Self-organization of Small Pluripotent Stem Cells (SPSC) into EB-like Structures Under Autologous Plasma Conditions
Torbjörn Ogéus*
Stockholms Led- & Smärtklinik, Stockholm, Sweden
*Corresponding Author: Torbjörn Ogéus, Stockholms Led- & Smärtklinik, Stockholm, Sweden.
Received: 19 March 2026; Accepted: 25 March 2026; Published: 31 March 2026
Article Information
Citation: Torbjörn Ogéus. Self-organization of Small Pluripotent Stem Cells (SPSC) into EB-like Structures Under Autologous Plasma Conditions. Archives of Clinical and Biomedical Research. 10 (2026): 120-129.
View / Download Pdf Share at FacebookAbstract
Introduction: Rare primitive stem cell–like populations have been proposed to persist in adult tissues, including peripheral blood; however, their biological properties remain incompletely understood. In the present study, we investigated whether small peripheral blood–derived pluripotent-like cells, termed small pluripotent stem cells (SPSCs), possess an intrinsic capacity for spontaneous three-dimensional self-organization under minimal physiologic culture conditions.
Methods: Peripheral blood–derived cells enriched using the Ogéus Sparq cells® platform were cultured under non-adherent conditions in either autologous plasma or isotonic saline without the addition of conventional pluripotent stem cell media or exogenous growth factors. Aggregate formation was monitored using brightfield microscopy over 72 h, while nuclear organization was evaluated using Hoechst staining. Immunofluorescence analysis was performed to assess the expression of pluripotency-associated markers SSEA-4 and SOX2.
Results: SPSCs reproducibly formed compact embryoid body (EB)–like aggregates when maintained in autologous plasma, with early clustering observed within 12 h and well-defined spheroidal structures appearing by 24–48 h. By 72 h, aggregates typically measured approximately 150–220 μm in diameter. Hoechst staining revealed dense and relatively homogeneous nuclear organization throughout the aggregates, supporting the presence of a compact cellular architecture rather than hollow or necrotic cores. Immunofluorescence demonstrated SSEA-4–associated membrane staining and nuclear SOX2 expression within EB-like structures. In contrast, cells maintained in isotonic saline remained largely dispersed and failed to form comparable aggregates.
Conclusions: These findings provide morphological and molecular evidence that small peripheral blood–derived cells can undergo spontaneous EB-like self-organization under physiologic plasma conditions without exogenous growth factor supplementation. Further studies will be required to determine whether these aggregates possess functional pluripotency through tri-lineage differentiation.
Keywords
Pluripotent stem cells; Peripheral blood stem cells; Embryoid bodies; EB-like aggregates; Autologous plasma; SOX2; SSEA-4; Threedimensional cell organization
Article Details
1. Introduction
Pluripotent stem cells are defined by their ability to self-renew and differentiate into derivatives of all three embryonic germ layers [1]. Traditionally, pluripotency has been associated with embryonic stem cells or induced pluripotent stem cells generated through genetic or epigenetic manipulation [2]. However, an increasing body of literature suggests that rare, developmentally primitive cell populations may persist in adult tissues, including peripheral blood.
Several studies have described very small embryonic-like stem cells (VSELs) and other small stem cell populations in adult organisms, often characterized by small size and reported expression of pluripotency-associated factors such as OCT4, SSEA-4, NANOG, and SOX2. While the biological significance and interpretation of these findings remain debated, multiple reports propose that such cells may represent a quiescent, stress-responsive population mobilized by tissue injury or physiological stress [3-5]. However, whether small circulating pluripotent-like cells in adult peripheral blood possess intrinsic self-organizing properties comparable to classical pluripotent stem cells remains largely unexplored.
In our previous work, we demonstrated that a population of small peripheral blood–derived cells enriched using a minimally manipulative protocol expressed the aforementioned pluripotency-associated markers OCT4, SSEA-4, SOX2, and NANOG [6]. In the present study, the term small pluripotent stem cells (SPSC) is used to describe this enriched population of small peripheral blood–derived cells obtained using the Ogéus Sparq cells® workflow and previously characterized by expression of pluripotency-associated markers.
Although marker expression alone does not establish functional pluripotency, these observations supported the rationale to further investigate whether adult peripheral blood may harbor cells with intrinsic pluripotency-associated properties.
