Acute Allergic Reactions and Severe Anaphylaxis: Underlying Causes, Management Strategies, and Future Directions
Aris Ellorin1, Devendra K. Agrawal1*
1Department of Translational Research, College of Osteopathic Medicine of the Pacific, Western University of Health Sciences, Pomona, California 91766, USA
*Corresponding author: Devendra K. Agrawal, MSc, PhD (Biochem), PhD (Med Sci), MBA, MS (ITM), FAAAAI, FAHA, FAPS, FIACS Director and Professor, Department of Translational Research, Western University of Health Sciences 309 E. Second Street, Pomona, California 91766, USA
Received: 28 March 2026; Accepted: 02 April 2026; Published: 06 April 2026
Article Information
Citation: Aris Ellorin, Devendra K. Agrawal. Acute Allergic Reactions and Severe Anaphylaxis: Underlying Causes, Management Strategies, and Future Directions. Archives of Internal Medicine Research. 9 (2026): 64-72.
View / Download Pdf Share at FacebookAbstract
Anaphylaxis represents the most severe and potentially fatal manifestation of allergic disease, characterized by sudden multi-system involvement and rapid hemodynamic compromise. While management protocols have improved, global incidence continues to rise and preventable deaths persist — driven largely by delayed epinephrine administration and inadequate long-term follow-up. This narrative review synthesizes current evidence on the immunological and non-immunological mechanisms underlying anaphylaxis, evaluates diagnostic criteria from major allergy societies, and appraises both immediate and long-term treatment strategies. Contributing factors to persistent morbidity — including epinephrine underuse, barriers to autoinjector access, and the emergence of biphasic and refractory phenotypes — are examined in depth. Advances in novel epinephrine delivery platforms, biologic therapies targeting the IgE and cytokine axes, and immunomodulatory strategies including oral and venom immunotherapy are highlighted as promising avenues for improving outcomes. This review also underscores the need for validated predictive biomarkers, equitable device access, and prospective trials to close the gaps that continue to drive preventable mortality.
Keywords
Allergic diseases; Anaphylaxis; Emergency medicine; Epinephrine; IgE; Immune tolerance; Immunotherapy; Mast cells; MRGPRX2; Platelet-Activating Factor
Article Details
1. Introduction
Allergic diseases affect approximately up to 30% of the global population and impose a substantial public health burden [1-8]. Among them, anaphylaxis is the most severe manifestation, characterized by acute multi-system involvement and rapid hemodynamic deterioration [9-12]. The case-fatality rate, while low at 0.25–0.33% of hospitalizations, translates to 63–99 deaths annually in the United States alone [13]. A recent systematic review reported a 7.4% annual increase in all-cause anaphylaxis incidence globally between 1990 and 2017, with pediatric rates rising disproportionately in suburban compared to urban settings [14,15].
Timely intramuscular epinephrine remains the only intervention consistently shown to reduce morbidity and mortality, yet community underuse is widespread — documented in up to 64% of pre-hospital events [16,17]. Contributing factors include poor autoinjector carriage, inadequate patient and provider training, socioeconomic barriers, and fear of adverse effects [16-19]. The emergence of biphasic and refractory anaphylaxis phenotypes further complicates management, as recurrent or epinephrine-resistant reactions carry substantially higher morbidity [20].
Recent advances offer meaningful clinical progress. Next-generation intranasal epinephrine formulations and high-dose autoinjectors address delivery barriers, while biologics targeting IgE and cytokine axes and allergen immunotherapies inducing sustained tolerance expand long-term options [11, 21-23]. Precision biomarkers such as hereditary α-tryptasemia, platelet-activating factor (PAF) acetylhydrolase deficiency, and elevated baseline serum tryptase, and abnormal density and function of immune cell functions have emerged as candidates for improved individual risk stratification [24-37]. This review highlights these developments and outlines a research agenda for areas where critical gaps remain.
2. Epidemiology and Disease Burden
2.1 Prevalence and Incidence
The lifetime prevalence of anaphylaxis is estimated between 1.6% and 5.1% [16,38]. Global incidence varies substantially by geography and methodology: population-based studies report rates from approximately 26 per 100,000 person-years in South Korea to 82.5 per 100,000 in Western Australia [39,40]. A recent systematic review and meta-analysis by Pühringer and colleagues confirmed a 7.4% annual increase in all-cause anaphylaxis incidence worldwide from 1990 to 2017, with food-triggered cases in pediatric populations demonstrating particularly steep temporal trends [14,41]. Perioperative anaphylaxis, though less common, carries a mortality rate of up to 4.8% and occurs in approximately 100 per million procedures [42].
