Therapeutic and Molecular Analysis of Natural Pterostilbene on Hela Cell Line
Ahmed Kareem Hussein Alatafi1*, Preeti Shrivastava2, Khushboo Agrawal3
1Department of Biology, College of Science, University of Diyala, Iraq
2Department of Biochemistry, Mata JijaBai Govt. PG girl’s college,indore, India
3University College Dublin, Ireland
*Corresponding authors: Ahmed Kareem Hussein Alatafi, Department of Biology, College of Science, University of Diyala, Iraq.
Received: 24 February 2026; Accepted: 14 March 2026; Published: 26 March 2026
Article Information
Citation: Ahmed Kareem Hussein Alatafi, Preeti Shrivastava, Khushboo Agrawal. Therapeutic and Molecular Analysis of Natural Pterostilbene on Hela Cell Line. Journal of Biotechnology and Biomedicine. 9 (2026): 84-93.
DOI: 10.26502/jbb.2642-91280213
View / Download Pdf Share at FacebookAbstract
Pterostilbene, a natural stilbene derivative, has garnered significant attention for its antioxidant, anti-inflammatory, and potential anticancer properties. This study aimed to investigate the therapeutic and molecular effects of blackberry derived pterostilbene on HeLa cervical cancer cell line. The present study was conducted in two phases to explore both the phytochemical potential of different extracts of Blackberries and therapeutic potential of isolated natural Pterostilbene on HeLa cell line. Initially, four blackberry extracts (BBM, BBW, BBH, and BBE) were evaluated for their total phenolic & flavonoid content (TPC and TFC) and antioxidant activity. Among them, BBW exhibited the highest phenolic concentration and ABTS radical scavenging activity, indicating its strong antioxidant potential. In the second phase, Pterostilbene was isolated from blackberry and tested for its cytotoxic effects on cancer cell line. Treatment with pterostilbene led to significant inhibition of cell viability, suggesting strong anticancer activity. Importantly, gene expression analysis revealed a marked upregulation of M-CSF and no effect on GM-CSF; these findings highlight the dual importance of natural plant extracts for antioxidant support and pterostilbene as a promising candidate for targeted anticancer therapy through cytotoxic effect on cancer cell line and modulation of immune-related cytokine signaling.
Keywords
Pterostilbene; HeLa cells; Anticancer; Cytotoxicity; Antioxidant
Article Details
Introduction
Cancer remains one of the leading causes of mortality worldwide, necessitating continuous efforts to identify effective therapeutic agents with minimal adverse effects [1]. In this quest, natural compounds derived from dietary sources have gained considerable attention due to their potential chemo preventive and therapeutic properties [2]. Among such compounds, pterostilbene stands out as a promising bioactive agent. A dimethylated derivative of resveratrol, pterostilbene is primarily found in blueberries, grapes, and heartwood of red sandalwood (Pterocarpus santalinus). It is recognized for its enhanced bioavailability, potent antioxidant properties, and a broad spectrum of biological activities, including anti-cancer effects [3]. Cervical cancer is one of the most prevalent cancers among women globally [4], with HeLa cells, derived from human cervical adenocarcinoma, being widely utilized as a model for studying cervical cancer mechanisms and treatments. Current therapeutic approaches for cervical cancer, including surgery, chemotherapy, and radiation therapy, are associated with significant limitations, such as drug resistance and systemic toxicity [5]. Thus, identifying novel therapeutic agents capable of selectively inducing cancer cell death while sparing normal cells remains critical. Recent studies have demonstrated that pterostilbene exerts cytotoxic effects on a variety of cancer cell lines by modulating several molecular pathways [6,7]. These pathways include the inhibition of cell proliferation, induction of apoptosis, regulation of cell cycle progression, and suppression of metastasis. In cervical cancer cells, emerging evidence suggests that pterostilbene could effectively inhibit cellular growth by modulating molecular targets such as p53, reactive oxygen species (ROS), and pathways involving inflammatory and apoptotic markers [8,9]. This study focuses on evaluating the therapeutic potential of natural pterostilbene against the HeLa cell line. It also aims to elucidate the molecular mechanisms underlying its effects. Specifically, the research examines pterostilbene’s influence on cell proliferation, apoptosis induction, ROS generation, and key signaling pathways involved in cancer progression. By combining cellular assays with molecular analyses, this investigation provides insights into the potential use of pterostilbene as an adjunctive or alternative therapeutic agent for cervical cancer. This research holds promise in advancing our understanding of natural stilbenes as bioactive compounds with potent anticancer activities. The findings could contribute to the development of targeted, less toxic treatment strategies for cervical cancer patients.