A classical in vitro hallmark of pluripotent stem cells is their capacity to self-organize into three-dimensional embryoid bodies (EBs) under non-adherent conditions [7]. EB formation reflects intrinsic cell–cell interactions and developmental programs and is widely used as an early indicator of pluripotency-associated behavior prior to directed differentiation assays. Importantly, EB-like self-organization can be assessed even before tri-lineage differentiation is performed, provided the interpretation is framed carefully as morphological and marker-based evidence rather than definitive functional proof [8].
In the present study, we sought to extend prior findings by investigating whether peripheral blood–derived small pluripotent stem cells (SPSC) demonstrate spontaneous EB-like aggregation under minimal culture conditions. Specifically, we examined (i) aggregation dynamics and growth over time, (ii) nuclear organization within EB-like structures, and (iii) organized expression of the pluripotency-associated markers SSEA-4 and SOX2 in aggregates formed without conventional pluripotent stem cell media or exogenous growth factor supplementation.
2. Methods
2.1 Cell isolation and plasma-based culture (Ogéus Sparq cells® platform)
Peripheral blood–derived cells were processed using the Ogéus Sparq cells® platform, a proprietary, non-expansive workflow developed to enrich and preserve small circulating cell populations under minimally manipulated conditions. The resulting enriched cell fraction is referred to in this study as small pluripotent stem cells (SPSC).
Briefly, peripheral whole blood (typically 60 mL in anticoagulant-containing tubes) was subjected to differential centrifugation according to our previously published cold-enrichment protocol [6]. Key steps included:
- - Initial centrifugation at 600 × g for 10 min at room temperature to separate plasma and cellular components.
- - Transfer of the autologous plasma supernatant to new tubes.
- - Subsequent centrifugation of the plasma supernatant at 1200 × g for 10 min at room temperature to pellet small cell populations while preserving viability.
- - Controlled cooling of the enriched fraction to 4.5 °C for 4.5 hours for stabilization.
The final enriched pellet was gently resuspended in 0.5 mL autologous plasma (or isotonic saline for control conditions) at a concentration of approximately 1–5 × 105 cells/mL, estimated using an automated cell counter (Luna™, Logos Biosystems). Cells were immediately plated in ultra-low attachment 24-well plates and maintained under non-adherent conditions at 37 °C in 5% CO5 (Thomas Scientific Midi 40 CO5 incubator) without addition of exogenous growth factors or conventional pluripotent stem cell media, allowing observation of spontaneous self-organization under minimal physiologic conditions.
For full methodological details, including exact volumes, buffers (if any), and quality control steps, refer to Ogéus and Roos [6].
All human peripheral blood samples used in this study were obtained from commercially available donor blood sources. All blood products were collected by licensed blood banks under standard donor consent procedures for research use. Samples were provided in anonymized form and contained no identifiable donor information. The present study involved only in vitro analysis of previously collected biological material.
2.2 Formation of EB-like aggregates and monitoring
Aggregation was monitored at baseline and after 12, 24, 48, and 72 hours using brightfield microscopy. Aggregate morphology (cluster formation, compaction, and spheroid appearance) was documented, and aggregate diameters were measured using calibrated microscopy images when applicable.
2.3 Nuclear staining
To visualize nuclear organization and assess whether aggregates represented cellular structures rather than debris, live nuclear staining was performed using Hoechst 33342. NucBlue™ staining was applied at endpoint when applicable.
2.4 Immunofluorescence staining
Aggregates were fixed in situ using a commercially available fixative solution (Invitrogen 4% Paraformaldehyde Fixative Solution in PBS) and processed directly in 24-well plates to preserve aggregate integrity. After washing and blocking, immunofluorescence staining was performed using the following primary antibodies:
- • SSEA-4 (surface marker)
- • SOX2 (nuclear transcription factor)
Appropriate species-specific Alexa Fluor–conjugated secondary antibodies were applied. For SOX2 staining, a brief permeabilization step (0.1% Triton X-100 in PBS) was performed prior to antibody incubation. All staining steps were performed using sufficient buffer volumes and gentle handling to minimize mechanical disruption of aggregates.
2.5 Imaging
Fluorescence imaging was performed using fluorescence microscopy. Images were captured using a Magus Lum D400 fluorescence microscope with a MAGUS CLM30 digital camera. The images were captured with acquisition of single-channel images and merged overlays (including brightfield where applicable) to evaluate co-localization patterns and aggregate architecture.
2.6 Experimental design and reproducibility
Experiments were performed using peripheral blood obtained from commercial blood bank sources. Blood samples originated from at least five independent donors (approximately 60 mL per donor) and were used to assess reproducibility across biologically independent samples. All samples were provided in anonymized form. Cell preparations from each donor were processed using the same protocol and distributed across a total of 24 wells under identical culture conditions.
