Efficacy and Safety of Perioperative Analgesia Provided by Ultrasound Guided Erector Spinae Plane Block (ESPB) Compared to Continuous Intercostal Wound Infiltration (IC-WI) in Minimally Invasive Mitral Valve Surgery (MICS-MVR)
Martin Roesslein1, Yasir Al-Hamami2, Thore Kalinowski1, Vincent Effner1, Wolfgang Bothe2, Axel Semmelmann1, Torsten Loop1*, Felix Ulbrich1
1Department of Anesthesiology and Critical Care, Medical Center-University of Freiburg, Faculty of Medicine, University of Freiburg, 79106 Freiburg, Germany
2Department of Cardiovascular Surgery, Heart Centre Freiburg, University of Freiburg, Faculty of Medicine, University of Freiburg, 79106 Freiburg, Germany
*Corresponding Author: Torsten Loop MD, Professor of Anesthesia, Department of Anesthesiology and Critical Care, Medical Center-University of Freiburg, Faculty of Medicine, University of Freiburg Hugstetter Strasse 55, 79106 Freiburg, Germany.
Received: 18 March 2026; Accepted: 25 March 2026; Published: 16 April 2026
Article Information
Citation: Martin Roesslein, Yasir Al-Hamami, Thore Kalinowski, Vincent Effner, Wolfgang Bothe, Axel Semmelmann, Torsten Loop, Felix Ulbrich. Efficacy and Safety of Perioperative Analgesia Provided by Ultrasound Guided Erector Spinae Plane Block (ESPB) Compared to Continuous Intercostal Wound Infiltration (IC-WI) in Minimally Invasive Mitral Valve Surgery (MICS-MVR). Anesthesia and Critical Care. 8 (2026): 54-61.
View / Download Pdf Share at FacebookAbstract
Purpose: To compare postoperative analgesic efficacy and opioid consumption between erector spinae plane block (ESPB) and continuous intercostal wound infiltration (IC-WI) in patients undergoing minimally invasive mitral valve surgery (MICS-MVR).
Patients and methods: In this retrospective registry study, patients undergoing MICS-MVR received either an ultrasound-guided ESPB at anesthesia induction or surgeon-administered continuous IC-WI at wound closure. All patients received standardized multimodal analgesia. The primary outcome was cumulative morphine consumption during the first 72 hours postoperatively. Secondary outcomes included pain during coughing assessed using the Numeric Rating Scale (NRS), extubation in the operating room, opioid-related adverse events, and early mobilization.
Results: A total of 221 patients were included in the analysis (ESPB: n = 113; IC-WI: n = 108). Mean cumulative morphine consumption over 72 hours did not differ significantly between groups (IC-WI: 151±10.3 mg vs. ESPB: 176±9.3 mg; p=0.07). Patients in the IC-WI group reported significantly lower NRS pain scores immediately after surgery compared with the ESPB group (3.1±0.3 vs. 4.3±0.3; p<0.003). Pain scores were comparable during the subsequent postoperative course. A significantly higher proportion of patients in the ESPB group were extubated in the operating room (37/113; p<0.001).
Conclusion: Ultrasound-guided ESPB provides postoperative analgesia almost comparable to IC-WI in patients undergoing MICS-MVR. ESPB was associated with lower intraoperative sufentanil requirements and a higher likelihood of immediate endotracheal extubation in the operating room. These findings support ESPB as an effective regional analgesic technique in minimally invasive cardiac surgery and as a potential component of Enhanced Recovery After Cardiac Surgery pathways.
Keywords
Truncal blocks; Continuous wound analgesia; Minimally invasive cardiac surgery; Pain management; ERACS
Article Details
Introduction
Postoperative pain after thoracic and cardiac surgery, including coronary artery bypass grafting (CABG) and minimally invasive mitral valve repair or replacement (MICS-MVR), is often severe. Inadequate pain control causes significant patient discomfort, may delay functional recovery, and is associated with an increased risk of postoperative pulmonary complications and prolonged hospital stay [1-3].
Postoperative pain is multifactorial and results from surgical trauma related to sternotomy or thoracotomy, the presence of intercostal chest drains, and other perioperative factors. Over the past decade, cardiac surgical techniques have increasingly shifted toward less invasive approaches. In parallel, less invasive regional anesthetic techniques have gained importance and have been incorporated into Enhanced Recovery After Cardiac Surgery (ERACS) protocols [4,5].