Material and Method
Cell Culture and treatment
HeLa cells (ATCC Number: CCL-2) were purchased from ATCC (Manassas, VA, USA) and cultured in ATCC-formulated Eagle’s Minimum Essential Medium with 10% FBS and 10 mg/mL gentamicin at 37°C in 5% CO2. For all experiments, HeLa cells were seeded at 2 × 105 cells/mL. The cells were treated with PTE-2 at the indicated concentration for 48 h.
Sample collection
The fruit part of plant Rubus fruticosus (blackberry)( 500 gm) was collected and samples were washed with running tap water surface sterilized with 0.1 % mercuric chloride and allowed to air dry in shade for two to four weeks. Precaution was taken to avoid direct contact of sun light to avoid the chances of destroying the active compounds present in samples. After drying, the samples were grinded finely and stored in airtight container in dry and cool place to avoid contamination and deterioration.
Extraction
In the current investigation, plant material was extracted utilizing the continuous hot percolation method with Soxhlet equipment. The powdered substance of Rubus fruticosus (blackberry) was placed in a thimble of soxhlet. The air-dried powder (5 g) was successively extracted by Hot-extraction method i.e. soxhlet extraction with solvents of different polarity i.e. Hexane, chloroform, Methanol and Hot water. 450 ml of hexane solvent was used for extraction and time duration was maintained for 6-8 hrs. (4-6 cycles per hour i.e 24-48 cycles). The hexane solvent containing extract was dried, collected in sterile container and stored in air tight container. Similarly different extracts were prepared in different solvents. All the dried extracts were weighed. All the extracts were diluted to 20 ml in their respective solvents. The dried extract was weighed, and each extract's % yield was calculated using the following formula:
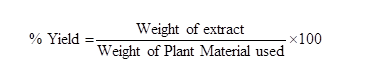
Prepared extracts was observed for organoleptic characters (percentage yield, colour and odour) and was packed in air tight container and labelled till further use [10].
Phytochemical investigation
An experiment was conducted to assess the presence or absence of numerous phytoconstituents utilizing a detailed qualitative phytochemical analysis. Reactions to testing were based on colour intensity or precipitate formation [11].
Quantitative Phytochemical Estimation
TPC
The total phenolic content of Rubus fruticosus (blackberry) extract was determined using the Folin-Ciocalteu Assay. The Rubus fruticosus (blackberry) extracts (0.2 mL from stock solution) were mixed with 2.5 mL of Folin-Ciocalteu Reagent and 2mL of 7.5% sodium carbonate. This mixture was diluted up to 7 mL with distilled water. Then the resulting solutions were allowed to stand at room temperature for 2 hrs before the absorbance was measured spectrophotometrically at 765 nm. Calibration curves were composed using standard solutions of Gallic Acid Equivalent (GAE) mg/gm. Concentration of 05, 10, 15, 20, and 25 µg/mL of Gallic aid was prepared. The Folin-ciocalteu reagent is sensitive to reducing compounds including polyphenols. They produce a colour upon reaction. Then colour was measured spectrophotometrically [12].
TFC
The flavonoid content was determined using Aluminium chloride method. 0.5 ml of Rubus fruticosus (blackberry) extract solution was mixed with 2 ml of distilled water. Then, 0.15 ml of sodium nitrite (5%) was added and mixed properly. After that, wait for 6 minutes before adding 0.15 ml Aluminium chloride (10 %) and allowed to stand for 6 minutes. Then, 2 ml of 4 % sodium hydroxide was added. The mixture was shaken and mixed thoroughly. Absorbance of mixture was estimated at 415 nm using UV spectrophotometer. Calibration curves were composed using standard solutions of Quercetin Equivalent (RE) mg/gm. Concentration of 20, 40, 60, 80, 100, 120, 140, and 160 µg/mL of Quercetin was prepared. The calibration curve was used to determine the total flavonoid concentration, which was expressed as mg Quercetin equivalent per gram of dry extract weight [13].
Antioxidant Activity
DPPH assay
The antioxidant activity of Rubus fruticosus (blackberry) extract was determined by using the DPPH free radical scavenging assay. 1 mg/ml methanol solution of extracts/standard was prepared. To create different concentrations of Rubus fruticosus (blackberry) extracts/standard (20-100µg/ml), a 1mg/mL stock solution was mixed with 2mL of 0.1mM DPPH solution. The resultant mixture was vortexed and incubated for 30 minutes at room temperature in a relatively dark atmosphere before being measured at 517 nm with a UV spectrophotometer (Shimadzu 1700). For the control, add 3 ml of 0.1mM DPPH solution and incubate for 30 minutes at room temperature in the dark. Absorbance of the control was taken against methanol (as blank) at 517 nm [14]. Percentage antioxidant activity of sample/standard was calculated by using formula:
% Inhibition = [(Ab of control- Ab of sample)/ Ab of control x 100]
FRAP assay
FRAP assay used was essentially as previously described [15]. Samples were diluted to 1mg/ml. An aliquot (100 μl) of the appropriately diluted extract was added to 3 ml of the standard reaction solution and the absorbance was measured at 593 nm at 0 time and after 6 min standing at room temperature. The measurement was taken in triplicate. FeSO4 was used to generate the standard curve in the range of 200–1000 μM. The FRAP values for standard and samples were calculated and expressed as μM Fe[II]/gm dry wt.
