A Chemically Synthesized Resveratrol Dimer is a Potent Inhibitor of Vascular Endothelial Growth Factor (VEGF)
Shengying Lin1, Maggie Suisui Guo1, Roy Wai-Lun Tang1, Yutong Ye1, Yuen Man HO1, Ran Duan1, Ka Wing Leung1, Jianlong Zhou2, Tina Ting-Xia Dong1, Karl Wah-Keung Tsim1*
1Center for Chinese Medicine, Division of Life Science, The Hong Kong University of Science and Technology, Clear Water Bay, Kowloon, Hong Kong, China
2Guangxi International Zhuang Medicine Hospital, Guangxi University of Chinese Medicine, 530200, Nanning, China
*Corresponding Author: Prof. Karl Wah-Keung Tsim, Center for Chinese Medicine, Division of Life Science, The Hong Kong University of Science and Technology, Clear Water Bay, Hong Kong, China.
Received: 06 February 2026; Accepted: 20 February 2026; Published: 20 March 2026
Article Information
Citation: Shengying Lin, Maggie Suisui Guo, Roy Wai-Lun Tang, Yutong Ye, Yuen Man HO, Ran Duan, Ka Wing Leung, Jianlong Zhou, Tina Ting-Xia Dong, Karl Wah-Keung Tsim. A Chemically Synthesized Resveratrol Dimer is a Potent Inhibitor of Vascular Endothelial Growth Factor (VEGF). Archives of Clinical and Biomedical Research. 10 (2026): 108-116.
View / Download Pdf Share at FacebookAbstract
Vascular endothelial growth factor (VEGF) plays a critical role in angiogenesis, influencing various physiological and pathological processes. Indeed, the inhibitors of VEGF have been developed as angiogenic medications, e.g. bevacizumab (avastin). Here, we aim to explore the synthesis and evaluation of a novel resveratrol dimer, named RE-16, as a potential anti-VEGF agent. By structural modifications of resveratrol, RE- 16 was synthesized using a chemical linker to dimerize the resveratrol scaffold, resulting in a compound with improved binding affinity to VEGF, as compared to its parent compound. The binding assays of RE-16 to VEGF, including ultrafiltration and Biacore surface plasmon resonance (SPR), demonstrated the superior affinity of RE-16 in comparing to resveratrol. In line with this physical binding, the biological evaluations using human umbilical vein endothelial cells (HUVECs) revealed that the treatment of RE-16 effectively inhibited the VEGF-mediated biological activities in dosedependent manners, exhibiting better potency than the parental molecule-- resveratrol. The favourable ADMET properties of RE-16 were also predicted through computational analysis, suggesting good intestinal absorption and low toxicity. Overall, RE-16 demonstrates promising potential as a therapeutic agent for the VEGF-mediated diseases, warranting further pharmacological development and clinical investigation.
Keywords
VEGF inhibitor; Computational docking; Angiogenesis; Resveratrol-type analogues; Wet age-related macular degeneration; Cancer
Article Details
1. Introduction
The vascular endothelial growth factor (VEGF) family, containing VEGF-A, VEGF-B, VEGF-C, plays a crucial role in regulating the formation of new blood networks [1-2]. VEGF family conserves a cysteine-knot fragment that enables dimerization and receptor binding to multiple VEGF receptors, including VEGFR1, VEGFR2 and VEGFR3. VEGF-A, acting as the most fundamental member in the family, exists in various isoforms, such as VEGF-A111, 121, 145, 165, 183, 189, and 206, of which VEGF-A165 appears as the most abundant isoform in nature [3-4]. Interestingly, VEGF-A165 comprises of an N-terminal receptor-binding domain and a C-terminal heparin-binding domain, which allows the subsequent interaction with extracellular matrix and systematic distribution. It is worth noting that VEGF-A165 usually binds to VEGFR2 with a KD constant of 1-10 nm, leading to the formation of homodimer at physiological level [5-6].
The VEGF-A/VEGFRs signalling pathway is vital in regulating various biological functions and diseases [7]. In mice with a knock-out VEGFR-1 gene, impaired blood vessel formation was observed, ultimately leading to cell death. Therefore, VEGF-A is a crucial component in the regulation of angiogenesis through its interaction with the VEGFR-2 receptor. Angiogenesis refers to the process of forming new blood vessels from the existing capillaries, which therefore results in the establishment of a complete, organized, and mature vascular network [8-9]. Angiogenesis primarily occurs during embryonic development, tissue repair, menstrual cycle, muscle growth, and the regeneration of organ linings. Indeed, this process is tightly regulated by the body. Additionally, angiogenesis plays a significant role in the progression of several malignant tumours, including melanoma, breast cancer, colorectal cancer, non-small cell lung cancer, and renal cell carcinoma [10-12]. Taken these together, VEGF-A is a critical protein factor in the regulation of angiogenesis through its interaction with the VEGFR-2 receptor, and such a pathway has appeared as an interesting drug target in modulating angiogenesis-mediated disease.
There have been a couple of reported inhibitors that are utilized to disrupt VEGF/VEGFR pathway, and which have been approved by FDA as drugs [13-15]. For instance, bevacizumab and ramucirumab, antibodies targeting to VEGF, have been employed as clinical anti-cancer therapeutics, while six of others have been approved to treat ocular vascular disease, such as pegaptanib, ramucirumab, brolucizumab and so on [13-14]. Nevertheless, the majority of these drugs often lead to side effects associated with high dose, off-target toxicity, relapse as well as drug resistance [14-15]. This inspired us to develop novel agents targeting VEGF-mediated disease and potentially serve as alternative options in clinical application.