2.2 Underuse of Epinephrine
A persistent and alarming gap exists between the severity of anaphylaxis and real-world epinephrine administration. Only 21% of children and 7% of adults experiencing anaphylaxis in community settings receive epinephrine prior to hospital arrival [43]. Underuse is similarly documented within emergency departments, where guideline-concordant first-line epinephrine administration remains inconsistent [44]. This disparity reflects multiple intersecting barriers: cost and limited availability of autoinjectors, patient and caregiver anxiety regarding injection, misconceptions about contraindications, and insufficient training in device use [18,19,44]. Addressing these barriers is a public health priority, as pre-hospital epinephrine administration is consistently associated with reduced biphasic risk and shorter emergency department stays [20,45].
3. Diagnostic Criteria and Clinical Definitions
Consensus definitions from the World Allergy Organization (WAO) and NIAID/FAAN require the acute onset of characteristic symptoms involving at least two organ systems following exposure to a likely allergen, or isolated hypotension after a known trigger [38]. The 2020 WAO guidance refined these criteria to better differentiate anaphylaxis from isolated cutaneous or respiratory reactions [38]. This update also emphasized recognition of atypical presentations, particularly in infants, older adults, and perioperative settings [38]. Ongoing discussions about further revision include whether and how to incorporate cardiovascular-specific entities such as Kounis syndrome, in which coronary artery spasm or acute myocardial infarction is precipitated by the anaphylactic cascade [46].
Diagnosing anaphylaxis remains clinically challenging, as evidenced by the subtle but meaningful differences in criteria across major authoritative bodies (see Table 1) [38]. These discrepancies complicate epidemiological comparisons and contribute to under-recognition in atypical presentations. Ongoing efforts to harmonize definitions internationally will be essential for advancing both research reproducibility and consistent clinical practice [12,38].
4. Pathophysiology and Molecular Mechanisms
4.1 IgE-Mediated Anaphylaxis
Anaphylaxis is primarily driven by an immunoglobulin E (IgE)-mediated mechanism in previously sensitized individuals [47]. Upon re-exposure to a causative allergen, cross-linking of allergen-specific IgE bound to high-affinity FcεRI receptors on mast cells and basophils triggers rapid degranulation [47]. This releases histamine, tryptase, and proteoglycans into the systemic circulation [47-49].
Table 1: Anaphylaxis criteria by guidelines: Side-by-side comparison of consensus criteria for the clinical diagnosis of anaphylaxis from the World Allergy Organization (WAO), NIAID/FAAN, and other leading societies. The diagram emphasizes clinically relevant differences in organ system thresholds, recognition of atypical presentations, and conditional hypotension criteria. This comparison illustrates the ongoing need for harmonized definitions to support epidemiological research and consistent clinical practice [21,38,46].

Downstream signaling cascades subsequently generate prostaglandins, leukotrienes, and platelet-activating factor (PAF), which collectively drive bronchoconstriction, vascular leak, and amplified systemic inflammation [47-49]. Elevated PAF levels have been associated with greater anaphylaxis severity [26]. Effectors cells are recruited by TNF-α resulting in the sustenance of multi-organ symptoms including urticaria, angioedema, bronchospasm, hypotension, and gastrointestinal distress [47]. This cascade helps explain why antihistamines alone are inadequate and why epinephrine — acting across multiple receptor classes simultaneously — remains the cornerstone first-line intervention [49].
4.2 Non-IgE Mediated Pathways and Emerging Mechanisms
While IgE-mediated anaphylaxis is the most prevalent form, the reaction can also occur through several IgE-independent mechanisms, including direct mast cell activation, immune complex–driven complement activation, cytotoxic reactions, neuropeptide release, and T-cell–mediated pathways [50]. Research using murine models has illuminated the role of IgG antibodies and FcγR-expressing effector cells — including basophils, macrophages, and neutrophils — in triggering PAF-mediated anaphylaxis [50]. However, the exact significance of IgG-mediated mechanisms in human anaphylaxis remains incompletely defined.