ABTS assay
To prepare the ABTS reagent, dissolved 0.180 g of ABTS in 50 mL of water (7 mM) and 0.033 g of potassium persulfate in 50 mL of water (2.45 mM). Mixed these solutions in a 1:1 ratio and incubated in the dark for 12–16 hours. Diluted the mixture to achieve an absorbance of ~0.7 at 734 nm. For analysis, mixed 5 µL of sample extract with 3.395 mL of the ABTS reagent, incubated in the dark for 10 minutes, and measured absorbance at 734 nm. A control was prepared using ABTS and methanol, and a standard curve of ascorbic acid was prepared with dilutions ranging from 1 to 5 µg/mL. The ABTS+ scavenging effect (%) was calculated as (Ab−Aa)/Ab×100, where Ab is the absorbance of ABTS with methanol, and Aa is the absorbance of ABTS with the sample/standard. CEAC was determined based on the ascorbic acid standard curve.
Column chromatography
Residue was subjected to column chromatography for compound isolation. Silica gel (60–120 mesh) was used as the stationary phase in a glass column (3×50 cm). The column was initially equilibrated with hexane. The dried ethyl acetate extract was adsorbed onto a small quantity of silica gel, dried, and loaded onto the column. Elution was carried out using a gradient of hexane and ethyl acetate, starting from 90:10 and progressing to 0:100. Fractions of 10 mL each were collected and monitored by thin-layer chromatography (TLC) using a mobile phase of hexane:ethyl acetate (7:3). The presence of pterostilbene was confirmed by the appearance of a distinct spot under UV light (254 nm) with an Rf value of approximately 0.6.
HPLC
The analysis was conducted using a reversed-phase C18 column, operating under isocratic conditions with a mobile phase composition of acetonitrile (ACN) and water in a 50:50 ratio. The flow rate of the mobile phase was maintained at 1 mL per minute. Detection was performed at a wavelength of 306 nm, ensuring optimal sensitivity for trans-pterostilbene. Standard and sample injections were carried out, with retention times and other peak parameters recorded and analyzed for quantification. The results included peak areas, heights, and retention times, as well as additional parameters like peak width. This method ensures accurate identification and quantification of trans-pterostilbene based on its specific retention time and spectral properties.
Cytotoxicity Assay (Cell viability assay)
Cell viability was measured using the MTT assay. To perform an MTT assay, prepared the necessary solutions, ensuring the MTT solution was stored at 4°C protected from light and the solubilization solution had no precipitates. Seeded 25,000 HeLa cells per well in a 96-well plate with 250 μL of phenol-red-free DMEM (10% FBS). Treated cells with the cytotoxic compound (e.g., 5 μM Doxorubicin) and incubated for 24 hours. Washed wells three times with PBS, then added 125 μL of DMEM and 25 μL of MTT solution per well. Incubated at 37°C for 2 hours, then added 100 μL of solubilization solution and gently mix. Measured absorbance at 570 nm using a spectrophotometer to assess cell viability. The cell inhibition rate was calculated as 100% × (control group A values - experimental group A values) / control group A values.
REAL TIME PCR
The Real-Time PCR analysis was conducted to evaluate the expression of M-CSF and GM-CSF genes in HCT-116 cells using GAPDH as the housekeeping gene for normalization. Specific primers for GAPDH, M-CSF, and GM-CSF were used based on established protocols [16,17]. A standard curve was prepared by serially diluting cDNA (150 ng/ml to 0.01 ng/ml) to determine PCR efficiency and amplification factors. Efficiencies of the target and housekeeping genes were close to 100%, ensuring reliable quantification. The comparative Ct method (2^-ΔΔCt) was used to determine the fold change in gene expression. GAPDH-normalized Ct values were obtained for untreated HeLa and cells treated with PTE-2 fraction.
|
Gene |
Forward (5’-3’) |
Reverse (5’-3’) |
|
GAPDH |
AACGGATTTGGTCGTATTG |
GTACTCCAGAAGACCAGAGG |
|
GM-CSF |
GGGAGCATGTGAATGCCATC |
GGCTCCTGGAGGTCAAACAT |
|
M-CSF (CSF-1) |
TCCAACTACATTGTCAAG GGCAATGCCCGCCT |
CAGAT TGGTATAGTCCCGCTCTCTCCTGTCCT |
Primers used
Results
Percentage Yield
In phytochemical extraction the percentage yield is very crucial in order to determine the standard efficiency of extraction for a specific plant, various sections of the same plant or different solvents used.
|
Sample |
Final dry weight |
Extraction yield (%) |
|
BBM (Methanol) |
2.3 |
46 |
|
BBW (Hot Water) |
2.4 |
48 |
|
BBH (Hexane) |
1.3 |
26 |
|
BBE (Ethanol) |
1.7 |
34 |
Table 1: Percentage Yield of
The percentage yield of phytochemical extraction, a critical parameter for assessing the efficacy of extraction of specific plants, was examined using various solvent system.Table1 presents the final dry weight and corresponding extraction yield for the samples. The methanol extract yielded 46%, while the hot water extract achieved a 48% yield. The hexane extract resulted in a 26% yield and Ethanol extract yielded.