Herbal medicines have been extensively used to treat various diseases, including VEGF-mediated diseases [16-17]. Through our previous screening platform, we have identified a few phytochemicals that expressed fundamental anti-VEGF efficacy [18-21]. Intriguingly, resveratrol and its analogous have been shown to bind to VEGF protein and attenuate VEGF-mediated angiogenic activities [18-21]. As a common polyphenol in grapes and vegetables, resveratrol has been previously identified as a clinically safe phytochemical with diverse functions [22-23]. Although resveratrol suppresses the VEGF-induced angiogenesis, the draggabilities of which are relatively low, including poor efficacy and low bioavailability due to poor aqueous solubility and rapid metabolism [24]. Here, we attempted to introduce a chemical linker and dimerized the resveratrol scaffold to generate novel resveratrol analogues. As a result, we were able to obtain RE-16 that displayed promising binding affinity to VEGF protein and expressed more significant effectiveness to VEGF-mediated biological activities than resveratrol. This evidently suggested that RE-16 could serve as a potent anti-VEGF agent for the subsequent pharmacological development.
2. Materials and Methods
2.1 Chemistry
All chemicals and solvents in use were obtained from commercial sources. NMR spectra (1H NMR and 13C NMR) was conducted by Bruker Advance 400 (1H: 400 MHz; 13C: 101 MHz) (Bruker, Billerica, MA, USA). The abbreviations for spin multiplicity are used as follows: s = singlet; d = doublet; t = triplet; m = multiplet. HRMS were provided through Agilent 6550 iFunnel QTOF.
2.2 Synthesis of RE-16
To a solution of pterostilbene (1 eq.) in THF (3 mL/mmol) was added NaH (1.1 equiv.) very slowly at 0°C, followed by the addition of 1,5-Dichloro-3-pentanone (0.5 equiv.) in THF (3 mL/mmol) dropwise. The reaction mixture was stirred at room temperature for 20 hours. The solvent was removed under vacuum, and the crude product was purified by column chromatography (EtOAc/petroleum ether) to yield the final product in 22% yield. 1H NMR (400 MHz, CDCl3) δ 2.90 (t, J = 6.3 Hz, 4H), 3.82 (s, 12H), 4.24 (t, J = 6.3 Hz, 4H), 6.37 (t, J = 2.0 Hz, 2H), 6.64 (d, J = 2.0 Hz, 4H), 6.81-6.91 (m, 6H), 7.01 (d, J = 16.0 Hz, 2H), 7.42 (d, J = 8.6 Hz, 4H). 1C NMR (101 MHz, CDCl3) δ 43.7 (2C), 54.6 (4C), 59.1 (2C), 99.1 (2C), 104.8 (4C), 113.9 (4C), 127.0 (4C), 129.1 (2C), 130.2 (4C), 140.1 (2C), 158.2 (2C), 160.8 (4C), 205.9 (1C). HRMS detection for [M+H]+: found 594.1475.
2.3 Biological evaluations
2.3.1 Materials
The reagents utilized in this report, including Dulbecco’s Modified Eagle Medium (DMEM), fetal bovine serum (FBS), and other related substances, were sourced from Thermo Fisher Scientific (Waltham, MA, USA). Various antibody species were procured from Cell Signaling Technology (CST; Beverly, MA, USA). VEGF-A165 was obtained from Haoyuan Chemexpress (Shanghai, China). Additionally, MTT [3-(4,5-dimethyl-2-thiazolyl)-2,5-diphenyl-2H-tetrazolium bromide] and the DCFH-DA probe (20,70-dichlorofluorescein diacetate) were obtained from Sigma-Aldrich (St. Louis, MO, USA).
2.3.2 Cell cultures
Human umbilical vein endothelial cells (HUVECs), deriving from endothelium of umbilical cord veins, macrophage RAW264.7 and Caco-2 cells, were obtained from American Type Culture Collection (ATCC, Manassas, VA, USA). During the cell culturing process, the cell lines were supplemented with DMEM culture medium, along with 10% fetal bovine serum (FBS) and 1% penicillin/streptomycin (100 U/mL and 100 μg/mL). The cells were maintained in a humidified CO2 incubator set to 5% CO2 and 37 °C. The cell lines were subculture when cell confluency exceeded 80%.
2.3.3 Binding affinity assay
Binding affinities between the analytes and VEGF-A165 protein were assessed using a Biacore T200 (Cytiva) following previous protocol [18]. Specifically, different concentrations of resveratrol and RE-16 were prepared by diluting in the running buffer (with tested concentrations of 1.25 to 20 µM) and flowed over the chip surface. The binding interactions between the VEGF protein and the tested ligands (either resveratrol or RE-16) were monitored in real time. A control group without immobilized VEGF protein was included as a blank. The data were subsequently analysed using the GE Biacore T200 control software.
2.3.4 Wound closure assay
Wound closure assay was conducted on cultured HUVECs to investigate cell migration in vitro, following the procedures outlined by Guo et al. [25]. In brief, HUVECs were plated in a 12-well plate at a density of 3 x 105 cells per well, and treatments were administered once the cells reached full confluence. After washing the cells with PBS, they were treated with the respective drugs. Images of each cell scrape were taken at 0 hours (At0) and 20 hours (At20) using a microscope and imaging software (Zen) at 10x magnification. The open area of each scrape image at different time points was analysed using the software TScratch (CSE lab, Switzerland). The wound recovery rate was quantified using the following equation: Wound closure (%) = (At0 – At20) / At20 x 100%.