The exact time interval between blood collection and experimental processing could not be determined, as samples were obtained from commercial blood bank sources. However, all samples were handled under standard blood bank storage conditions prior to use.
3. Results
3.1 Spontaneous EB-like aggregation and growth over time
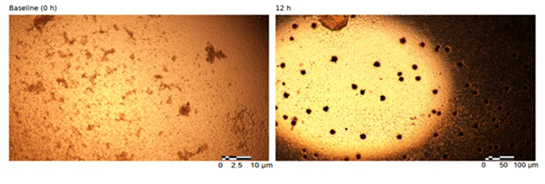
Within 12 hours of incubation under non-adherent conditions, SPSC began forming small multicellular clusters. Over time, clusters progressively compacted, resulting in well-defined, spherical, three-dimensional aggregates by approximately 24–48 hours. By 72 hours, aggregates typically reached diameters of approximately 150–220 µm, consistent with early EB-like structures.
The apparent reduction in aggregate number over time likely reflects progressive compaction and fusion of smaller clusters into larger EB-like structures, a phenomenon commonly observed during early embryoid body formation.
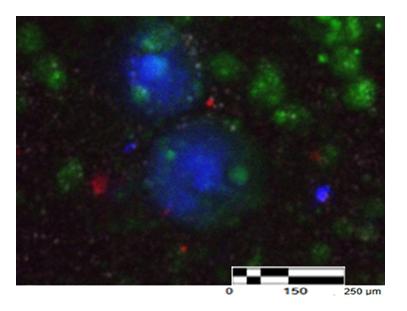
Brightfield imaging demonstrated spheroidal EB-like aggregates with a prominent central dark region in many structures. This appearance prompted further assessment of whether the central region represented a hollow lumen, a necrotic core, or a densely cellular architecture (Figure 1).
Quantitatively, hundreds of aggregates were already observed per well at early time points under plasma conditions. The diameter of these aggregates progressively increased from approximately 1–5 µm at baseline to approximately 150–220 µm by 72 hours. In contrast, cells maintained in isotonic NaCl remained largely dispersed throughout the observation period and did not demonstrate measurable aggregate growth. Qualitative overview of aggregation dynamics is summarized in Table 1, with quantitative measurements provided in Table 2.
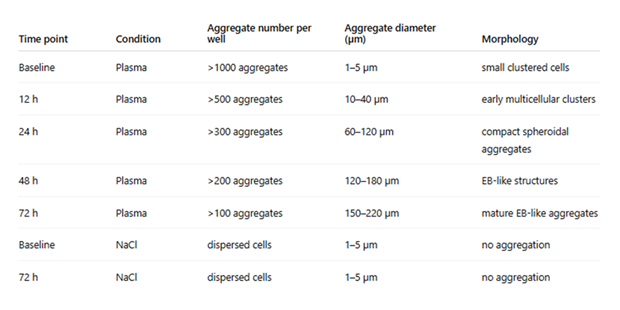
Table 1: Temporal dynamics of SPSC aggregation under plasma and saline conditions. Under plasma conditions, a large number of small aggregates were observed at baseline, which progressively compacted and increased in diameter over time. In contrast, cells maintained in isotonic NaCl remained dispersed and did not demonstrate measurable aggregation or growth.
3.2 Nuclear organization indicates a compact cellular core
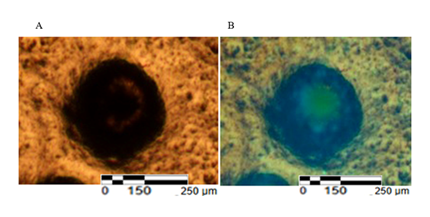
To determine the cellular nature of EB-like aggregates and specifically interpret the central dark region observed in brightfield imaging, nuclei were visualized using Hoechst staining. Hoechst imaging revealed a high density of discrete, Hoechst-positive nuclei throughout the EB-like structures, including the central region. Nuclear fluorescence intensity was remarkably homogeneous across central and peripheral regions, indicating intact nuclear architecture and comparable DNA content throughout the aggregates. The detection of discrete Hoechst-positive nuclei within these structures indicates the presence of DNA-containing cellular units rather than extracellular vesicles or platelets.
This uniform nuclear signal argues against a hollow lumen and is inconsistent with necrosis, debris, or acellular material, which would be expected to produce heterogeneous, fragmented, or absent nuclear fluorescence (Figure 2,3).