Preliminary Phytochemical study
Table 2: Phytochemical testing of extract.
The Phytochemical analysis of crude extracts from the sample (BBM, BBW, BBH, and BBE) Revealed varying compositions of bioactive compounds, as summarized in Table 2. Sample BBM exhibited the presence of flavonoids, tannins, carbohydrates and steroids, with a positive wagner’s test but negative results for alkaloids, Mayer’s test, Dragendorff’s test, and steroids. Sample BBW displayed positive results for flavonoids, phenolics, with a notable presence of saponin, while testing negative for alkaloid, carbohydrate, glycosides and steroids. Sample BBH exhibited flavonoids,phenolics tannins and carbohydrate, with a positive reaction for saponin but no alkaloids and steroids were detected. Sample BBE prove the presence of alkaloids, glycosides, tannins and saponins and steroids, indicating a diverge phytochemical profile.
Quantitative Analysis
Preliminary phytochemical testing of crude extracts confirmed the presence of phenolics and flavonoids in plant material. To estimate their amount total phenolic (TPC) and total flavonoid content (TFC) assays were performed.
Total Phenolic content (TPC) and Total Flavonoids content (TFC) estimation
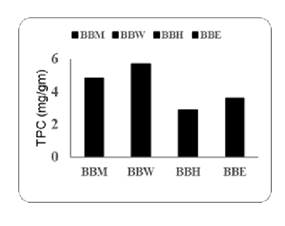
The total phenolic content (TPC) of the extracts was showed as mg/g equivalent of Gallic Acid. The results are presented in Table 3 and Figure 1. The TPC values for the different samples were as follows: the value for BBM was 4.838 mg/g, for BBW was 5.706 mg/g, for BBH was 2.868 mg/g, and for BBE was 3.602 mg/g. The highest TPC was observed in the BBW extract, while the lowest was recorded in the BBH extract.
Table 5: Total Phenolic Content of Extracts.
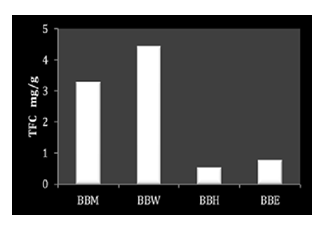
Moreover, the total flavonoid content (TFC) of the extracts displayed as mg/g equivalent of Quercetin. The results are summarized in Table 4. The TFC values were as follows: for BBM 3.287 mg/g, for BBW 4.431 mg/g, for BBH 0.544 mg/g, and for BBE 0.773 mg/g. The highest TFC was found in the BBW extract, while the lowest was observed in the BBH extract.
In vitro Antioxidant Assays
In the present investigation, the in vitro anti-oxidant activity of extracts of Rubus fruticosus (blackberry) was evaluated by DPPH radical scavenging activity and ABTS assay. The results are summarized in Tables.
DPPH 1, 1- diphenyl-2-picryl hydrazyl Assay and 3.5.3 ABTS Assay
|
S.No |
Sample |
% Inhibition |
|
1 |
BBM |
63.450 |
|
2 |
BBW |
65.321 |
|
3 |
BBH |
45.672 |
|
4. |
BBE |
62.865 |
Table 3: DPPH radical scavenging activity of blackberry extracts.
|
S.No |
Sample |
% Inhibition |
|
1 |
BBM |
80.359 |
|
2 |
BBW |
88.024 |
|
3 |
BBH |
75.059 |
|
4. |
BBE |
25.696 |
Table 4: ABTS assay of blackberry extracts.
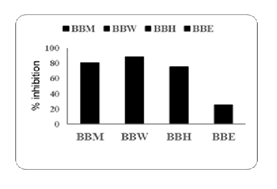
The DPPH radical scavenging activity of blackberry extracts was examined, and the results are presented in Table 5.Percent inhibition (% Inhibition) for each sample are as follows: BBM showed % Inhibition of 63.450, BBW showed % Inhibition of 65.321, BBH had a % Inhibition of 45.672, and BBE displayed % Inhibition of 62.865.The antioxidant capacity of blackberry extracts was further assessed using the ABTS assay, with results summarized in Table 6. Percent Inhibition for each sample was: BBM with % Inhibition of 80.359, BBW with a % Inhibition of 88.024, BBH with a % Inhibition of 75.059, and BBE with a % Inhibition of 25.696. The corresponding % Inhibition values are visually represented in the accompanying bar chart, highlighting the varying antioxidant potentials among the extracts.