2.3.5 SDS/PAGE and western blotting
The western blotting study was performed as previously described [26]. The samples were then analyzed using SDS-PAGE on a 10% gel. For blocking, a solution of 5% skim milk in Tris-buffered saline (pH 7.4) containing 0.1% Tween-20 (TBST) was utilized. The antibodies employed included anti-rabbit phospho-p44/42 MAPK, anti-rabbit p44/42 MAPK (CST), and an anti-rabbit horseradish peroxidase (HRP) secondary antibody (Sigma-Aldrich). ECL Western blotting substrate (Thermo Fisher Scientific) was used to visualize the blots, which were captured with the Chemidoc Touch Imaging System (Bio-Rad Laboratories, Hercules, CA, USA). The intensities of each band were quantified and analyzed using Image Lab software.
2.3.6 Cell proliferation
After incubating for 24 hours, 100 μL of VEGF at a concentration of 10 ng/mL, with or without various concentrations of analytes, was added to each well, followed by the addition of 10 μL of MTT solution (5 mg/mL). Both resveratrol and RE-16 were tested at concentrations of 0.1 to 10 µM. The mixture was incubated at 37 °C for 4 hours, after which 150 μL of DMSO was added to dissolve the formazan salt formed during the reaction. The samples were collected and measured using a microplate reader (Thermo Fisher Scientific) at a wavelength of 570 nm. Proliferation values were calculated using the following formula: proliferation (%) = (experimental value − low control) / (high control − low control) × 100%.
2.3.7 Computational docking study
The docking study was performed as previously described [26]. Specifically, the chemical structures of resveratrol and RE-16 were created using ChemDraw (version 20.0, https://perkinelmerinformatics.com/, accessed on 04-01-2026), while the protein structure of VEGF-A was downloaded from Protein Data Bank (PDB code: 1FLT, https://www.rcsb.org/, accessed on 04-01-2026). Residues 1-165 of VEGF were selected as the target domain for binding simulations. Virtual screening was conducted using SEESAR software (Version 14.0; https://www.biosolveit.de/, accessed on 04-01-2026). ADMET properties, including TPSA, Log P, and intestinal absorption, were predicted using the Optibrium mode.
2.3.8 Ultrafiltration
The binding affinity was assessed using a previously established method [26]. Specifically, the tested ligands at a concentration of 2 µM were mixed with or without 2 nM VEGF protein (MCE, Monmouth Junction, USA) and incubated in Milli-Q water at 4°C for 2 hours. The mixture was then transferred to a 0.5 mL ultrafiltration tube (2000 MW cutoff; Sartorius Stedim Biotech, Göttingen, Germany). After several rounds of centrifugation, the unfiltered mixture containing both the protein and the tested compound was precipitated with acetonitrile, and the supernatant was further analysed by HPLC at a wavelength of 210 nm.
3. Results
3.1 Synthesis of RE-1
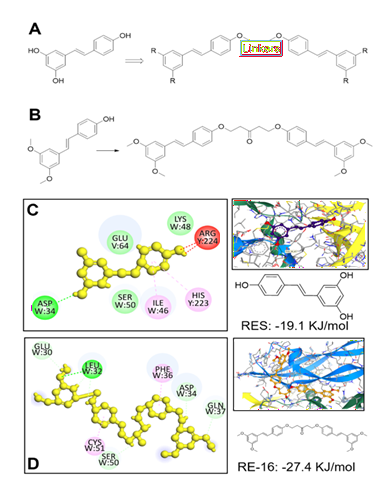
As reported previously, resveratrol was able to bind the active site of VEGF-A protein and acted as effective anti-VEGF agent [18-19]. The para-phenol fragment of resveratrol appears synthetically available, allowing us to introduce various building blocks to yield a group of novel resveratrol analogues with improved efficacy and bioavailability (Figure 1A). To begin with, we proposed to introduce a linker to the para-phenol scaffold connecting with another resveratrol in forming a dimer of resveratrol. Such a dimer was expected to show stronger binding affinity at the active site and display potent anti-VEGF effectiveness. In light of this, an analogue, named RE-16, was designed and synthesized for the following evaluation. As shown in Figure 1B, 1,5-dichloro-3-pentanone reagent was employed to introduce a pentanone linker through a one-pot reaction in the presence of NaH. The reaction went well, and we were able to obtain pure RE-16 compound, albeit the final yield was relatively low at ~22%. In our initial evaluation, we conducted a docking analysis and observed that both resveratrol and RE-16 were able to bind the active site and formed van der Waal interactions with SER50 residue of VEGF-A (Figure 1C and D). Interestingly, RE-16 showed lower docking energy at -27.4 KJ/mol vs -19.1 KJ/mol of resveratrol, indicating an increase of binding affinity to the protein and more robust anti-VEGF activity.
Figure 1: (A) Structural design of novel resveratrol analogue. Introduction of linkers into para-phenol scaffold leads to a dimerization of resveratrol. (B) Synthetic scheme towards RE-16. Reagents and conditions: NaH, 1,5-dichloro-3-pentanone, THF, 0 °C-RT, 20 hours, 22%. (C) Docking analysis of resveratrol (RES) against VEGF-A protein (PDB code: 1FLT). (D) Docking analysis of RE-16 against VEGF-A protein. Chemical structures were obtained from Chemdraw software, and protein structure was obtained from protein data bank.