At the molecular level, tyrosine kinases including Lyn, Syk, and Fyn modulate FcεRI signal transduction, and their dysregulation may contribute to exaggerated mast cell responses [48]. Activating mutations such as the D816V variant in the KIT tyrosine kinase — frequently observed in systemic mastocytosis — predispose affected individuals to recurrent, unexplained anaphylaxis through constitutive mast cell activation [24]. Emerging mediators including sphingosine-1-phosphate and nitric oxide are also gaining recognition for their roles in anaphylactic physiology [50]. Separately, the MAS-related G protein-coupled receptor member X2 (MRGPRX2) has been identified as a non-IgE target through which neuromuscular blocking agents and other drugs directly activate mast cells, providing a mechanistic basis for drug-induced anaphylaxis that bypasses the classical IgE sensitization requirement [50]. This evolving mechanistic landscape suggests that future therapeutic strategies and diagnostic biomarker panels may increasingly be individualized to the predominant pathway driving each patient’s reaction.
5. Triggers and Risk Amplifiers
Foods constitute the predominant trigger of anaphylaxis in pediatric patients, with peanuts, tree nuts, shellfish, and wheat accounting for most cases [51]. In adults, drug reactions — particularly to β-lactam antibiotics, NSAIDs, and radiocontrast media — are leading causes, with biologics representing an increasingly important emerging category [52]. Venom-induced anaphylaxis from Hymenoptera stings contributes disproportionately to anaphylaxis fatalities across all age groups [53].
Several comorbidities and pharmacological exposures substantially amplify reaction severity. Asthma, cardiovascular disease, and obesity increase the risk of fatal outcomes [12]. Concurrent β-blocker or ACE-inhibitor therapy blunts the physiological response to epinephrine and worsens refractory presentations [12]. Clonal mast-cell disorders — including systemic mastocytosis and hereditary α-tryptasemia — represent the most biologically distinct risk-amplifying conditions, predisposing affected individuals to unprovoked or disproportionately severe reactions through constitutive mediator release [24,54].
6. Clinical Phenotypes
Anaphylaxis manifests across three clinically distinct phenotypes with important management implications. Uniphasic reactions, the most common, resolve completely with treatment and do not recur [20]. Biphasic anaphylaxis involves recurrence of symptoms 1–48 hours after apparent resolution [20]. Reported incidence ranges from 4% to 20%, with large emergency department cohorts estimating the rate at approximately 7–16% [20,55,56]. Independent predictors of biphasic recurrence include delayed initial epinephrine administration of more than 60 minutes from onset, drug or idiopathic triggers, and angioedema at presentation [20]. Supporting this, a nationwide cohort study from U.S. emergency departments found that patients who received prehospital epinephrine had significantly lower odds of biphasic reactions and shorter emergency department stays, reinforcing early intervention as both therapeutic and preventive [45].
Refractory anaphylaxis is defined by persistent or progressive symptoms despite at least two appropriately dosed intramuscular epinephrine injections [20]. This phenotype is associated with delayed presentation, β-blocker use, obesity, and severe initial manifestations including cardiovascular collapse [12,57]. Cases meeting this threshold require escalation to intravenous epinephrine infusion, vasopressors, and advanced resuscitation measures [12,57]. Data from the European Anaphylaxis Registry indicate that refractory cases are underreported and that standardized recognition and management protocols remain inconsistently applied across institutions [57].
7. Immediate Management
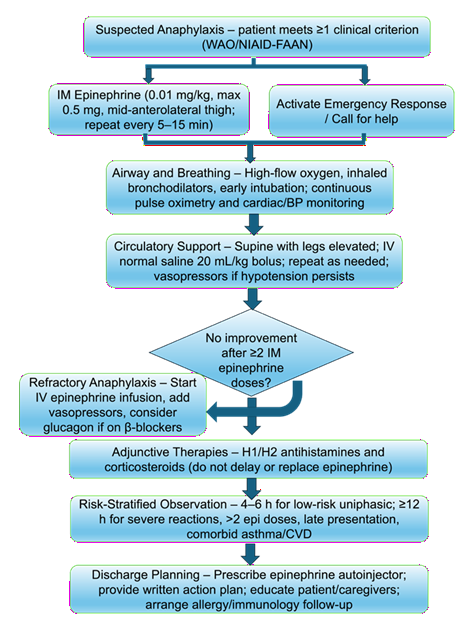
The cornerstone of acute anaphylaxis management is prompt intramuscular epinephrine [12]. Delivered at 0.01 mg/kg (maximum 0.5 mg) into the mid-anterolateral thigh, epinephrine simultaneously reverses bronchoconstriction, restores vascular tone, and suppresses further mediator release [12]. Early administration is associated with reduced biphasic risk and shorter emergency department observation time [20]. Despite this evidence, epinephrine remains underused in both community and hospital settings [16,18]. Provider anxiety, cost barriers, and persistence of misconceptions about side effects have been identified as modifiable targets for intervention [16,19,44] (Figure 1).