FRAP Assay
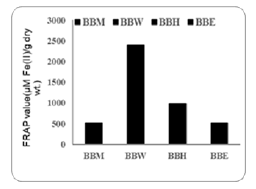
The ferric reducing antioxidant power assay revealed a concentration dependent increase in absorbance for the standard Feso4 with value ranging from 0.109±0.002 at 100µg/ml to 1.59±0 at 1500µg/ml, indicating as strong linear relationship. Among the blackberry extracts, BBW showed the highest FRAP value (224µgFe (II)/g dry weight, followed by BBH (971.42±98.97Fe (II)/g dry weight, while BBM and BBE showed lower activities (514.38±71 and 514.2µgFe (II)/g dry weight respectively). These results suggest varying antioxidant capabilities among the blackberry extracts, with BBW displaying the most potent reducing power.
HPLC
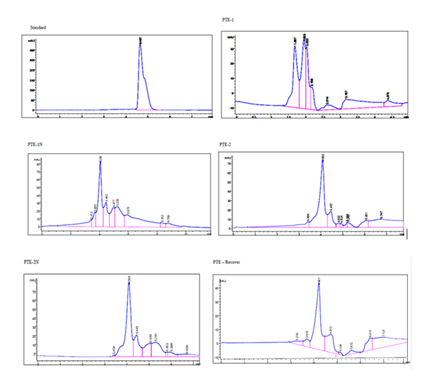
The HPLC analysis results for Trans-Pterostilbene showed a retention time of 4.467 minutes with a peak area of 4823.001 mAU*s, accounting for 100% of the total area. Several additional samples (PTE-1, PTE-1N, PTE-2, and PTE-2N) exhibited multiple peaks. For instance, in PTE-1, a notable peak at 1.687 minutes showed an area percentage of 25.90%, while PTE-2 highlighted a peak at 4.082 minutes with an area contribution of 43.42%. The data reveals distinct component profiles based on their retention times and areas, demonstrating the separations achievable under specified conditions (mobile phase ACN: water, flow rate 1 mL/min, detection at 306 nm).
MTT Assay
MTT assay (IC50): 318.85 µg/ml
|
conc. (µg/ml) |
%Cytotoxicity± std. dev. |
|
50 |
16.228±0.439 |
|
75 |
19.636±0.595 |
|
100 |
23.434±0.313 |
|
150 |
27.361±0.258 |
|
200 |
40.409±0.779 |
|
250 |
43.979±0.5 |
|
300 |
45.764±0.351 |
|
350 |
56.994±0.281 |
|
400 |
65.303±0.313 |
|
450 |
82.928±0.595 |
|
500 |
95.748±0.49 |
Table 5: MTT assay of PTE2 fraction.
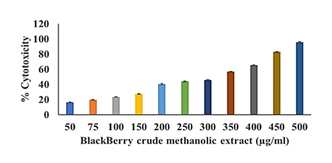
The MTT assay was performed to assess the cytotoxicity of the PTE2 fraction on HeLa cell line, revealing a 50% inhibitory concentration (IC50) of 318.85 μg/ml. The assay results, detailed across a range of concentrations, demonstrated a dose-dependent increase in cytotoxicity at 50 μg/ml, cytotoxicity was 16.228 ± 0.439%; at 75 μg/ml, it was 19.636 ± 0.595%; at 100 μg/ml, 23.434 ± 0.313%; at 150 μg/ml, 27.361 ± 0.258%; at 200 μg/ml, 40.409 ± 0.779%; at 250 μg/ml, 43.979 ± 0.5%; at 300 μg/ml, 45.764 ± 0.351%; at 350 μg/ml, 56.994 ± 0.281%; at 400 μg/ml, 65.303 ± 0.313%; at 450 μg/ml, 82.928 ± 0.595%; and at 500 μg/ml, the highest cytotoxicity was recorded at 95.748 ± 0.49%. These findings indicate that the PTE2 fraction exhibits significant cytotoxic effects, with the most pronounced impact observed at the highest tested concentration.
PCR
Real Time PCR of M-CSF and GM-CSF genes in HCT-116
For standard curve analysis cDNA was serially diluted from 150 to 0.01 ng/ml concentrations.
|
Gene |
Amplification factor |
PCR Efficiency (%) |
Efficiency between control and target gene |
|
GAPDH |
1.97 |
97.33 |
|
|
M-CSF |
1.97 |
96.72 |
0.0155 |
|
GM-CSF |
2.02 |
101.77 |
-0.1073 |
Table 6: PCR Efficiency table.