Although neuraxial regional anesthesia techniques such as thoracic epidural analgesia and thoracic paravertebral block are well established and provide effective analgesia, their use in cardiac surgery is limited due to the need for systemic anticoagulation. Consequently, alternative regional techniques with a more favorable safety profile are increasingly being explored.
In recent years, several ultrasound-guided chest wall blocks—including the erector spinae plane block (ESPB), serratus anterior plane block (SAPB), and pectoral nerve blocks (PECS)—have been described and investigated for analgesia in thoracic procedures, including cardiac surgery [6,7]. The ultrasound-guided ESPB was first described by Forero et al. [8] in 2016 and targets the interfascial plane deep to the erector spinae muscle [8]. Owing to its technical simplicity, easily identifiable sonographic landmarks, and its presumed safety in anticoagulated patients, ESPB has become one of the most commonly applied regional analgesic techniques in cardiac surgery, although high-quality evidence supporting its safety in this setting remains limited [9-11].
Intercostal nerve block or continuous intercostal wound infiltration (IC-WI) has also been shown to provide effective postoperative analgesia after thoracic surgery and can be readily performed by the surgeon, but has numerous disadvantages, such as a limited duration of analgesia, variable efficacy, the risk of systemic toxicity from local anesthetics, technical challenges and complications, as well as infections and impaired wound healing [12-16].
The hypothesis of this study was that a preoperative ESPB in patients undergoing MICS-MVR provides postoperative pain relief that - as measured by pain scores and cumulative opioid consumption in the first 72 hours after surgery - is virtually comparable to that of continuous IC-WI. Furthermore, we hypothesized that preoperative ESPB is associated with lower intraoperative opioid consumption, fewer postoperative complications, and a higher rate of immediate extubation in the operating room.
The primary endpoint was cumulative postoperative morphine consumption over a 72-hour period. Secondary endpoints included postoperative pain intensity, opioid-related adverse effects (e.g., postoperative nausea and vomiting), surgical characteristics (including duration of chest tube drainage and need for revision surgery), and early extubation.
2. Material and Methods
Participant recruitment and group allocation
This retrospective registry study was approved by the local ethics committee of the University Hospital (reference number AZ 25-1138-S1-retro) and registered in the German Clinical Trials Register (DRKS, ID DRKS00033343). The study was based on a retrospective cohort of patients who underwent minimally invasive mitral valve repair or replacement (MICS-MVR) via an anterolateral thoracotomy between May 2022 and October 2023.
Eligible patients were retrospectively identified from the hospital’s electronic medical records using International Classification of Diseases (ICD) and Operations and Procedures (OPS) codes. Following the introduction of the erector spinae plane block (ESPB) into routine anesthetic practice at our institution in late 2022, ESPB was offered to all eligible patients without contraindications after obtaining written informed consent. Patients who did not receive ESPB during the study period were treated according to the standard surgical analgesic protocol and underwent continuous intercostal wound infiltration (IC-WI) with catheter placement.
Accordingly, group allocation was determined by routine clinical practice and procedural availability rather than randomization, consistent with the retrospective observational design of the study. ESPB was performed by the attending anesthesiologist, whereas IC-WI catheters were placed intraoperatively by the surgical team as part of standard care.
This resulted in a total cohort of 232 patients who underwent MICS-MVR at our single center. Data were retrieved retrospectively from the hospital information system PATIDOK® (version 2.0, PCS Professional Clinical Software GmbH, Klagenfurt, Austria) of the University Heart Center. Patient-related information, including pre-existing medical conditions, regular medication, exposure to noxious substances, and externally initiated therapeutic measures, was extracted from admission records. Additional demographic data (e.g., age and body weight) as well as information on conservative treatments were obtained from the same source. Clinical and perioperative data were extracted from electronic anesthesia records (Metavision®, version MV5, IMDsoft GmbH, Düsseldorf, Germany) and transferred in anonymized form to a pre-structured Microsoft Excel® database. Access to sensitive data and analyses was restricted to designated in-house computers in accordance with institutional data protection policies.