3.2 Binding activity to VEGF
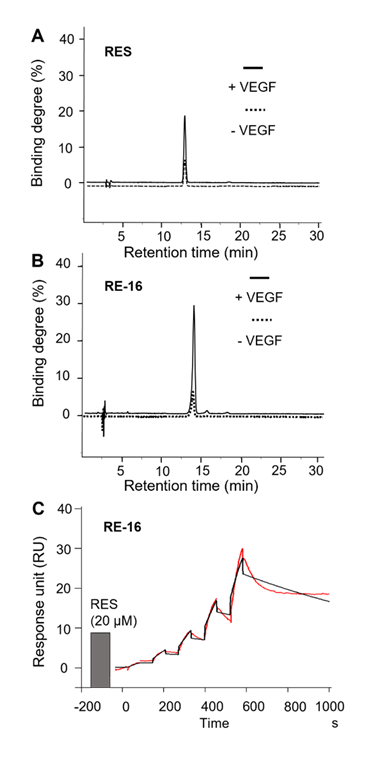
To further determine the binding affinity of RE-16 against VEGF-A, we developed a series of VEGF-based screening platforms. In an ultrafiltration assay, RE-16 could significantly bind to VEGF-A, and the binding degree was higher than that of resveratrol (30% vs 19%), suggesting that the novel dimer had better affinity to VEGF-A than its parental phytochemical (Figure 2A and B). In a Biacore SPR assay, we found that RE-16 displayed fundamental binding performance to VEGF-A and showed significantly higher response units than resveratrol at concentration of 20 mM, which was in consistence with the observation in the ultrafiltration study (Figure 2C).
Figure 2: Binding performances of resveratrol (RES) (A) and RE-16 (B) in the ultrafiltration assay. Binding degree was determined through HPLC analysis at wavelength of 210 nm. (C) Binding activity of RE-16 in Biacore assay at concentrations up to 20 µM. The binding interactions were monitored in real time and analysed using the GE Biacore T200 control software.
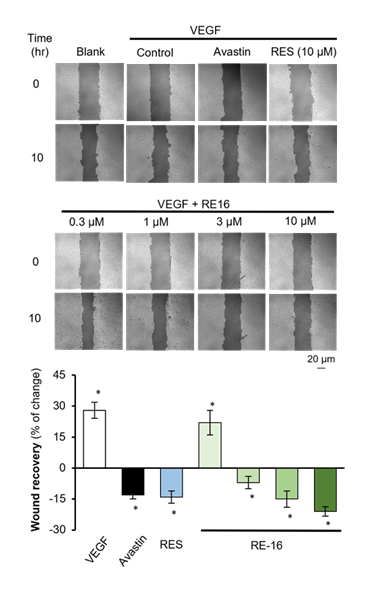
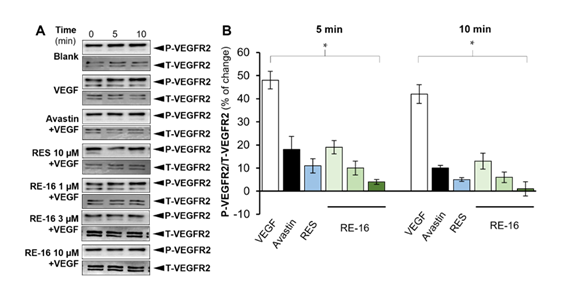
HUVEC is an endothelial cell line deriving from peripheral vasculature and has been widely utilized in VEGF-related vascular research [27]. During our investigation, we established a HUVEC culture model and found that VEGF-A triggered efficient wound recovery, as compared with the blank group (Figure 3). Subsequently, avastin (bevacizumab), a well-known inhibitor of VEGF-A, was employed as a control and attenuated the wound recovery induced by VEGF-A. When co-treated with VEGF-A, RE-16 was able to significantly reduce the efficiency in dose-dependent manners from 0.1 µM to 10 µM, and the inhibitory effectiveness was stronger than resveratrol. In the following western blotting study, RE-16 robustly blocked the VEGF-induced phosphorylation of VEGFR2 from 0.1 µM to 10 µM and expressed better potency than avastin and/or resveratrol (Figure 4).
Figure 3: RE-16 attenuated wound recovery induced by VEGF-A in HUVECs from 0.3 µM to 10 µM. Pictures of cell scrape at 0 and 10 hours were taken by a microscope and further interpreted by imaging software (Zen). The asterisks statistically represent significant differences such that * p < 0.05 in comparison with the blank group (no drug treatment) in mean ± SD, n = 4.
Figure 4: (A) RE-16 inhibited interaction between VEGF-A and VEGFR2 by blocking phosphorylation of VEGHR2 in HUVECs from 1, 3 and 10 µM in western blotting assay. (B) The intensities of each band were interpreted by software Image Lab (6.1 version). Values were calculated as the percentage of changes in comparison with blank group (no drug treatment). The asterisks statistically represent significant differences such that * p < 0.05 in comparison with the blank group (medium only) in mean ± SD, n = 4.
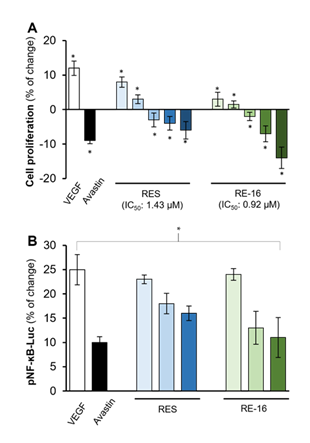
Figure 5: (A) RE-16 and resveratrol (RES) reduced VEFG-mediated cell proliferation in HUVECs from 0.1 µM to 10 µM. Avastin was used as a positive control at a concentration of 200 μg/mL. Values were calculated as the percentage of changes in comparison with blank group (no drug treatment). (B) RE-16 and resveratrol (0.1, 1 and 10 µM) suppressed NF-κB activities mediated by VEGF-A. RAW 264.7 cells were transfected with pNF-κB (Thermo Fisher Scientific) through jetPRIME before being incubated for 24 hours. The refresh medium was replaced every four hours prior to drug treatments twice after 24 and 48 hours, respectively. The luminescent intensity was interpreted by luminometer. The asterisks statistically represent significant differences such that * p < 0.05 in comparison with the blank group (medium only) in mean ± SD, n = 4.