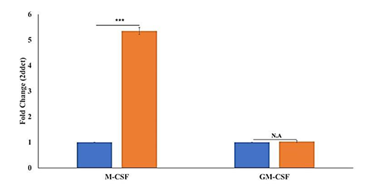
The experiment showed the 5.29-fold upregulation of M-CSF in PTE-2 treated sample compared to non- treated HeLa., While1.04 fold (minimal change) in GM-CSF. The high fold change for M-CSF suggests significant upregulation in the Hela- PTE-2 condition, while the GM-CSF remains relatively constant across both conditions.
Discussion
The phytochemical investigation of the crude extracts of black berry i.e BBM, BBW, BBH, and BBE revealed a diverse presence of bioactive compounds. The results indicate that all extracts exhibited positive tests for flavonoids and phenolics except BBE although it was positive for broadest spectrum of phytochemicals, including alkaloids, Dragendorff's test positives, Wagner's test positives, and steroids. In contrast, BBM and BBW demonstrated notable absences in alkaloids and steroids. Similar studies reported that Blackberries contain numerous phytochemical including polyphenols, flavonoids, anthocyanins, salicylic acid, ellagic acid, and fiber [18].These findings propose that variation in chemical compositions among the extracts, which may correlate with their potential biological activities. This variability highlights the influence of plant source and extraction techniques on phytochemical profiles [19].
Quantitative analysis further elucidated the phenolic (TPC) and flavonoid (TFC) contents. BBW exhibited the highest TPC, followed by BBM, BBE and BBH. The TFC values presented a different trend; with BBW recording the highest TFC wile BBE had the lowest. These variations underscore the differential extraction efficiencies and the influence of the extraction method on the yield of specific phytochemicals. A preliminary analysis using HPLC showed that the blueberry, blackberry, and strawberry samples tested contained a range of phenolic acids and various types of flavonoids [20]. The raised TPC and TFC in BBW and BBM suggest a rich antioxidant potential, which aligns with the known roles of phenolics and flavonoids in scavenging free radicals [21]. The DPPH assay results demonstrate the antioxidant potential of the blackberry extracts (BBM, BBW, BBH, and BBE), these extracts showed varying degrees of DPPH radical scavenging activity, with inhibition percentages ranging from 45.672% to 65.450% . Among the extracts, BBW displayed the highest inhibition, followed by BBM, BBH, and BBE. These results advocate that blackberry extracts contain bioactive compounds, such as anthocyanins and phenolic acids, which are known to contribute to antioxidant activity [22]. The variability in inhibition may be attributed to differences in extraction methods or the concentration of active constituents.
Antioxidant activity of black berry extracts was determined by various methods, FRAP assay highlight the antioxidant capacity of standard FeSO4 and blackberry extracts, respectively. the blackberry extracts BBM, BBW, BBH, and BBE displayed varying FRAP values, ranging from 514.28 to 971.42 µM Fe(II)/g dry weight . BBW displayed the highest FRAP value followed by BBH, BBM, and BBE. These differences propose that the antioxidant potential of blackberry extracts may be influenced by the presence of polyphenolic compounds, such as anthocyanin’s , which are known to contribute to reducing power. The superior FRAP values of BBW and BBH compared to BBW and BBE indicate that certain extraction methods or blackberry varieties may yield advanced antioxidant activity, These findings suggest that blackberry extracts could assist as natural antioxidants in food or pharmaceutical applications. On the other hand The DPPH radical scavenging activity of blackberry extracts, demonstrates varying grades of antioxidant potential among the samples tested. These results suggest that BBW displays the highest radical scavenging activity, followed closely by BBM and BBE, while BBH shows the least efficacy among the tested samples. BBW’s superior performance could indicate an advanced content of these bioactive compounds, potentially due to differences in extraction methods. Conversely, the lower activity of BBH might suggest a reduced presence of such compounds or the presence of intrusive substances that diminish its antioxidant capacity.
The results of the ABTS assay demonstrate significant antioxidant activity across the samples tested (BBM, BBW, BBH, and BBE). These findings indicate a strong capacity of blackberry extracts to neutralize free radicals, with BBW exhibiting the highest inhibition and BBE the lowermost. For instance, BBW’s superior performance could indicate a higher phenolic content or a more efficient extraction process compared to BBE.
Blackberry extract was fractionated and identified by HPLC. The HPLC analysis of Trans-Pterostilbene delivered additional insights into the chemical profiles of the extracts. The retention time of 4.462 minutes and a peak area of 4823.001 mAU* for the standard sample direct a robust detection of this compound. However, the presence of multiple peaks in additional samples (PTE-1, PTE-IN, PTE-2, and PTE-2N) proposes a complex mixture of constituents. Notably, PTE-1 exhibited a significant peak at 1.687 minutes, while PTE-2 highlighted a peak at 4.082 minutes, representing distinct component profiles. In contrast, the PTE-Recover sample exhibits multiple smaller peaks alongside a prominent peak, which may show the presence of degradation products or impurities [23].