Airway management proceeds in parallel with epinephrine administration. High-flow supplemental oxygen should be initiated immediately [12]. Patients with bronchospasm benefit from inhaled bronchodilators, while those with impending airway compromise require early intubation [12]. Ketamine is a preferred induction agent in this context given its bronchodilatory properties and hemodynamic stability profile [58-60]. Circulatory support requires supine positioning with leg elevation to optimize venous return, followed by isotonic crystalloid boluses of 20 mL/kg for persistent hypotension after epinephrine, with vasopressor escalation if cardiovascular collapse remains refractory [12,60] (Figure 1).
Figure 1: Flow chart illustrating recommended steps for the acute management of anaphylaxis, adapted from current WAO and emergency medicine practice guidelines. Key interventions include prompt intramuscular epinephrine, airway support (high-flow oxygen, bronchodilators, intubation as indicated), circulatory stabilization (supine positioning, isotonic fluid bolus, vasopressors for refractory hypotension), adjunctive antihistamines and corticosteroids, and risk-stratified observation periods [12,38,60].
Adjunctive agents — including H1 and H2 antihistamines and corticosteroids — address cutaneous symptoms such as urticaria and may reduce late-phase inflammatory activity, but neither class prevents biphasic reactions or substitutes for epinephrine [12,16]. Observation following clinical stabilization is essential to detect symptom recurrence. Evidence-based protocols support 4–6 hours of monitoring for low-risk uniphasic presentations [12,16]. Extended observation of at least 12 hours is recommended for severe initial reactions, delayed epinephrine administration, or cases with significant comorbidities [12,16].
8. Long-Term Management and Secondary Prevention
Long-term anaphylaxis management requires a personalized, multi-pronged strategy centered on trigger avoidance, immunotherapy, and structured education. Allergen avoidance remains the most reliably effective prevention strategy [12,61-63]. Accurate trigger identification, often requiring referral to an allergist for skin-prick testing, specific IgE measurement, and controlled challenges, is foundational to developing tailored avoidance plans [12,63].
Allergen-specific immunotherapy (AIT) offers the most durable long-term risk reduction for select populations. Venom immunotherapy (VIT) provides a 90% reduction in the risk of future anaphylaxis from Hymenoptera stings and is considered standard of care for patients with a documented history of venom-triggered systemic reactions [53]. Oral immunotherapy (OIT) has expanded options for patients with peanut and other food allergies, enabling clinically meaningful desensitization through carefully titrated exposure protocols [23]. However, adherence to daily maintenance dosing is critical, and clinicians must monitor closely for treatment-emergent eosinophilic esophagitis [23].
Biologic therapies have emerged as an important adjunct for patients with severe, recurrent, or refractory allergic disease. Omalizumab, a monoclonal anti-IgE antibody, reduces free IgE levels and raises the allergen threshold required to trigger mast cell activation [64,65]. Dupilumab and anti-IL-5 agents similarly modulate the Th2-driven inflammatory milieu underlying allergic sensitization [43]. Reviews of clinical trial data suggest that these agents can meaningfully reduce reaction frequency and corticosteroid burden during allergen immunotherapy [43]. However, optimal patient selection, treatment duration, and combination sequencing with AIT remain active areas of investigation [64].
Novel epinephrine delivery platforms represent a meaningful advance for device adherence and patient acceptability. Intranasal epinephrine formulations have demonstrated pharmacokinetic profiles comparable to standard autoinjectors in clinical studies [21]. High dose autoinjectors provide more rapid peak plasma concentrations in patients for whom standard dosing may be inadequate [22]. Patient and caregiver preference data suggest that many individuals favor needle-free delivery methods, indicating that these platforms may help reduce some behavioral barriers to pre-hospital epinephrine use. [21,22].
Education and structured emergency action plans constitute the behavioral infrastructure of secondary prevention. Comprehensive training programs increase autoinjector carriage and correct technique, and community-level educational initiatives have been associated with improved preparedness and guideline-concordant management in community anaphylaxis [63].
Advances in pharmacogenomics and biomarker science are helping to individualize care. Genetic screening for hereditary α-tryptasemia identifies patients at higher risk for severe reactions [24]. The KIT D816V mutation can guide decisions about the need for prolonged hospital observation and personalized monitoring strategies in mastocytosis-related anaphylaxis [24]. Dynamic biomarkers including serial tryptase levels and PAF concentrations further refine individual risk assessment in the acute setting [25,26].