In parallel, RE-16 showed effective inhibition to the VEGF-induced proliferation of HUVECs in a dose-dependent manner (Figure 5A), and the IC50 value was lower than that of resveratrol (0.92 µM vs 1.43 µM), suggested that RE-16 was a more potent anti-VEGF agent than resveratrol. Furthermore, it was previously reported that VEGF-A, after binding to its corresponding receptor, such as VEGFR2, activated the downstream signalling factors, including NF-ĸB, and therefore mediated the inflammation-regulated angiogenesis [28-29]. Indeed, VEGF-A was found to enhance the expression of NF-ĸB, and the application of avastin could effectively reduce the activity of NF-ĸB in the presence of VEGF-A. As anticipated, RE-16 displayed better suppression to NF-ĸB by inhibiting VEGF-A from 0.1 µM to 10 µM than resveratrol (Figure 5B), which agreed with the observation above. Taken together, these evidently indicated RE-16 could act as a promising anti-VEGF agent and impede the VEGF-mediated downstream biological activities through disrupting its correlation with VEGFR2.
3.3 Pharmacological profile of RE-16
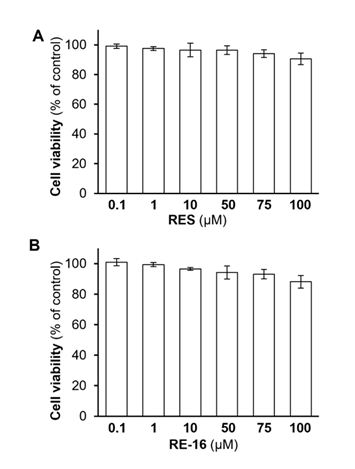
To outline the pharmacological draggability of RE-16, we first conducted an MTT assay in HUVECs to determine the potential toxicity of RE-16. As shown in Figure S1, no obvious cell apoptosis was observed from RE-16 group when treated at relatively low concentrations, implying that the reagent was unlikely to induce toxicity in cultured cells. In addition, we performed a stability test in three conditions, i.e. acidic, basic and neutral, and revealed that RE-16 was relatively stable under neutral and acidic condition (RSD < 10%), despite that very mild degradation was detected under basic condition (Tabel 1). Besides, it was observed in a Caco-2 cell permeability assay that RE-16 was able to penetrate through the assay with a Papp value of 16.4 × 10-6 cm/s at a concentration of 10 µM (Figure 6A), which was anticipated to display good bioavailability and absorption according to Ahmed et al [30] (Table 1).
|
Analytes |
Precision |
|||
|
Intra-day (n=6) a |
Inter-day (n=6) b |
|||
|
Mean Peak Area c |
RSD (%) |
Mean Peak Area |
RSD (%) |
|
|
RE-16 (acidic) |
1347.1 |
4.71 |
1254.9 |
5.07 |
|
RE-16 (neutral) |
1975.5 |
3.99 |
2032.7 |
7.97 |
|
RE-16 (basic) |
945.3 |
6.48 |
994.7 |
10.06 |
Table 1: Stability of RE-16 and resveratrol in HPLC analysis. The stability was determined by the intra-day and inter-day deviations through HPLC analysis at a wavelength of 210 nm. The relative standard deviation (RSD) was used as an indicator of stability. a b the intra- and inter-day stability tests were determined by analysing 6 replicates of the standard solution of the two analytes during a single day and six replicates of the samples examined on 5 successive days, respectively; c mean peak area was calculated from the average number of 6 replicates.
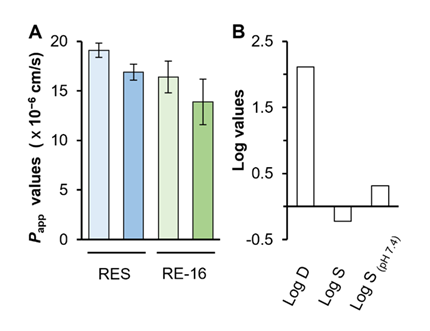
Figure 6: (A) RE-16 expressed promising permeability in Caco-2 cells. The integrity of the cell monolayer was assessed by measuring transepithelial electrical resistance (TEER) with an EVOM Epithelial Volt/Ohm Meter (WPI, Sarasota, FL, USA), along with evaluating the permeability of lucifer yellow (a marker for paracellular leakage) across the cell monolayer. The permeabilities values were determined through mass spectrometry. Both resveratrol and RE-16 were tested at concentration of 1 µM and 10 µM. Values were calculated as percentage of changes compared with the blank control (only medium in use), in mean ± SD, n = 4. Papp value was calculated from the following equation: ; standard deviation (SD) was calculated from four replicates. (B) In silico prediction of Log D and Log S values. Log D and Log S values were obtained from SEESAR software (Version 14.0; https://www.biosolveit.de/, accessed on 04-01-2026) using the Optibrium mode (Table 2).
|
Analyte |
MW (Da) |
TPSA (< 140 Å2) |
Log P (< 5) a |
BBB (−) |
CYPs Toxicity b |
HIA (+) c |
P-gp (−) d |
|
RES |
228.2 |
60.7 |
2.97 |
− |
Medium |
+ |
− |
|
RE-16 |
706.7 |
140.7 |
7.66 |
− |
Medium |
+ |
− |
Table 2: ADMET profile of resveratrol and RE-16. Data was obtained through Optibrium mode in SEESAR software. a Lipinski's Rule of Five; b potential toxicity against cytochrome P450 enzymes (CYPs); c HIA: human intestinal absorption (over 30%). d P-gp: P-glycoprotein, an ATP-dependent active transporter.