Collectively, these findings support the interpretation that the brightfield “dark core” corresponds to a densely packed, nucleus-rich cellular core rather than an empty cavity or degenerative center.
Given the exceptionally small size of the constituent cells (approximately 1–5 µm), a very high nuclear packing density would be expected. Such dense packing would also increase light attenuation in brightfield imaging, providing a plausible optical explanation for the central dark appearance even in aggregates that remain viable and highly cellular (Figure 4).
3.3 Expression of pluripotency-associated markers within EB-like aggregates
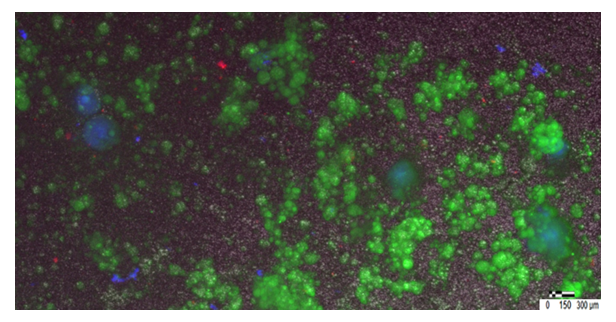
Immunofluorescence analysis demonstrated organized expression of pluripotency-associated markers within and adjacent to EB-like structures.
SSEA-4: SSEA-4–associated signals were observed as discrete, punctate membrane-associated staining, frequently localized to peripheral regions of EB-like aggregates and in closely associated clustered cells. This distribution is consistent with a surface epitope in compact three-dimensional structures, where antigen accessibility is often greatest at the aggregate boundary.
SOX2: SOX2 immunoreactivity was observed as discrete intracellular signals compatible with nuclear localization in a subset of cells. Co-localization with Hoechst-positive nuclei supported nuclear SOX2 expression within densely packed cellular regions of EB-like aggregates.
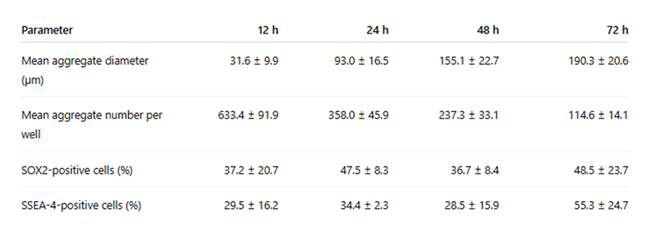
Table 2: Data are presented as mean ± standard deviation (SD). Aggregate diameter measurements were obtained from 23 representative aggregates per time point. Aggregate number per well was determined across 24 independent wells. Marker-positive cells were estimated by semi-quantitative analysis of immunofluorescence images relative to total Hoechst-positive nuclei. Experiments were performed using anonymized blood obtained from at least five independent donors sourced from a licensed blood bank.
Merged images demonstrate EB-like structures with abundant nuclear staining together with SSEA-4 and/or SOX2 signal, supporting the presence of a pluripotency-associated subpopulation within spontaneously formed aggregates.
To quantitatively assess aggregation dynamics, aggregate diameter and aggregate number per well were measured across time points. Mean aggregate diameter increased progressively from 12 h to 72 h, while the number of aggregates per well decreased, consistent with progressive compaction and fusion of smaller clusters into larger EB-like structures. Semi-quantitative analysis of immunofluorescence images further confirmed the presence of SOX2-positive and SSEA-4-positive cells within aggregates. The quantitative summary is presented in Table 2.
3.4 Reproducibility across multiple aggregates within the same field
At 72 hours, SOX2-positive nuclei were observed across multiple EB-like aggregates within the same field of view, indicating that SOX2 expression was not restricted to a single structure but occurred reproducibly across several independently formed aggregates (Figure 5,6).
4. Discussion
4.1 EB-like self-organization under minimal, plasma-based conditions
In the present study, peripheral blood–derived SPSC demonstrated a reproducible capacity to spontaneously self-organize into compact EB-like three-dimensional aggregates under minimal culture conditions. Notably, this behavior was observed in autologous plasma without addition of conventional pluripotent stem cell media or exogenous growth factors. This suggests that at least a subset of the cell population possesses intrinsic survival and organizational properties sufficient to support early EB-like aggregation.
Notably, aggregation occurred in autologous plasma without the use of conventional pluripotent stem cell media or exogenous growth factors, suggesting that physiologic plasma conditions alone may provide sufficient cues to support early three-dimensional organization.