9. Discussion
This review addresses a clinically relevant and evolving topic: the persistent gap between the availability of effective treatments and their real-world uptake [16-18]. Several prior reviews have addressed anaphylaxis mechanisms or management in isolation, but the present synthesis integrates pathophysiology, phenotypic classification, emerging therapeutics, and patient-level barriers within a unified framework [48,50].
A particularly actionable implication concerns the consistent failure to administer epinephrine promptly. Pre-hospital administration remains below 40% in most community settings, and within emergency departments, guideline-concordant first-line epinephrine use is inconsistently documented [16,17]. This is not exclusively a knowledge failure — it reflects structural gaps in autoinjector accessibility, prescribing rates, and follow-up education [19]. Policy interventions modeled on public access defibrillation programs may offer a scalable template for increasing community-level epinephrine availability [19].
The mechanistic findings reviewed here also suggest important precision medicine opportunities. The identification of MRGPRX2-mediated mast cell activation in drug-induced anaphylaxis and the role of KIT mutations in mastocytosis-related anaphylaxis collectively indicate that anaphylaxis is not a single immunological entity but a syndromic spectrum [24,50]. As biomarker science advances, diagnostic panels encompassing PAF levels, tryptase kinetics, and genetic variants may enable individualized risk stratification that goes beyond current binary treatment algorithms [25,26].
A notable limitation of this review is the variable quality and heterogeneity of underlying studies [20,55,56]. Many key estimates — including biphasic reaction rates and epinephrine underuse figures — derive from retrospective, single-center, or regionally limited cohorts [20,55,56]. Prospective, multi-site, population-representative studies are needed to generate estimates robust enough to inform guideline revision [12,14].
10. Conclusion
Anaphylaxis management has advanced considerably, yet the burden of preventable deaths remains unacceptably high [13,16]. Bridging the gap between current evidence and clinical practice demands simultaneous attention to biomedical, behavioral, and systemic dimensions of care [12,20]. The field requires prospective validation of biphasic and refractory risk prediction tools, large multi-center cohort studies to validate dynamic biomarker panels for bedside use, and rigorous randomized trials to define optimal sequences of biologic and immunotherapeutic interventions [23-26,65]. Equally important are policy-level efforts to ensure equitable autoinjector access and the integration of structured emergency action plans into every patient's discharge planning [19,63]. Addressing these priorities collectively represents the most direct path to improving outcomes for patients with severe allergic disease worldwide.
Key Points
- • Anaphylaxis incidence increases approximately 7.4% annually, with pediatric rates rising disproportionately in suburban compared to urban settings
- • Pre-hospital epinephrine is consistently associated with reduced biphasic risk and shorter emergency department stays, yet underuse persists in up to two-thirds of community events
- • Delayed epinephrine (>60 min), drug or idiopathic triggers, and angioedema are the strongest independent predictors of biphasic recurrence
- • Defined by failure of ≥2 intramuscular doses, refractory anaphylaxis requires urgent escalation to intravenous epinephrine and vasopressors
- • Venom immunotherapy reduces future sting anaphylaxis risk by 90%; OIT and anti-IgE/IL-4/IL-5 biologics reduce food-allergic reaction frequency and threshold
- • Intranasal epinephrine and high-dose autoinjectors show pharmacokinetic profiles comparable to standard devices, with emerging evidence suggesting patient preference for needle-free delivery
- • Autoinjector cost, limited public access, and training disparities fuel preventable deaths, particularly in low-resource settings
- • Prospective biomarker validation, population-representative epidemiological studies, and rigorous immunotherapy sequencing trials are the most critical unmet needs
Acknowledgement:
The work reported in this publication was supported by the National Institute of Allergy and Infectious Diseases of the National Institutes of Health under Award number R25AI179582. Aris Ellorin received a 2025 summer research education fellowship under this award. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Competing interests:
Both authors have read the manuscript and declare no conflict of interest. No writing assistance was utilized in the production of this manuscript.
Consent for publication:
Both authors have read the manuscript and consented for publication.
References
- Alska E, Doligalska A, Napiórkowska-Baran K, et al. Global Burden of Allergies: Mechanisms of Development, Challenges in Diagnosis, and Treatment. Life (Basel) 15 (2025): 878.
- Agrawal DK, Shao Z. Pathogenesis of allergic airway inflammation. Curr Allergy Asthma Rep, 10 (2010): 39-48.
- Bharadwaj AS, Bewtra AK, Agrawal DK. Dendritic cells in allergic airway inflammation. Can J Physiol Pharmacol 85 (2007): 686-699.