Next, we investigated the ADMET profile of RE-16 through computational analysis. As a result, RE-16 was found to display good intestine absorption (positive HIA value) and relatively low toxicity to CYPs, and was expected to show no interaction with BBB (blood brain barrier) as well as P-gp that led to active transportation and low bioavailability, although slightly higher Lop P was recorded (Log P > 5) (Table 2). Also, the Log D, Log S and Log S pH 7.4 values were determined as 2.11, -0.221 and 0.316, respectively (Figure 6B), which were believed to provide crucial balance between lipophilicity and hydrophilicity and therefore result in promising bioavailability [31-32].
4. Discussions
Angiogenesis plays an important role in several diseases’ progression, such as malignant tumours and retinopathies. The VEGF/VEGFR axis, including VEGF/VEGFR interaction, VEGFR tyrosine kinase phosphorylation and VEGFR receptor downstream signalling, is the key process of angiogenesis [33-34]. This process involves the activation, proliferation, and migration of endothelial cells, mediated by a variety of pro-angiogenic and anti-angiogenic factors. In healthy adults under normal physiological conditions, endothelial cells remain largely inactive, with a mitotic frequency of only 0.5% [33-34]. In several non-neoplastic angiogenic diseases, such as immune disorders, e.g., rheumatoid arthritis, psoriasis, and Crohn's disease, diabetic retinopathy, and age-related macular degeneration (AMD), angiogenesis can become dysregulated and excessive due to the overexpression of pro-angiogenic factors and/or the inactivation of anti-angiogenic factors [34-35].
Ever since the hypothesis of neovascularization, as proposed in 1970s, the anti-angiogenic therapeutic has become a major strategy to tackle angiogenesis-mediated diseases, including cancer and AMD [36-37]. The combination of VEGFs and VEGFRs has led to the activation of various receptors and eventually resulted in angiogenesis and lymphangiogenesis, and such an interaction has attracted considerable attention as the main target of anti-angiogenesis therapy [36-37]. There have been a few anti-angiogenic drugs being approved by the FDA, which have been divided into three classes, i.e., antibody, fusion protein, and small molecule [38-40]. So far, totally seven drugs targeting VEGFs/VEGFRs pathway have been approved by the FDA to treat cancer and ocular vascular disease, and none of them are small molecules. It is worth noting that, in general, antibody and fusion proteins contain large molecular size and display poor penetration into cell membrane and low bioavailability, which leads to off-target side effect and safety issue in clinical applications [38-40].
Phytochemicals from natural products have been widely utilized in clinical applications and have gained significant attention for their potential efficacy against various disease, including angiogenesis-mediated disease [41]. For instance, baicalin, from Chinese skullcap, has been employed as an effective cancer treatment by suppressing VEGF-regulated pathway. In parallel, curcumin was found to inhibit cancer growth through blocking TNF-α and angiogenesis-related activities [41]. Despite these successes, it has remained extremely challenging to transform natural occurring phytochemicals into medicinal therapeutics, and this is mainly due to the challenges of large-scale isolation and relatively low efficacy [41-42]. To develop as drugs for clinical applications, the chemical modification of these phytochemicals is a prerequisite.
Computational docking has been an efficient in silico tool utilized in drug discovery [26,43], which determines the protein binding domain responsible for ligand binding and reveals the affinity at the targeted protein. Besides, the computational model could also be used to predict ADMET properties and outline the pharmacological profile of newly designed chemical analogues prior to biological evaluations [26,43]. In our previous investigation, we have identified resveratrol as a potent anti-VEGF and appeared as a promising hit molecule for the following hit-to-lead drug development [18-21]. This inspired us to conduct structural modifications around resveratrol with the aid of computational analysis to further identify novel molecules with better potency and potentially lower toxicity. Indeed, during our investigation, we have identified a novel resveratrol dimer, RE-16, that expressed potent efficacy to VEGF-mediated activity and could be considered as an effective anti-VEGF inhibitor for the subsequent pharmaceutical development.
5. Conclusion
In summary, we have successfully synthesized and evaluated RE-16, a novel resveratrol dimer, as a potential anti-VEGF agent. The structural modifications made to the resveratrol scaffold, particularly the introduction of a chemical linker to create a dimerized compound, which significantly enhanced its binding affinity to VEGF-A. Binding assays demonstrated that RE-16 exhibits superior affinity and potency in inhibiting VEGF-mediated biological activities, as evidenced by its effectiveness in reducing cell proliferation and migration in HUVECs. Furthermore, RE-16 was shown to inhibit the phosphorylation of VEGFR2 and suppress the downstream signalling pathways associated with angiogenesis, including NF-κB activation. The compound's favourable ADMET properties, predicted through computational analysis, suggested that it has the potential for good intestinal absorption and low toxicity, making it a promising candidate for the development of pharmacological products. Future studies should focus on the in vivo efficacy and safety of RE-16, as well as its potential for clinical application in treating VEGF-mediated diseases. Overall, RE-16 could serve as a valuable addition to the toolbox of anti-angiogenic agents available for pharmaceutical development and clinical use.