The PTE-2 fraction was preceded for cytoxicity assay with HeLa cell line.
The MTT assay results show the cytotoxic effects of PTE-2 across a range of concentrations (50–500 µg/ml), with an IC50 value of 318.85 µg/ml. This IC50 value advocates that PTE-2 exhibit cytotoxicity; requiring a concentration of approximately 318.85 µg/ml to inhibit 50% of cell viability. The percentage cytotoxicity increases with concentration, ranging from 16.228% ± 0.439 at 50 µg/ml to 95.748% ± 0.49 at 500 µg/ml, demonstrating a dose-dependent relationship. This trend aligns with previous studies on plant-derived extracts, which often display dose-dependent cytotoxicity due to the presence of bioactive compounds such as polyphenols and flavonoids [24]. The observed cytotoxicity could be credited to the antioxidant properties of blackberry constituents, which may encourage apoptosis or necrosis at elevated concentrations. The induction of apoptosis by polyphenolic compounds is associated with the caspase family and the effect on p53, Bax, Bcl-2, Bcl-X1 levels [25]. The IC50 value of 318.85 µg/ml offers a benchmark for future studies to optimize the therapeutic window of blackberry extracts, potentially for applications in cancer research or antimicrobial therapies.
The data show the relative gene expression levels of M-CSF and GM-CSF in Hela cells under different conditions, as determined by the relative real time PCR. The fold change analysis reveals a significant 5.29-fold up regulation of M-CSF in the treated sample (PTE-2 TREATMENT to HeLa cell line) compared to the untreated control (Hela cells), while GM-CSF exhibits a minimal change of 1.04-fold across both conditions. In an another study macrophage-colony stimulating factor was significantly amplified by cisplatin and 6-mercaptopurine in HT-29 [26] This marked increase in M-CSF expression suggests a robust response to the treatment, potentially indicative of enhanced macrophage colony-stimulating factor activity, which is known to play a critical role in monocyte and macrophage survival [27]. In contrast, the expression of GM-CSF implies that this granulocyte-macrophage colony-stimulating factor may not be significantly influenced by the treatment, highlighting a selective regulatory mechanism.
The observed differential regulation may reflect distinct signaling pathways or transcriptional controls activated by the BBM condition, warranting further investigation into the underlying molecular mechanisms.
Conclusion
The therapeutic and molecular analysis of natural pterostilbene on the HeLa cell line highlights its potential as a promising anti-cancer agent. Pterostilbene demonstrated significant cytotoxic activity against HeLa cells; this study contributes to the growing body of evidence supporting the use of natural bioactive compounds like pterostilbene in cancer therapeutics. Our findings suggests that PTE-2 ( fraction containing Pterostilbene) possesses significant dose dependent cytotoxicity activity against HeLa cell line, which warrants further investigation to elucidate its mechanism of action. Further studies focusing on apoptosis induction, cell cycle arrest are recommended to comprehensively assess the anticancer properties of PTE-2 fraction. Moreover, Efficacy of Pterostilbene, combined with its natural origin suggests its potential for development as a novel therapeutic agent for cervical cancer. Pterostilbene can be used in combination with other naturally occurring anticancer compounds or can be tried with other anticancer drugs. Further in-depth studies, including in vivo models and clinical trials, are necessary to establish its mechanistic pathways and optimize its use in integrative cancer treatment
Acknowledgement
The authors wish to express their gratitude to the Sumerian Scriptum Synthesis Publisher (SSSP), Baqubah 32001, Diyala Province, Iraq, for their financial support of this work through the Large Research Group Project under Grant No. (G.N.R.2022.SSSP). for further details, please refer to (doi.org/10.62909/G.N.R.2022.SSSP).
Conflict of Interest
The authors declare there are no conflicts of interests regarding the publication of this manuscript.
References
- Brown JS, Amend SR, Austin RH, et al. Updating the definition of cancer. Molecular Cancer Research 21 (2023): 1142-1147.
- Gullett NP, Amin AR, Bayraktar S, et al. Cancer prevention with natural compounds. Seminars in Oncology 37 (2010): 258-280.
- Obrador E, Salvador-Palmer R, Jihad-Jebbar A, et al. Pterostilbene in cancer therapy. Antioxidants 10 (2021): 492.
- Kent A. HPV vaccination and testing. Reviews in Obstetrics and Gynecology 3 (2010): 33.
- Denny L. Cervical cancer: prevention and treatment. Discovery Medicine 14 (2012): 125-131.
- Li Z, Zhang J, You S, et al. Pterostilbene upregulates MICA/B via the PI3K/AKT signaling pathway to enhance the capability of natural killer cells to kill cervical cancer cells. Experimental Cell Research 435 (2024): 113933.