- Hall S, Agrawal DK. Key mediators in the immunopathogenesis of allergic asthma. Int Immunopharmacol 23 (1): 316-329.
- Nabipur L, Mouawad M, Agrawal DK. Bioaerosols and Airway Diseases: Mechanisms of Epithelial Dysfunction, Immune Activation, and Strategies for Exposure Mitigation. Arch Intern Med Res 8 (2025): 178-191.
- Kim CK, Agrawal DK. Glucocorticoid Insensitivity in Severe Asthma: Underlying Molecular Mechanisms, Challenges, and Emerging Therapies. Arch Intern Med Res 8 (2025): 107-120.
- Chaudhary F, Lee W, Escander T, Agrawal DK. Exploring the Complexities of Atopic Dermatitis: Pathophysiological Mechanisms and Therapeutic Approaches. J Biotechnol Biomed 7 (2024): 314-328.
- Hall SC, Agrawal DK. Increased TREM-2 expression on the subsets of CD11c+ cells in the lungs and lymph nodes during allergic airway inflammation. Sci Rep 7 (2017): 11853.
- Yazdani AN, DeMarco N, Patel P, et al. Adverse Hematological Effects of COVID-19 Vaccination and Pathomechanisms of Low Acquired Immunity in Patients with Hematological Malignancies. Vaccines (Basel) 11 (2023): 662.
- Quirk SK, Rainwater E, Shure AK, Agrawal DK. Vitamin D in atopic dermatitis, chronic urticaria and allergic contact dermatitis. Expert Rev Clin Immunol 12 (2016): 839-847.
- Zhang M, Murphy RF, Agrawal DK. Decoding IgE Fc receptors. Immunol Res 37 (2007): 1-16.
- Golden DBK, Wang J, Waserman S, et al. Anaphylaxis: A 2023 practice parameter update. Ann Allergy Asthma Immunol 132 (2024): 124-176.
- Ma L, Danoff TM, Borish L. Case fatality and population mortality associated with anaphylaxis in the United States. J Allergy Clin Immunol 133 (2014): 1075-1083.
- Pühringer V, Jilma B, Herkner H. Population-based incidence of all-cause anaphylaxis and its development over time: a systematic review and meta-analysis. Front Allergy 4 (2023): 1249280.
- Gurkha D, Podolsky R, Sethuraman U, et al. Comparison of anaphylaxis epidemiology between urban and suburban pediatric emergency departments. BMC Pediatr 23 (2023): 85.
- Shaker MS, Wallace DV, Golden DBK, et al. Anaphylaxis-a 2020 practice parameter update, systematic review, and Grading of Recommendations, Assessment, Development and Evaluation (GRADE) analysis. J Allergy Clin Immunol 145 (2020): 1082-1123.
- Dami F, Enggist R, Comte D, et al. Underuse of Epinephrine for the Treatment of Anaphylaxis in the Prehospital Setting. Emerg Med Int 15 (2022): 5752970.
- Glassberg B, Nowak-Wegrzyn A, Wang J. Factors contributing to underuse of epinephrine autoinjectors in pediatric patients with food allergy. Ann Allergy Asthma Immunol 126 (2021): 175-179.e3.
- Baichoo N, Fishbein J, Ponda P. Access to Epinephrine Autoinjectors, the Patient Perspective. Allergy. 2025 Oct; 80 (2025): 2879-2881.
- Pongdee T, Dribin TE, et al. Incidence, predictors, and treatment outcomes of biphasic anaphylactic reactions: a multicenter study. Asian Pac J Allergy Immunol 42 (2024): 97-104.
- Tal Y, Segal N, Levi-Schaffer F, et al. A 13.2 mg epinephrine intranasal spray demonstrates comparable pharmacokinetics and pharmacodynamics to the intramuscular autoinjector and manual syringe. J Allergy Clin Immunol Pract 12 (2024): 101-110.
- Dhami S, Panesar SS, Roberts G, et al. Pharmacokinetics and pharmacodynamics of adrenaline autoinjectors: a systematic review. Clin Exp Allergy 51 (2021) :408-420.
- Anderson B, Wong L, Adlou B, et al. Oral Immunotherapy in Children: Clinical Considerations and Practical Management. J Asthma Allergy 14 (2021): 1497-1510.
- Giannetti MP, Weller E, Bormans C, et al. Hereditary alpha-tryptasemia in 101 patients with mast cell activation-related symptomatology including anaphylaxis. Ann Allergy Asthma Immunol 126 (2021): 655-660.