Anesthetic management
All patients received standardized intraoperative monitoring, including continuous electrocardiography, pulse oximetry, invasive arterial blood pressure monitoring (arterial line placed prior to induction), and depth of anesthesia monitoring using the bispectral index (BIS). General anesthesia was induced intravenously with sufentanil (0.3–0.5 µg·kg-¹), propofol (1–2 mg·kg-¹), and rocuronium (0.4–0.6 mg·kg-¹). Anesthesia was maintained with sevoflurane, titrated to achieve a BIS target range of 40–60, while maintaining heart rate and arterial blood pressure within ±20% of baseline values.
Central venous access was established via the right or left internal jugular vein, and transesophageal echocardiography was routinely performed intraoperatively.
Additional intraoperative boluses of sufentanil were administered at the discretion of the attending anesthesiologist, based on hemodynamic responses and clinical judgment. Postoperatively, analgesic management followed the institutional standard protocol. Pain intensity was routinely assessed using the Numeric Rating Scale (NRS), and rescue analgesia was administered if NRS scores were ≥3. The standardized multimodal analgesic regimen comprised systemic opioids in combination with non-opioid analgesics, such as paracetamol or metamizole.
Ultrasound-guided erector spinae plane block
At the beginning of anesthetic induction, an ultrasound-guided erector spinae plane block (ESPB) was performed on the surgical side with the patient in the lateral position, awake and responsive. A sterile-covered ultrasound transducer was initially positioned over the fifth rib and subsequently moved medially to identify the costotransverse junction and the transverse process.
After local infiltration of the skin, a block needle was advanced in-plane under real-time ultrasound guidance until contact with the transverse process was achieved. Correct needle placement was confirmed by injection of normal saline, demonstrating hydrodissection of the fascial plane between the transverse process and the erector spinae muscle [17,18].
Following confirmation of the appropriate needle position, 20 mL of ropivacaine 0.375% were administered incrementally with repeated aspiration. A catheter was then advanced 5 cm beyond the needle tip into the erector spinae plane. After attachment of a bacterial filter, the catheter was secured with sterile adhesive dressing and directed cranially toward the shoulder. Catheter tunneling was not performed due to an increased bleeding risk.
According to the institutional protocol, patients in the ESPB group received intermittent boluses of 20 mL ropivacaine 0.2% every three hours over the entire observation period. No catheter- or puncture-related complications requiring deviation from the planned anesthetic management or premature catheter removal were observed.
Intercostal wound infiltration
In the IC-WI group, local anesthetic infiltration of the intercostal space at the thoracotomy site was performed at the end of surgery. Intercostal wound catheters were placed under sterile conditions by an experienced cardiothoracic surgeon.
The catheter was positioned within the intercostal space corresponding to the dermatomal level of the surgical incision, most commonly the fourth or fifth intercostal space. Using an introducer needle, the puncture was performed perpendicular to the skin and advanced medially and slightly caudally toward the inferior margin of the rib. The catheter was subsequently inserted into the fascial plane between the internal and innermost intercostal muscles and secured at the skin with adhesive fixation.
Following catheter placement, 10 mL of ropivacaine 0.2% were infiltrated along the surgical incision. Thereafter, continuous infusion of ropivacaine 0.2% was administered via the intercostal wound catheter at a fixed rate of 6 mL/h over the entire observation period. No dose adjustments were performed. No catheter-related complications necessitating changes in postoperative management were observed.
Pain management and assessment
Postoperatively, analgesic management followed the institutional standard protocol. Pain intensity was routinely assessed using the Numeric Rating Scale (NRS), and rescue analgesia was administered if NRS scores were ≥3. Pain scores were analyzed separately for the day of surgery (postoperative day 0) and for postoperative days 1, 2, and 3, each defined as a 24-hour interval starting at 12:00 a.m. Intraoperative sufentanil consumption and cumulative postoperative opioid requirements over 72 hours (until midnight of postoperative day 3) were recorded and converted into oral morphine milligram equivalents (MME), both as absolute values and normalized to body weight. The standardized multimodal analgesic regimen comprised systemic opioids in combination with non-opioid analgesics, such as paracetamol or metamizole.
Surgical and postoperative characteristics
Surgical parameters were analyzed, including type and duration of surgery and cardiopulmonary bypass time. Allogenic blood transfusion was quantified. Incidence of PONV, duration until the first full mobilization were calculated. Postoperative complications were only considered if confirmed by a physician and documented in the hospital information system or in the corresponding discharge letters. Categorical variables were coded as binary variables (0/1), while ordinal variables were converted into numerical values for statistical analysis.