In established EB protocols, aggregate formation is typically performed under defined conditions and often involves media formulations designed for pluripotent stem cells (e.g., factors such as bFGF, TGF-β modulation, or defined serum replacements) [9,10]. In contrast, autologous plasma represents a physiologically relevant environment containing a complex mixture of proteins, metabolites, lipids, extracellular vesicles, and endogenous signaling molecules [11-13]. Although the present study does not dissect mechanisms, the observation that EB-like self-organization occurred in plasma raises the possibility that endogenous plasma components may provide sufficient cues to support early embryonic-like organization in small circulating pluripotent-like cells.
4.2 Interpretation of marker patterns in compact 3D aggregates
The observed marker distribution patterns are biologically coherent with compact EB-like structures. SSEA-4 signals were predominantly peripheral and patchy, which is commonly seen in three-dimensional aggregates where surface epitopes are more accessible at the periphery and where spatial gradients develop during early maturation [13,14]. SOX2 staining was punctate and heterogeneous, consistent with two non-exclusive explanations: (i) limited antibody penetration in compact 3D structures, and (ii) emergence of cellular heterogeneity during early aggregate maturation. Importantly, such spatially restricted SOX2 patterns have been reported in EB-like systems and do not inherently contradict pluripotency-associated properties.
4.3 The “dark core” reflects high cellular density rather than lumen/necrosis
Central dark regions in spheroidal aggregates are sometimes interpreted as luminal cavities or necrotic cores, particularly in larger embryoid bodies [15,16]. Here, however, the presence of dense, discrete, uniformly intense Hoechst-positive nuclei throughout the EB-like structures — including the central region — provides compelling evidence that the aggregates are highly cellular and maintain nuclear integrity across the structure. Hoechst dyes bind double-stranded DNA and require intact nuclear architecture; necrotic or acellular regions would be expected to exhibit reduced, fragmented, or heterogeneous fluorescence rather than uniform signal.
Given the exceptionally small constituent cell size (approximately 1–5 µm), the observed architecture implies an unusually high packing density. Consequently, these EB-like structures may contain substantially higher cell numbers than conventional embryoid bodies of similar diameter formed by larger pluripotent stem cells. This is proposed as a plausible inference based on size and packing, but future work using direct cell counting per aggregate would be needed to quantify cell numbers.
4.4 Converging evidence with prior OCT4A/NANOG observations
The present findings complement prior observations of OCT4A and NANOG expression in cells enriched using the same workflow. Taken together, the combined expression of OCT4A, NANOG, SOX2, and SSEA-4, along with reproducible EB-like self-organization and growth over time, supports the presence of a pluripotency-associated cell population within adult peripheral blood [17-19]. Alternative explanations such as aggregation of heterogeneous circulating cell populations cannot be excluded and will require further investigation using lineage tracing or single-cell analysis.
Similar small pluripotency-associated cell populations have previously been described in adult tissues and bone marrow as very small embryonic-like stem cells (VSELs), characterized by small size and expression of pluripotency markers such as OCT4 and SSEA antigens [20,21].
Although the existence and functional pluripotency of very small embryonic-like stem cells (VSELs) and related populations have been debated in the literature for over two decades, recent independent reviews highlight accumulating evidence supporting their presence in adult tissues and their potential role in tissue homeostasis and repair. In particular, an updated 2025 review by Thetchinamoorthy and Ratajczak synthesizes data from the original discovery team as well as independent laboratories, emphasizing VSELs' broad differentiation capacity and emerging applications in regenerative medicine [22]. These converging observations align with our findings of spontaneous EB-like self-organization in peripheral blood-derived SPSCs under physiologic plasma conditions, providing additional morphological support for the concept of rare, developmentally primitive cells persisting in adult circulation.
4.5 Environmental dependence of aggregation and mechanistic implications
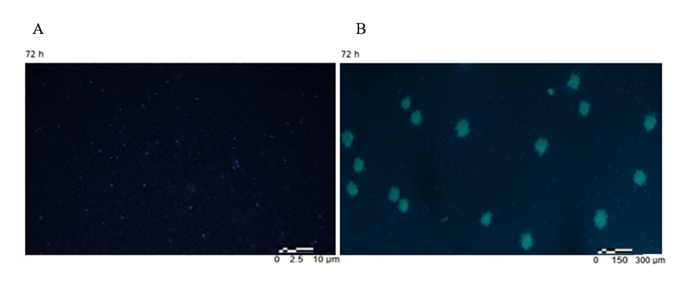
A clear environmental dependence of aggregation behavior was observed when comparing SPSCs incubated in autologous plasma supernatant versus isotonic saline over 72 hours. Cells maintained in isotonic NaCl remained largely dispersed, displaying only isolated Hoechst-positive nuclei without evident formation of multicellular aggregates (Figure S1). In contrast, cells incubated in plasma rapidly formed clearly defined, compact multicellular clusters with increased nuclear density and progressive spatial organization (Figure S2).