- Khalaf R, Prosty C, Davalan W, et al. Diagnostic Utility of Biomarkers in Anaphylaxis: A Systematic Review and Meta-Analysis. J Allergy Clin Immunol Pract 13 (2025): 1342-1349.e12.
- Galvan-Blasco P, Gil-Serrano J, Sala-Cunill A. New Biomarkers in Anaphylaxis (Beyond Tryptase). Curr Treat Options Allergy 9 (2022): 303-322.
- Tamura N, Agrawal DK, Suliaman FA, et al. Effects of platelet activating factor on the chemotaxis of normodense eosinophils from normal subjects. Biochem Biophys Res Commun 142 (1987): 638-644.
- Tamura N, Agrawal DK, Townley RG. Leukotriene C4 production from human eosinophils in vitro. Role of eosinophil chemotactic factors on eosinophil activation. J Immunol 141 (1988): 4291-4297.
- Hall SC, Agrawal DK. Toll-like receptors, triggering receptor expressed on myeloid cells family members and receptor for advanced glycation end-products in allergic airway inflammation. Expert Rev Respir Med 10 (2016): 171-184.
- McGee HS, Yagita H, Shao Z, Agrawal DK. Programmed Death-1 antibody blocks therapeutic effects of T-regulatory cells in cockroach antigen-induced allergic asthma. Am J Respir Cell Mol Biol 43 (2010): 432-442.
- Shao Z, Makinde TO, McGee HS, et al. Fms-like tyrosine kinase 3 ligand regulates migratory pattern and antigen uptake of lung dendritic cell subsets in a murine model of allergic airway inflammation. J Immunol 183 (2009): 7531-7538.
- McGee HS, Agrawal DK. Naturally occurring and inducible T-regulatory cells modulating immune response in allergic asthma. Am J Respir Crit Care Med 180 (2009): 211-225.
- Makinde TO, Steininger R, Agrawal DK. NPY and NPY receptors in airway structural and inflammatory cells in allergic asthma. Exp Mol Pathol 94 (2013): 45-50.
- Gupta GK, Agrawal DK. CpG oligodeoxynucleotides as TLR9 agonists: therapeutic application in allergy and asthma. BioDrugs 24 (2010): 225-235.
- Agrawal DK, Cheng G, Kim MJ, et al. Interaction of suplatast tosilate (IPD) with chloride channels in human blood eosinophils: a potential mechanism underlying its anti-allergic and anti-asthmatic effects. Clin Exp Allergy 38 (2008): 305-312.
- Gaurav R, Agrawal DK. Clinical view on the importance of dendritic cells in asthma. Expert Rev Clin Immunol 9 (2013): 899-919.
- Gaurav R, Bewtra AK, Agrawal DK. Chloride Channel 3 Channels in the Activation and Migration of Human Blood Eosinophils in Allergic Asthma. Am J Respir Cell Mol Biol 53 (2015): 235-245.
- Cardona V, Ansotegui IJ, Ebisawa M, et al. World allergy organization anaphylaxis guidance 2020. World Allergy Organ J 13 (2020): 100472.
- Jeong K, Lee JD, Kang DR, et al. A population-based epidemiological study of anaphylaxis using national big data in Korea: trends in age-specific prevalence and epinephrine use in 2010-2014. Allergy Asthma Clin Immunol 14 (2018): 31.
- Liew WK, Williamson E, Tang ML. Anaphylaxis fatalities and admissions in Australia. J Allergy Clin Immunol 123 (2009): 434-442.
- Baseggio Conrado A, Ierodiakonou D, Gowland MH, et al. Food anaphylaxis in the United Kingdom: analysis of national data, 1998-2018. BMJ. 2021 Feb 17;372:n251. Erratum in: BMJ 372 (2021): n733.
- Pouessel G, Tacquard C, Tanno LK, et al. Anaphylaxis mortality in the perioperative setting: Epidemiology, elicitors, risk factors and knowledge gaps. Clin Exp Allergy 54 (2024): 11-20.
- Miles LM, Ratnarajah K, Gabrielli S, et al. Community use of epinephrine for the treatment of anaphylaxis: a review and meta-analysis. J Allergy Clin Immunol Pract 9 (2021): 2321-2333.
- Choi YJ, Kim J, Jung JY, et al. Underuse of Epinephrine for Pediatric Anaphylaxis Victims in the Emergency Department: A Population-based Study. Allergy Asthma Immunol Res 11 (2019): 529-537.
- Hlady AL, Weinman AF, Zhang Y, et al. Outcomes associated with prehospital epinephrine in adult and pediatric patients with anaphylaxis. Ann Allergy Asthma Immunol 133 (2024): 592-599.e8.