Statistical Analysis
Due to the retrospective study design, no formal a priori sample size calculation was performed. All consecutive eligible patients treated during the study period were included in the analysis (n = 232). Continuous variables are presented as mean ± standard deviation (SD) or median with interquartile range (IQR), depending on data normality which was tested by D'Agostino-Pearson omnibus test. Categorical variables are reported as counts and percentages. Comparisons between groups were performed using the Student’s t-test or analysis of variance (ANOVA) for normally distributed variables and the Mann–Whitney U test for non-normal data. Categorical variables were compared using the Chi-square test or Fisher’s exact test, as appropriate. A two-sided p-value < 0.05 was considered statistically significant. All statistical analyses were performed using GraphPad Prism (Version 7.0, 225 Franklin Street. Fl. 26, Boston, MA 02110, USA).
3. Results
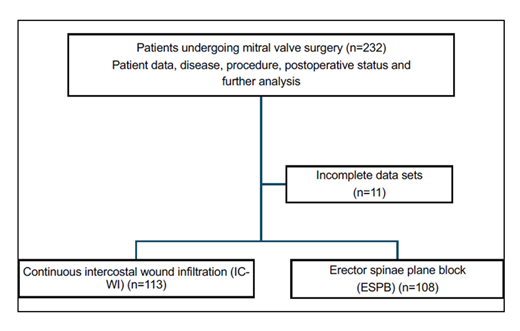
A total of 232 patients were initially enrolled in the study. Eleven patients were excluded due to incomplete datasets, primarily missing key intraoperative or postoperative variables required for analysis (e.g., analgesic dosages, pain scores, or duration of catheter use). The final analysis included 221 patients, with 108 in the ESPB group and 113 in the IC-WI group (Figure 1). Baseline characteristics - including age, sex, body mass index (BMI), ASA classification, NYHA status, and comorbidities - were comparable between the groups (Table 1).
|
Overall (n=221) |
IC-WI (n=113) |
ESPB (n=108) |
p-value |
|
|
Age (yr) |
64 10 |
63 10 |
64 11 |
0.48 |
|
Female, n (%) |
64 (29) |
33 (29) |
31 (29) |
>0.9 |
|
Male, n (%) |
155 (71) |
80 (71) |
75 (71) |
>0.9 |
|
Height (cm) |
175 10 |
175 9 |
174 10 |
>0.9 |
|
Weight (kg) |
78 16 |
79 17 |
78 15 |
0.73 |
|
BMI (kg/m2) |
26 4 |
25 4 |
26 4 |
0.71 |
|
ASA 1/2/3/4, n |
3/14/195/8 |
2/11/96/4 |
1/3/100/4 |
|
|
NYHA I/II/III/IV, n |
41/125/54/1 |
21/65/27/0 |
20/60/27/1 |
|
|
Comorbidities n, (%) |
||||
|
Arterial Hypertension |
104 (47) |
52 (46) |
52 (49) |
0.78 |
|
Coronary heart disease |
36 (16) |
17 (15) |
19 (18) |
0.71 |
|
Diabetes |
15 (7) |
5 (4) |
10 (9) |
0.18 |
|
Type of procedure |
||||
|
MVR |
123 |
66 |
57 |
0.12 |
|
MVR + TVR |
54 |
32 |
22 |
0.21 |
|
MVR + ACB |
44 |
15 |
29 |
0.01 |
|
Duration of |
||||
|
surgery (min) |
213 58 |
212 |
241 56 |
0.85 |
|
CPB time (min) |
157±40 |
154±40 |
157±40 |
0.22 |
|
Transfusion (units per patient) |
||||
|
Red Blood Cell Concentrates |
0.6 2.3 |
0.6 1.7 |
0.7 2.9 |
0.64 |
|
Fresh Frozen Plasma |
0.2 0.7 |
0.1 0.4 |
0.2 0.9 |
0.3 |
|
Thrombocytes |
0.7 2.2 |
0.9 1.8 |
0.6 2.6 |
0.41 |
Notes: Patient characteristics and intraoperative data. The data are shown as numbers of patients and percentages or mean ± standard deviation. Abbreviations: BMI = body mass index; ASA = American Society of Anesthesiologists; NYHA = New York Heart Association; MVR = mitral valve repair or replacement; TVR = tricuspidal valve repair or replacement; ACB = Aortocoronary bypass; CPB = cardiopulmonary bypass
Table 1: Patient characteristics and intraoperative data.