Similar aggregation behavior was observed across multiple independent donor samples, suggesting that potential variability in storage time did not substantially influence the observed results.
While the use of commercially sourced blood introduces variability in pre-analytical conditions, the consistent observation of organized EB-like aggregation across multiple independent donor samples, together with the presence of intact Hoechst-positive nuclei and pluripotency-associated marker expression, argues against the possibility that these structures represent random debris, extracellular vesicles, or platelet aggregates, although definitive lineage characterization will require further functional validation (Figure S3).
This marked divergence strongly suggests that aggregation is not a passive consequence of cell concentration, sedimentation, or random mechanical collision. If clustering were purely stochastic, similar aggregate formation would be expected under both conditions. Instead, the data indicate that biological cues present in physiological plasma — but absent in saline — actively support cell survival, cell–cell adhesion, and early three-dimensional organization [23,26].
Autologous plasma constitutes a complex, physiologically relevant microenvironment containing adhesion-mediating proteins (e.g., fibrinogen, fibronectin fragments), complement components, extracellular vesicles, lipoproteins, metabolites, and soluble signaling factors [11,23-28]. These elements likely facilitate initial adhesive interactions, stabilize multicellular contacts, and promote progressive compaction and fusion of smaller clusters into larger EB-like structures — phenomena well documented in other stem cell suspension cultures [24,29,30].
In conventional embryoid body protocols, aggregate formation often depends on defined media supplemented with exogenous growth factors or matrix components [9,10,13]. The present observation that spontaneous EB-like self-organization occurs in unsupplemented autologous plasma raises the possibility that endogenous plasma-derived molecules alone can provide sufficient microenvironmental support to reveal intrinsic organizational properties of small circulating pluripotent-like cells — properties that remain undetectable under minimal salt-based conditions.
Future studies will be required to identify the specific plasma components responsible (e.g., through depletion experiments, proteomic profiling, or fractionation) and to dissect the molecular drivers of this phenomenon—such as extracellular vesicles, plasma adhesion proteins, or autocrine signaling loops—providing further insight into whether these aggregates represent early embryoid body-like entities capable of maturation and lineage-specific differentiation.
4.6 Limitation: functional pluripotency requires tri-lineage differentiation
While EB-like morphology and expression of pluripotency-associated markers are widely used indicators of pluripotency-associated states, they do not alone constitute definitive proof of functional pluripotency. Confirmation requires demonstration of differentiation potential across ectodermal, mesodermal, and endodermal lineages. Accordingly, future experiments will focus on transferring EB-like aggregates to lineage-specific differentiation conditions to assess tri-lineage differentiation capacity.
Preliminary experiments transferring these EB-like aggregates to Matrigel-based differentiation conditions have already demonstrated early expression of lineage-specific markers for all three germ layers at day 6 post-plating (data to be reported in a forthcoming study).
4.7 Regulatory and translational considerations
The ability to observe EB-like self-organization in autologous plasma, without exogenous growth factors or conventional pluripotent media, is not only biologically notable but also potentially relevant from a translational perspective. Autologous plasma represents a patient-derived, physiological environment and may reduce exposure to non-physiologic components commonly used in standard pluripotent stem cell culture systems. While regulatory classification is context-dependent and beyond the scope of the present study, minimizing ex vivo manipulation and avoiding non-native additives are important considerations in multiple regulatory frameworks.
4.8 Key Findings
- • Compact EB-like structures formed within 72 h from peripheral blood–derived small cells cultured in autologous plasma, without use of conventional pluripotent stem cell media.
- • Brightfield imaging revealed a prominent central dark region that corresponded to a high density of discrete, uniformly Hoechst-positive nuclei, supporting a compact cellular core rather than lumen/necrosis/debris.
- • SSEA-4-positive cells were consistently detected within/adjacent to EB-like structures with punctate membrane-associated staining.
- • SOX2 exhibited nuclear localization in a subset of cells and co-localized with Hoechst staining within dense aggregate regions.
- • The exceptionally small constituent cell size (≈1–5 µm) implies unusually high packing density and potentially high cell numbers per aggregate.