6. Data Statement
All data is available from corresponding author upon reasonable request.
7. Funding Source
This work was supported by Zhongshan Municipal Bureau of Science and Technology (2019AG035); Hong Kong Innovation Technology Fund (ITCPD/17-9); TUYF19SC02, GBA Institute of Collaborate Innovation (GICI-022); Hong Kong RGC-GFC 16100921; Hong Kong RGC Theme-based Research Scheme (T13-605/18-W); PD18SC01 and HMRF18SC06; and Shenzhen Science and Technology Innovation Committee (ZDSYS201707281432317); The Key-Area Research and Development Program of Guangdong Province (2020B1111110006); Special Project of Foshan University of Science and Technology in 2019 (FSUST19-SRI10).
8. Authorship Contribution Statement
Shengying Lin: Writing – original draft, Visualization, Validation, Methodology, Visualization; Maggie Suisui Guo: Methodology; Roy Wai-Lun Tang: Software; Yutong Ye: Formal analysis; Yuen Man HO: Investigation; Ran Duan: Resources; Ka Wing Leung: Project administration, Validation; Jianlong Zhou: Investigation; Tina Ting-Xia Dong: Funding acquisition; Karl Wah-Keung Tsim: Supervision, Conceptualization, Writing – review & editing.
9. Declaration of Competing Interest
All authors declare no competing interest.
References
- Shibuya M. Structure and function of VEGF/VEGF-receptor system involved in angiogenesis. Cell Struct Funct 26 (2001): 25-35.
- Brozzo MS, Bjelic S, Kisko K, et al. Thermodynamic and structural description of allosterically regulated VEGFR-2 dimerization. Blood 119 (2012): 1781-1788.
- Iyer S, Darley PI, Acharya R. Structural insights into the binding of vascular endothelial growth factor-B by VEGFR-1D2. J Biol Chem 285 (2010): 23779-23789.
- Park J, Keller G, Ferrara N. The Vascular Endothelial Growth Factor (VEGF) isoforms: differential deposition into the subepithelial extracellular matrix and bioactivity of extracellular matrix-bound VEGF. Mol Biol Cell 4 (1993): 1317-1326.
- Grünewald F, Prota A, Giese A, et al. Structure-function analysis of VEGF receptor activation and the role of coreceptors in angiogenic signaling. Biochim Biophys Acta 1804 (2010): 567-580.
- Park SA, Jeong MS, Ha K, et al. Structure and function of vascular endothelial growth factor and its receptor system. BMB Rep 51 (2018): 73-78.
- Potente M, Gerhardt H, Carmeliet P. Basic and therapeutic aspects of angiogenesis. Cell 146 (2011): 873-887.
- Ho V, Fong G. Vasculogenesis and angiogenesis in VEGF receptor-1 deficient mice. Methods Mol Biol 1332 (2015): 161-176.
- Ball C, Shuttleworth A, Kielty C. Vascular endothelial growth factor can signal through platelet-derived growth factor receptors. J Cell Biol 177 (2007): 489-500.
- Callan A, Heckman J, Tah G, et al. VEGF in diabetic retinopathy and age-related macular degeneration. Int J Mol Sci 26 (2025): 4992.
- Shahidatul-Adha M, Zunaina E, Aini-Amalina M. Evaluation of vascular endothelial growth factor (VEGF) level in the tears and serum of age-related macular degeneration patients. Sci Rep 12 (2022): 4423-4431.
- Ghalehbandi S, Yuzugulen J, Pranjol ZI, et al. The role of VEGF in cancer-induced angiogenesis and research progress of drugs targeting VEGF. Eur J Pharmacol 949 (2023): 175586.
- Elsheikh RH, Chauhan MZ, Sallam AB. Current and novel therapeutic approaches for treatment of neovascular age-related macular degeneration. Biomolecules 12 (2022): 1629.
- Qi S, Deng S, Lian Z, et al. Novel drugs with high efficacy against tumor angiogenesis. Int J Mol Sci 23 (2022): 6934.
- Lytvynchuk L, Sergienko A, Lavrenchuk G, et al. Antiproliferative, apoptotic, and autophagic activity of ranibizumab, bevacizumab, pegaptanib, and aflibercept on fibroblasts: implication for choroidal neovascularization. J Ophthalmol (2015): 934963.
- Fei XJ, Zhang X, Wang Q. Xijiao Dihuang Decoction alleviates ischemic brain injury in MCAO rats by regulating inflammation, neurogenesis, and angiogenesis. Evid Based Complement Alternat Med (2018): 5945128.
- Mao Y, Meng L, Liu H, et al. Therapeutic potential of traditional Chinese medicine for vascular endothelial growth factor. J Zhejiang Univ Sci B 23 (2022): 353-364.
- Hu WH, Duan R, Xia YT, et al. The binding of resveratrol to vascular endothelial growth factor (VEGF) suppresses angiogenesis by inhibiting the receptor signalling. J Agric Food Chem 67 (2019): 1127-1137.
- Hu WH, Chan GK, Duan R, et al. Synergy of ginkgetin and resveratrol in suppressing VEGF-induced angiogenesis: a therapy in treating colorectal cancer. Cancers (Basel) 11 (2019): 1828.
- Hu WH, Wang HY, Kong XP, et al. Polydatin suppresses VEGF-induced angiogenesis through binding with VEGF and inhibiting its receptor signaling. FASEB J 33 (2019): 532-544.