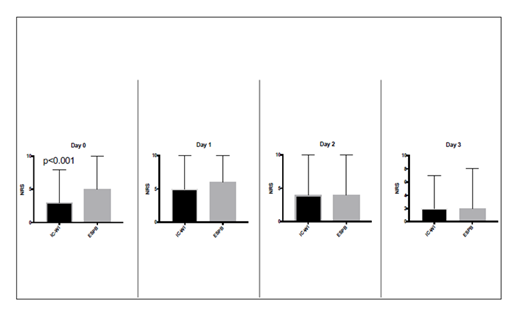
Postoperative pain scores, assessed using the Numeric Rating Scale (NRS), were significantly lower in the IC-WI group on the day of surgery (day 0; p<0.003). On postoperative days 1, 2, and 3, pain scores were similar between the groups (Table 2, Figure 2). The incidence of postoperative nausea and vomiting did not differ significantly (IC-WI: 52 patients vs. ESPB: 54 patients; p=0.7).
Intraoperative sufentanil consumption was significantly lower in the ESPB group (p<0.001). Cumulative postoperative morphine consumption over 72 hours was comparable between groups (median 176±9.3 mg vs. 151±10.3 mg; p=0.07), but when normalized to body weight, consumption was slightly higher in the ESPB group (2.3±1.3 mg/kg vs. 1.9±1.0 mg/kg; p=0.04; Table 2). Cumulative ropivacaine doses were similar between groups, whereas catheter indwelling time was significantly shorter in the ESPB group (3.9 vs. 4.5 days; p<0.001; Table 2).
Notably, none of the patients in the IC-WI group were extubated immediately postoperatively in the operating room, whereas 37 patients in the ESPB group were successfully extubated immediately (p<0.001; Table 2).
|
IC-WI n=113 |
ESPB n=108 |
p-value |
|
|
Pain NRS scores |
|||
|
Day 0 |
3.1±0.3 |
4.3±0.3 |
0.003 |
|
Day 1 |
5.2±0.2 |
5.4±0.2 |
0.3 |
|
Day 2 |
3.8±0.2 |
3.9±0.2 |
0.6 |
|
Day 3 |
2.5±0.2 |
2.5±0.2 |
0.8 |
|
Intraoperative opioids |
|||
|
Sufentanil (total dose in µg) |
200±6.8 |
168±6.6 |
0.001 |
|
Sufentanil (µg/kg) |
2.7±1 |
2.3±1 |
0.01 |
|
Postoperative rescue opioids (MME) |
|||
|
Total MME at 72h |
151±10.3 |
176±9.3 |
0.07 |
|
Total MME at 72h per kg BW |
1.9±1.0 |
2.3±1.3 |
0.04 |
|
Regional anesthesia outcome |
|||
|
Ropivacaine dose (total dose mg) |
1232±620 |
1141±492 |
0.174 |
|
Duration of pain catheter (days) |
4.5±1.4 |
3.9±1.2 |
0.001 |
|
Duration chest drain (h) |
69±3.8 |
62±3.2 |
0.15 |
|
Extubation in the operating room |
|||
|
Yes n (%) |
0 (0) |
39 (36) |
<0.001 |
|
No n (%) |
113 (100) |
69 (64) |
|
|
Postoperative complications |
|||
|
Re-Surgery n (%) |
9 (8) |
16 (15) |
0.13 |
|
Time until mobilization out of bed (hours) |
28±1.5 |
30±2.5 |
0.65 |
|
PONV n (%) |
52 (46) |
54 (50) |
0.7 |
|
Duration chest drain (h) |
69±3.8 |
62±3.2 |
0.15 |
Notes: Primary and secondary outcomes and postoperative variables. Abbreviations: NRS = numeric rating scale; MME = morphine milligram equivalents; PONV = postoperative nausea and vomiting.
Table 2: Primary and secondary outcomes, postoperative variables.