- Lieberman JA, Bingemann TA, Wang J. Diagnostic Challenges in Anaphylaxis. J Allergy Clin Immunol Pract 8 (2020): 1177-1184.
- Metcalfe DD, Peavy RD, Gilfillan AM. Mechanisms of mast cell signaling in anaphylaxis. J Allergy Clin Immunol 124 (2009): 639-46; quiz 647-648.
- Peavy RD, Metcalfe DD. Understanding the mechanisms of anaphylaxis. Curr Opin Allergy Clin Immunol 8 (2008): 310-315.
- Khan BQ, Kemp SF. Pathophysiology of anaphylaxis. Curr Opin Allergy Clin Immunol 11 (2011): 319-325.
- Stevens WW, Kraft M, Eisenbarth SC. Recent insights into the mechanisms of anaphylaxis. Curr Opin Immunol 81 (2023): 102288.
- Leung A-y, Estrada-Reyes E, Goto K, et al. Global trend of food-induced anaphylaxis: Up to date. Pediatr Allergy Immunol 36 (2025): e70246.
- Montañez MI, Mayorga C, Bogas G, et al. Epidemiology, mechanisms, and diagnosis of drug-induced anaphylaxis. Front Immunol 8 (2017): 614.
- Sahiner UM, Durham SR. Hymenoptera venom allergy: how does venom immunotherapy prevent anaphylaxis from bee and wasp stings? Front Immunol 10 (2019): 1959.
- Muraro A, Lemanske RF Jr, Castells M, et al. Precision medicine in allergic disease—food allergy, drug allergy, and anaphylaxis. Pediatr Allergy Immunol 31 (2020): 48-51.
- Lee S, Bellolio MF, Hess EP, et al. Time of Onset and Predictors of Biphasic Anaphylactic Reactions: A Systematic Review and Meta-analysis. J Allergy Clin Immunol Pract. 2015 May-Jun;3(3):408-16.e1-2. Erratum in: J Allergy Clin Immunol Pract 12 (2024): 808.
- Dribin TE, Sampson HA, Camargo CA Jr, et al. Persistent, refractory, and biphasic anaphylaxis: A multidisciplinary Delphi study. J Allergy Clin Immunol 146 (2020): 1089-1096.
- Francuzik W, Dölle-Bierke S, Knop M, et al. Refractory Anaphylaxis: Data From the European Anaphylaxis Registry. Front Immunol 10 (2019): 2482.
- Calleja-Alarcón S, Sánchez-Hurtado LA, Romero-Gutiérrez L, et al. Ketamine as a treatment of bronchospasm due to an anaphylaxis: a case report. Rev Med Inst Mex Seguro Soc 60 (2022): 356-360.
- Goto Y, Hagiwara Y, Watase H, et al. Association of ketamine use with lower risks of post-intubation hypotension in hemodynamically-unstable patients in the emergency department. Sci Rep 9 (2019): 17230.
- Pouessel G, Dribin TE, Tacquard C, et al. Management of Refractory Anaphylaxis: An Overview of Current Guidelines. Clin Exp Allergy 54 (2024): 470-488.
- Short HB, Walters B, Fabi M, et al. Evaluating Practice Patterns of Observation Periods Following Epinephrine Administration for Anaphylaxis Among Pediatric Patients. Cureus 16 (2024): e69419.
- Yin Y, Shoshan MB, Shaker M, et al. Cost-effectiveness of watchful waiting versus immediate emergency department transfer after epinephrine autoinjector use in Canada. Allergy Asthma Clin Immunol 21 (2025): 5.
- Järvinen KM, Celestin J. Anaphylaxis avoidance and management: educating patients and their caregivers. J Asthma Allergy 7 (2014): 95-104.
- Sindher SB, Hillier C, Anderson B, et al. Treatment of food allergy: oral immunotherapy, biologics and beyond. Ann Allergy Asthma Immunol 131 (2023): 29-36.
- Sindher SB, Long A, Chin AR, et al. Food allergy, mechanisms, diagnosis and treatment: Innovation through a multi-targeted approach. Allergy 77 (2022): 2937-2948.


 Impact Factor:
* 8.1
Impact Factor:
* 8.1
 Acceptance Rate:
78.21%
Acceptance Rate:
78.21%
 Time to first decision: 10.4 days
Time to first decision: 10.4 days
 Time from article received to acceptance:
2-3 weeks
Time from article received to acceptance:
2-3 weeks