5. Conclusion
In this study, peripheral blood–derived SPSCs demonstrated a reproducible capacity to self-organize into compact EB-like aggregates under autologous plasma conditions without exogenous growth factor supplementation or conventional pluripotent stem cell media. These EB-like structures exhibited dense nuclear organization with homogeneous Hoechst staining throughout the aggregate and expressed pluripotency-associated markers including SSEA-4 and nuclear SOX2 in spatially organized patterns.
When considered together with prior observations of OCT4A and NANOG expression in the same cell population, the present findings provide converging morphological and molecular evidence supporting the presence of a pluripotency-associated cell population in adult peripheral blood. While the data support pluripotency-associated properties, definitive confirmation of functional pluripotency will require tri-lineage differentiation assays, which will be addressed in future work.
To our knowledge, spontaneous EB-like aggregation of small peripheral blood–derived cells under plasma-based conditions has not previously been reported.
6. Acknowledgements
Ethical Considerations:
All blood donors provided informed consent before donating blood samples, all data collected was anonymous.
According to the Ethics Commission of Stockholm, Sweden Ethical review was not required for this preclinical laboratory analysis using anonymized human blood obtained from commercial blood bank sources. All donors provided informed consent at the time of donation permitting research use of the collected material.
The study was conducted in accordance with the ethical standards as laid down in the 1964 Declaration of Helsinki and its later amendments.
Consent to participate:
Informed consent was obtained by the blood collection organizations from all donors at the time of donation in accordance with standard blood bank procedures.
Consent for Publication:
This manuscript does not contain any individual person’s data. All data exposed in this manuscript was anonymized.
Funding:
The authors received no financial support for the research, authorship, and/or publication of this article.
Author Contributions:
The author TO, was the main contributor to the manuscript, data collection and sample analysis writing, review and editing. All authors have read and agreed to the published version of the manuscript.
All texts, design, literature review and drafting of this study were done by TO responsible for the submitted manuscript.
Competing Interests:
Ogéus Sparq cells® is a trademarked platform developed by the author. The present study reports biological observations obtained using this platform.
Availability of data and materials:
All data generated or analyzed during this study can be provided by the corresponding authors upon reasonable request and is available for review by the Editor-in-Chief of this journal.
References
- Romito A, Cobellis G. Pluripotent Stem Cells: Current Understanding and Future Directions. Stem Cells Int (2016): 9451492.
- Takahashi K, Yamanaka S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell 126 (2006): 663-676.
- Kucia M, Reca R, Campbell FR, et al. A population of very small embryonic-like (VSEL) CXCR4(+)SSEA-1(+)Oct-4+ stem cells identified in adult bone marrow. Leukemia 20 (2006): 857-869.
- Ratajczak MZ, Zuba-Surma EK, Wysoczynski M, et al. Very small embryonic-like stem cells: characterization, developmental origin, and biological significance. Exp Hematol 36 (2008): 742-751.
- Bhartiya D. Pluripotent Stem Cells in Adult Tissues: Struggling to Be Acknowledged Over Two Decades. Stem Cell Rev Rep 13 (2017): 713-724.
- Ogéus T, Roos A. Isolation and Characterization of Small Pluripotent Stem Cells (SPSCs) from Human Peripheral Blood: A Novel Cold-Enrichment Protocol. Archives of Clinical and Biomedical Research 9 (2025): 328-337.
- Rungarunlert S, Techakumphu M, Pirity MK, et al. Embryoid body formation from embryonic and induced pluripotent stem cells: Benefits of bioreactors. World J Stem Cells 1 (2009): 11-21.
- Zeevaert K, Elsafi Mabrouk MH, Wagner W, et al. Cell Mechanics in Embryoid Bodies. Cells 9 (2020): 2270.
- Kurosawa H. Methods for inducing embryoid body formation: in vitro differentiation system of embryonic stem cells. J Biosci Bioeng 103 (2007): 389-398.
- Discher DE, et al. Growth Factors, Matrices, and Forces Combine and Control Stem Cells. Science 324 (2009): 1673-1677.
- Scadden D. The stem-cell niche as an entity of action. Nature 441 (2006): 1075-1079.
- Yáñez-Mó M, et al. Biological properties of extracellular vesicles and their physiological functions. J Extracell Vesicles 4 (2015): 27066.
- Bratt-Leal AM, Carpenedo RL, McDevitt TC. Engineering the embryoid body microenvironment to direct embryonic stem cell differentiation. Biotechnol Prog 25 (2009): 43-51.
- Pettinato G, Wen X, Zhang N. Engineering Strategies for the Formation of Embryoid Bodies from Human Pluripotent Stem Cells. Stem Cells Dev 24 (2015): 1595-1609.