4. Discussion
In this study, we evaluated the analgesic efficacy of ultrasound-guided erector spinae plane block (ESPB) compared with continuous intercostal wound infiltration (IC-WI) in patients undergoing minimally invasive mitral valve surgery via lateral thoracotomy. Our results demonstrate that ESPB provides postoperative analgesia comparable to IC-WI while reducing intraoperative opioid requirements and facilitating immediate extubation in the operating room.
Minimally invasive cardiac surgery (MICS) has become an integral component of enhanced recovery pathways in cardiac surgery. However, thoracotomy-based approaches are frequently associated with significant postoperative pain due to intercostal nerve compression from rib retractors and chest drains [19,20]. This pain can impair respiratory mechanics and contribute to pulmonary complications such as atelectasis and pneumonia [21]. Consequently, effective regional analgesia has become an important component of perioperative management in MICS. Multimodal analgesic strategies that reduce opioid requirements are particularly desirable, as they improve pain control while minimizing opioid-related adverse effects.
The integration of regional anesthesia into the Enhanced Recovery After Cardiac Surgery (ERACS) framework has been associated with improved postoperative recovery, including reduced catecholamine requirements, shorter hospital stays, and higher patient satisfaction [22]. Continuous intercostal wound catheters (IC-WC) have previously demonstrated effectiveness in reducing postoperative pain after mitral valve repair procedures [23]. However, ESPB has recently emerged as a promising alternative regional technique in thoracic and cardiac surgery.
Several studies have reported improved analgesia and reduced opioid consumption with ESPB in cardiac surgery. In addition, the administration of ESPB has been associated with shorter ICU and hospital length of stay when compared to systemic analgesia [24,25]. Nevertheless, evidence remains inconsistent. A recent prospective placebo-controlled trial in patients undergoing mitral valve repair did not demonstrate a significant reduction in morphine consumption within the first 24 hours after surgery [26]. These conflicting findings highlight the need for further comparative studies of ESPB and other regional analgesic strategies in minimally invasive cardiac procedures.
To our knowledge, this study is the first to directly compare ESPB with continuous intercostal wound infiltration in patients undergoing minimally invasive mitral valve surgery via lateral thoracotomy. In our cohort, immediate postoperative pain scores were slightly higher in the ESPB group on the day of surgery. Several factors may explain this finding. First, block failure or incomplete spread of local anesthetic cannot be excluded. Second, patients in the ESPB group received lower intraoperative doses of sufentanil, which may have contributed to higher early postoperative pain perception. In addition, earlier extubation in the ESPB group may have allowed patients to report pain sooner, while in those intubated, pain assessment might have been less accurate.
Despite these early differences in pain scores, cumulative postoperative opioid consumption and the incidence of opioid-related adverse effects were comparable between the two groups, suggesting similar overall analgesic efficacy. The difference of postoperative MME normalized per bodyweight corresponds to 10 mg of oral morphine (per day in an adult of 80 kg bodyweight) between the groups, leaving doubts on the clinical relevance of the differences seen.
From a mechanistic perspective, ESPB provides more widespread but less dense somatic and visceral analgesia by blocking the dorsal and ventral rami of the thoracic spinal nerves through cranio-caudal spread of local anesthetic along the fascial plane [27-28]. Together with the timing of the application, these functional differences could have contributed to the reduced amount of intraoperative opioids. In contrast, IC-WI primarily targets somatic pain at the incision site and may therefore provide stronger immediate local analgesia at the site of the thoracotomy but less extensive regional coverage [29].
An important finding of this study was the higher rate of immediate extubation in the ESPB group. More than one-third of patients receiving ESPB were extubated in the operating room, whereas none of the patients receiving IC-WI underwent on-table extubation. Early extubation represents a key component of ERACS protocols and is associated with improved postoperative recovery [22]. As depicted above, the administration of ESPB before surgical incision may have contributed to improved intraoperative analgesia and reduced opioid requirements, thereby facilitating faster emergence from anesthesia. Although differences in clinical practice patterns or extubation strategies may also have influenced these results, the timing and quality of the regional techniques most likely played an important role. If our assumptions are confirmed by other studies, the ESPB might represent an adequate regional technique in cardiac surgery. From our data, facilitating fast and secure extubation conditions represents a functional advantage within the ERACS concept and might outweigh the limited view on the numeric analgesic efficacy.