- Hu WH, Zhang XY, Leung KW, et al. Resveratrol, an inhibitor binding to VEGF, restores the pathology of abnormal angiogenesis in retinopathy of prematurity (ROP) in mice: Application by intravitreal and topical instillation. Int J Mol Sci 23 (2022): 6455.
- Kawada N, Seki S, Inoue M, et al. Effect of antioxidants, resveratrol, quercetin, and N-acetylcysteine, on the functions of cultured rat hepatic stellate cells and kupffer cells. Hepatology 27 (1998): 1265-1274.
- Jang M, Cai L, Udeani GO, et al. Cancer chemopreventive activity of resveratrol, a natural product derived from grapes. Science 175 (1997): 218-220.
- Chen X, Song X, Zhao X, et al. Insights into the anti-inflammatory and antiviral mechanisms of resveratrol. Mediators Inflamm (2022): 7138756.
- Guo MS, Gao X, Hu W, et al. Scutellarin potentiates the skin regenerative function of self growth colony, an optimized platelet-rich plasma extract, in cultured keratinocytes through VEGF receptor and MAPK signaling. J Cosmet Dermatol 21 (2021): 4836-4845.
- Lin S, Tang R, Ye Y, et al. Drug screening of flavonoids as potential VEGF inhibitors through computational docking and cell models. Molecules 30 (2025): 257.
- Medina-Leyte D, Domínguez-Pérez M, Mercado I, et al. Use of human umbilical vein endothelial cells (HUVEC) as a model to study cardiovascular disease: a review. Appl Sci 10 (2020): 938.
- Tu J, Fang Y, Han D, et al. Activation of nuclear factor-κB in the angiogenesis of glioma: Insights into the associated molecular mechanisms and targeted therapies. Cell Prolif 54 (2021): e12929.
- Dabravolski SA, Khotina VA, Omelchenko AV, et al. The role of the VEGF family in atherosclerosis development and its potential as treatment targets. Int J Mol Sci 23 (2022): 931.
- Ahmed I, Leach D, Wohlmuth H, et al. Caco-2 cell permeability of flavonoids and saponins from Gynostemma pentaphyllum: The immortal herb. ACS Omega 5 (2020): 21561-21569.
- Stienstra C, Ieritano C, Haack A, et al. Bridging the gap between differential mobility, Log S, and Log P using machine learning and SHAP analysis. Anal Chem 95 (2023): 10309-10321.
- Duan Y, Fu L, Zhang X, et al. Improved GNNs for Log D7.4 prediction by transferring knowledge from low-fidelity data. J Chem Inf Model 63 (2023): 2345-2359.
- Apte R, Chen D, Ferrara N. VEGF in signaling and disease: beyond discovery and development. Cell 176 (2019): 1248-1264.
- Little K, Ma JH, Yang N, et al. Myofibroblasts in macular fibrosis secondary to neovascular age-related macular degeneration: the potential sources and molecular cues for their recruitment and activation. EBioMedicine 38 (2018): 283-291.
- Yang Y, Cao Y. The impact of VEGF on cancer metastasis and systemic disease. Semin Cancer Biol 86 (2022): 251-261.
- Pober JS, Sessa WC. Evolving functions of endothelial cells in inflammation. Nat Rev Immunol 7 (2007): 803-815.
- Patel N, Ohbayashi M, Nugent AK, et al. Circulating anti-retinal antibodies as immune markers in age-related macular degeneration. Immunology 115 (2005): 422-430.
- Elebiyo T, Rotimi D, Evbuomwan I, et al. Reassessing vascular endothelial growth factor (VEGF) in anti-angiogenic cancer therapy. Cancer Treat Res Commun 32 (2022): 100620.
- Escalante C, Zalpour A. Vascular endothelial growth factor inhibitor-induced hypertension: basics for primary care providers. Cardiol Res Pract (2011): 816897.
- Wang L, Liu WQ, Broussy S, et al. Recent advances of anti-angiogenic inhibitors targeting VEGF/VEGFR axis. Front Pharmacol 14 (2024): 1307860.
- Soumya SJ, Arya KR, Abhinand CS, et al. Multi-target and natural product-based multi-drug approach for anti-VEGF resistance in glioblastoma. Explor Drug Sci 2 (2024): 567-582.
- Abu-Reidah I, Taamalli A. Promising phytoconstituents in antiangiogenesis drug development. Nutraceuticals 4 (2024): 450-468.
- Spagnolli G, Massignan T, Astolfi A, et al. Pharmacological inactivation of the prion protein by targeting a folding intermediate. Commun Biol 4 (2021): 62-77.
Supplementary File:
Figure S1: Cell viability was assessed with MTT assay. HUVECs were seeded onto 96-well plates for 24 h, prior to drug treatments. After incubations for 24 h, a MTT solution in final concentration of 0.5 mg/mL was added to each well. Following another 3 h of incubation, DMSO solvent was used to dissolve the purple formazan crystals generated in each well. The absorbance of the samples at 570 nm was measured using a microplate reader (Thermo Fisher Scientific). The cell viability was calculated as follow: cytotoxicity (%) = (experimental value − low control) / (high control−low control) × 100%.


 Impact Factor:
* 5.8
Impact Factor:
* 5.8
 Acceptance Rate:
71.20%
Acceptance Rate:
71.20%
 Time to first decision: 10.4 days
Time to first decision: 10.4 days
 Time from article received to acceptance:
2-3 weeks
Time from article received to acceptance:
2-3 weeks