- Friedrich J, Seidel C, Ebner R, et al. Spheroid-based drug screen: considerations and practical approach. Nat Protoc 4 (2009): 309-324.
- Patel T, Jain N. Multicellular tumor spheroids: A convenient in vitro model for translational cancer research. Life Sci 358 (2024): 123184.
- Boyer LA, Lee TI, Cole MF, et al. Core transcriptional regulatory circuitry in human embryonic stem cells. Cell 122 (2005): 947-956.
- Rodda DJ, Chew JL, Lim LH, et al. Transcriptional regulation of nanog by OCT4 and SOX2. J Biol Chem 280 (2005): 24731-24737.
- Abujarour R, Valamehr B, Robinson M, et al. Optimized surface markers for the prospective isolation of high-quality hiPSCs using flow cytometry selection. Sci Rep 3 (2013): 1179.
- Kucia M, Reca R, Campbell F, et al. A population of very small embryonic-like (VSEL) CXCR4+SSEA-1+Oct-4+ stem cells identified in adult bone marrow. Leukemia 20 (2006): 857-869.
- Ratajczak MZ, Ratajczak J, Kucia M. Very Small Embryonic-Like Stem Cells (VSELs). Circ Res 124 (2019): 208-210.
- Thetchinamoorthy K, Jarczak J, Kieszek P, et al. Very small embryonic-like stem cells (VSELs) on the way for potential applications in regenerative medicine. Front Bioeng Biotechnol 13 (2025): 1564964.
- Anderson N, Anderson N. The Human Plasma Proteome. Mol Cell Proteomics 1 (2002): 845-867.
- Koike M, Sakaki S, Amano Y, et al. Characterization of embryoid bodies of mouse embryonic stem cells formed under various culture conditions and estimation of differentiation status of such bodies. J Biosci Bioeng 104 (2007): 294-299.
- Mosesson MW. Fibrinogen and fibrin structure and functions. J Thromb Haemost 3 (2005): 1894-1904.
- Lin RZ, Chang HY. Recent advances in three-dimensional multicellular spheroid culture for biomedical research. Biotechnol J 3 (2008): 1172-1184.
- Itskovitz-Eldor J, Schuldiner M, Karsenti D, et al. Differentiation of human embryonic stem cells into embryoid bodies compromising the three embryonic germ layers. Mol Med 6 (2000): 88-95.
- Théry C, Zitvogel L, Amigorena S. Exosomes: composition, biogenesis and function. Nat Rev Immunol 2 (2002): 569-579.
- Doetschman TC, Eistetter H, Katz M, et al. The in vitro development of blastocyst-derived embryonic stem cell lines: formation of visceral yolk sac, blood islands and myocardium. J Embryol Exp Morphol 87 (1985): 27-45.
- Carpenedo RL, Sargent CY, McDevitt TC. Rotary suspension culture enhances the efficiency, yield, and homogeneity of embryoid body differentiation. Stem Cells 25 (2007): 2224-2234.
Supplementary Files:
Hypothesis Box
|
Hypothesis: Small peripheral blood–derived pluripotent-like cells possess an intrinsic capacity for self-organization into compact EB-like structures when maintained in a physiologic plasma environment. Rationale: Autologous plasma contains a complex mixture of endogenous signaling molecules, extracellular vesicles, metabolites, and structural proteins that may collectively provide sufficient cues to support early embryonic-like organization. Unlike conventional pluripotent stem cell media, plasma-based conditions may allow small pluripotent-like cells to respond to endogenous regulatory signals closer to in vivo biology. Testable predictions: · EB-like formation will be reduced or altered under fully synthetic or growth factor–depleted conditions. · Plasma from different physiologic states (e.g., stress/hypoxia) may differentially influence aggregate formation. · Plasma-based culture may preserve endogenous marker expression patterns more faithfully than conventional media. |
- A) cells maintained in NaCl/saline show predominantly dispersed Hoechst-positive nuclei without evident aggregation. B) cells maintained in autologous plasma supernatant demonstrate formation of multicellular aggregates with increased nuclear density. Scale bar: 300 µm.






 Impact Factor:
* 5.8
Impact Factor:
* 5.8
 Acceptance Rate:
71.20%
Acceptance Rate:
71.20%
 Time to first decision: 10.4 days
Time to first decision: 10.4 days
 Time from article received to acceptance:
2-3 weeks
Time from article received to acceptance:
2-3 weeks