From a practical perspective, ESPB offers several other advantages. The technique can be performed under ultrasound guidance with a favorable safety profile and a relatively low risk of complications compared with neuraxial techniques. These characteristics make ESPB an attractive component of multimodal analgesia protocols in minimally invasive cardiac surgery.
Furthermore, cumulative postoperative opioid consumption and the incidence of postoperative nausea and vomiting (PONV) were comparable between the two groups, indicating that the analgesic efficacy of ESPB is similar to that of continuous IC-WI. Reducing opioid requirements is particularly relevant in cardiac surgery, as it may decrease the risk of opioid-related complications, such as respiratory depression and postoperative ileus, potentially contributing to enhanced recovery - although assessing this effect was beyond the scope of the present study.
5. Limitations
This study has several limitations. First, the retrospective single-center design and relatively small sample size limit the generalizability of the findings and introduce potential selection bias. Second, the absence of randomization and blinding may have influenced perioperative management and outcome assessment. Third, variability in operator experience with ESPB and IC-WI may have affected the consistency of the interventions. Finally, this study focused on short-term postoperative outcomes; long-term pain control, functional recovery, and postoperative complications were not evaluated.
6. Conclusion
In patients undergoing minimally invasive mitral valve surgery, ultrasound-guided ESPB provides postoperative analgesia comparable to continuous IC-WI and is associated with lower intraoperative opioid requirements and a higher rate of immediate extubation. These results support ESPB as a promising component of ERACS pathways, although prospective randomized studies are needed to confirm these findings.
Acknowledgments
No.
Disclosure
The author(s) report no conflicts of interest in this work.
References
- Lahtinen P, Kokki H, Hynynen M. Pain after cardiac surgery: a prospective cohort study of 1-year incidence and intensity. Anesthesiology 105 (2006): 794-800.
- Szelkowski LA, Puri NK, Singh R, et al. Current trends in preoperative, intraoperative, and postoperative care of the adult cardiac surgery patient. Curr Probl Surg 52 (2015): 531-69.
- Bordoni B, Marelli F, Morabito B, et al. Post-sternotomy pain syndrome following cardiac surgery: case report. J Pain Res 10 (2017): 1163-9.
- Noss C, Prusinkiewicz C, Nelson G, et al. Enhanced Recovery for Cardiac Surgery. J Cardiothorac Vasc Anesth 32 (2018): 2760-70.
- Batchelor TJP, Rasburn NJ, Abdelnour-Berchtold E, et al. Guidelines for enhanced recovery after lung surgery: recommendations of the Enhanced Recovery After Surgery (ERAS(R)) Society and the European Society of Thoracic Surgeons (ESTS). Eur J Cardiothorac Surg 55 (2019): 91-115.
- Kelava M, Alfirevic A, Bustamante S, et al. Regional Anesthesia in Cardiac Surgery: An Overview of Fascial Plane Chest Wall Blocks. Anesth Analg 131 (2020): 127-35.
- Hargrave J, Grant MC, Kolarczyk L, et al. An Expert Review of Chest Wall Fascial Plane Blocks for Cardiac Surgery. J Cardiothorac Vasc Anesth 37 (2023): 279-90.
- Forero M, Adhikary SD, Lopez H, et al. The Erector Spinae Plane Block: A Novel Analgesic Technique in Thoracic Neuropathic Pain. Reg Anesth Pain Med 41 (2016): 621-7.
- Tsui BCH, Fonseca A, Munshey F, et al. The erector spinae plane (ESP) block: A pooled review of 242 cases. J Clin Anesth 53 (2019): 29-34.
- Toscano A, Capuano P, Costamagna A, et al. Is continuous Erector Spinae Plane Block (ESPB) better than continuous Serratus Anterior Plane Block (SAPB) for mitral valve surgery via mini-thoracotomy? Results from a prospective observational study. Ann Card Anaesth 25 (2022): 286-92.
- Toscano A, Capuano P, Galata M, et al. Safety of Ultrasound-Guided Serratus Anterior and Erector Spinae Fascial Plane Blocks: A Retrospective Analysis in Patients Undergoing Cardiac Surgery While Receiving Anticoagulant and Antiplatelet Drugs. J Cardiothorac Vasc Anesth 36 (2022): 483-8.