- McCormack D, McFadden D. A review of pterostilbene antioxidant activity and disease modification. Oxidative Medicine and Cellular Longevity (2013): 575482.
- Alosi JA, McDonald DE, Schneider JS, et al. Pterostilbene inhibits breast cancer in vitro through mitochondrial depolarization and induction of caspase-dependent apoptosis. The American Journal of Surgery 211 (2016): 466-470.
- Zhang B, Wang XQ, Chen HY, et al. Involvement of the Nrf2 pathway in the regulation of pterostilbene-induced apoptosis in HeLa cells via ER stress. Journal of Pharmacological Sciences 126 (2014): 216-229.
- Baidya B, Gupta SK, Mukherjee T. An extraction-based verification methodology for MEMS. Journal of Microelectromechanical Systems 11 (2002): 2-11.
- Kokate CK, Purohit AP, Gokhale SB. Textbook of Pharmacognosy. Nirali Prakashan (2000): 1-4.
- Tangco JVV, Angustia DA, Jelynne PT. Nutritional analysis, phytochemical screening and total phenolic content of Basella alba leaves from Philippines. International Journal of Pharmacognosy and Phytochemical Research 7 (2015): 103-110.
- Parthasarathy S, Bin Azizi J, Ramanathan S, et al. Evaluation of antioxidant and antibacterial activities of aqueous, methanolic and alkaloid extracts from Mitragyna speciosa (Rubiaceae family) leaves. Molecules 14 (2009): 3964-3974.
- Athavale A, Jirankalgikar N, Nariya P, et al. Evaluation of in vitro antioxidant activity of panchagavya: a traditional ayurvedic preparation. International Journal of Pharmaceutical Sciences and Research 3 (2012): 2543-2549.
- Benzie IFF, Strain JJ. The Ferric Reducing Ability of Plasma (FRAP) as a measure of “antioxidant power”: The FRAP assay. Analytical Biochemistry 239 (1996): 70-76.
- Kumar B, Maharana BR, Thakre B, et al. 18S rRNA gene-based piroplasmid PCR: an assay for rapid and precise molecular screening of Theileria and Babesia species in animals. Acta Parasitologica 67 (2022): 1697-1707.
- Uemura H, Hasumi H, Ishiguro H, et al. Renin-angiotensin system is an important factor in hormone refractory prostate cancer. The Prostate 66 (2006): 822-830.
- Sellappan S, Akoh CC, Krewer G. Phenolic compounds and antioxidant capacity of Georgia-grown blueberries and blackberries. Journal of Agricultural and Food Chemistry 50 (2002): 2432-2438.
- Harborne JB. Phytochemical Methods: A Guide to Modern Techniques of Plant Analysis. Springer (1998).
- Huang WY, Zhang HC, Liu WX, et al. Survey of antioxidant capacity and phenolic composition of blueberry, blackberry and strawberry in Nanjing. Journal of Zhejiang University Science B 13 (2012): 94-102.
- Kahkonen P, Hopia AI, Vuorela HJ, et al. Antioxidant activity of plant extracts containing phenolic compounds. Journal of Agricultural and Food Chemistry 47 (1999): 3954-3962.
- Li J, Shi C, Shen D, et al. Composition and antioxidant activity of anthocyanins and non-anthocyanin flavonoids in blackberry from different growth stages. Foods 11 (2022): 2902.
- Blessy M, Patel RD, Prajapati PN, et al. Development of forced degradation and stability indicating studies of drugs: A review. Journal of Pharmaceutical Analysis 4 (2014): 159-165.
- Rao YK, Geethangili M, Fang SH, et al. Antioxidant and cytotoxic activities of naturally occurring phenolic and related compounds: a comparative study. Food and Chemical Toxicology 45 (2007): 1770-1776.
- Wang E, Liu Y, Xu C, et al. Antiproliferative and proapoptotic activities of anthocyanin and anthocyanidin extracts from blueberry fruits on B16-F10 melanoma cells. Food and Nutrition Research 61 (2017): 1325308.
- Calatayud S, Warner T, Mitchell J. Modulation of colony stimulating factor release and apoptosis in human colon cancer cells by anticancer drugs. British Journal of Cancer 86 (2002): 1316-1321.
- Stanley ER, Chitu V. CSF-1 receptor signaling in myeloid cells. Cold Spring Harbor Perspectives in Biology 6 (2014): a021857.


 Impact Factor:
* 5.3
Impact Factor:
* 5.3
 Acceptance Rate:
75.63%
Acceptance Rate:
75.63%
 Time to first decision: 10.4 days
Time to first decision: 10.4 days
 Time from article received to acceptance:
2-3 weeks
Time from article received to acceptance:
2-3 weeks