- Albi-Feldzer A, Mouret-Fourme EE, Hamouda S, et al. A double-blind randomized trial of wound and intercostal space infiltration with ropivacaine during breast cancer surgery: effects on chronic postoperative pain. Anesthesiology 118 (2013): 318-26.
- Razi SS, Stephens-McDonnough JA, Haq S, et al. Significant reduction of postoperative pain and opioid analgesics requirement with an Enhanced Recovery After Thoracic Surgery protocol. J Thorac Cardiovasc Surg 161 (2021): 1689-1701.
- Guerra-Londono CE, Privorotskiy A, Cozowicz C, et al. Assessment of Intercostal Nerve Block Analgesia for Thoracic Surgery: A Systematic Review and Meta-analysis. JAMA Netw Open 4 (2021): e2133394.
- Stamenkovic DM, Bezmarevic M, Bojic S, et al. Updates on Wound Infiltration Use for Postoperative Pain Management: A Narrative Review. J Clin Med 10 (2021).
- Douglas RN, Kattil P, Lachman N, et al. Superficial versus deep parasternal intercostal plane blocks: cadaveric evaluation of injectate spread. Br J Anaesth 132 (2024): 1153-9.
- Hamilton DL, Manickam B. The Erector Spinae Plane Block. Reg Anesth Pain Med 42 (2017): 276.
- Hamilton DL, Manickam B. Erector spinae plane block for pain relief in rib fractures. Br J Anaesth 118 (2017): 474-5.
- Yuan K, Cui B, Lin D, et al. Advances in Anesthesia Techniques for Postoperative Pain Management in Minimally Invasive Cardiac Surgery: An Expert Opinion. J Cardiothorac Vasc Anesth 39 (2025): 1026-36.
- Miyazaki T, Sakai T, Tsuchiya T, et al. Assessment and follow-up of intercostal nerve damage after video-assisted thoracic surgery. Eur J Cardiothorac Surg 39 (2011): 1033-9.
- Mehta Y, Arora D, Sharma KK, et al. Comparison of continuous thoracic epidural and paravertebral block for postoperative analgesia after robotic-assisted coronary artery bypass surgery. Ann Card Anaesth 11 (2008): 91-6.
- Werner A, Conrads H, Rosenberger J, et al. Effects of Implementing an Enhanced Recovery After Cardiac Surgery Protocol with On-Table Extubation on Patient Outcome and Satisfaction-A Before-After Study. J Clin Med 14 (2025).
- Hosono M, Yasumoto H, Kuwauchi S, et al. Comprehensive Pain Control Strategy in Minimally Invasive Mitral Valve Repair. Ann Thorac Cardiovasc Surg 28 (2022): 180-5.
- Morkos M, DeLeon A, Koeckert M, et al. The Use of Unilateral Erector Spinae Plane Block in Minimally Invasive Cardiac Surgery. J Cardiothorac Vasc Anesth 37 (2023): 432-6.
- Muhammad QUA, Sohail MA, Azam NM, et al. Analgesic efficacy and safety of erector spinae versus serratus anterior plane block in thoracic surgery: a systematic review and meta-analysis of randomized controlled trials. J Anesth Analg Crit Care 4 (2024): 3.
- Hoogma DF, Van den Eynde R, Al Tmimi L, et al. Efficacy of erector spinae plane block for minimally invasive mitral valve surgery: Results of a double-blind, prospective randomized placebo-controlled trial. J Clin Anesth 86 (2023): 111072.
- Sorenstua M, Zantalis N, Raeder J, et al. Spread of local anesthetics after erector spinae plane block: an MRI study in healthy volunteers. Reg Anesth Pain Med 48 (2023): 74-9.
- Varela V, Ruiz C, Montecinos S, et al. Spread of local anesthetic injected in the paravertebral space, intertransverse processes space, and erector spinae plane: a cadaveric model. Reg Anesth Pain Med 49 (2023): 228-32.
- Mowbray A, Wong KK, Murray JM. Intercostal catheterisation. An alternative approach to the paravertebral space. Anaesthesia 42 (1989): 958-61.


 Impact Factor:
* 3.1
Impact Factor:
* 3.1
 Acceptance Rate:
77.58%
Acceptance Rate:
77.58%
 Time to first decision: 10.4 days
Time to first decision: 10.4 days
 Time from article received to acceptance:
2-3 weeks
Time from article received to acceptance:
2-3 weeks
