Silent Signals in the Blood: Reframing Platelet Indices as Early Markers of Malaria Severity
Mohammed Mustafa Hassan1, Bashir Abdrhman Bashir*,2
1Department of Hematology, Faculty of Graduate College, Kassala University, Kassala, Sudan
2Department of Hematology, Faculty of Medical Laboratory Sciences, Port Sudan Ahlia University, Port Sudan, Sudan
* Corresponding author: Bashir Abdrhman Bashir, Department of Hematology, Faculty of Medical Laboratory Sciences, Port Sudan Ahlia University, Port Sudan, Sudan.
Received: 25 March 2026; Accepted: 30 March 2026; Published: 03 April 2026
Article Information
Citation: Mohammed Mustafa Hassan, Bashir Abdrhman Bashir. Silent Signals in the Blood: Reframing Platelet Indices as Early Markers of Malaria Severity. Archives of Microbiology and Immunology. 9 (2026): 36-41.
View / Download Pdf Share at FacebookAbstract
Background: Malaria is frequently associated with thrombocytopenia. The relationship between malaria and thrombocytopenia is thought to reflect platelet destruction or consumption during malaria infection. Platelets provide functions beyond blood coagulation, including combating infection and regulating inflammation.
Objective: To measure platelet indices and their correlation with parasite burden and clinical severity.
Methods: A comparative cross-sectional analysis was performed on 217 participants, including 167 malaria-positive and 50 malaria-negative participants. Platelet parameters, comprising platelet count (PLT), plateletcrit (PCT), platelet large cell count (PLCC), mean platelet volume (MPV), platelet distribution width (PDW), and platelet large cell ratio (PLCR), were examined. Nonparametric statistical approaches were used, and relationships with parasitemia were evaluated using Spearman’s rank correlation test.
Results: Malaria-positive individuals exhibited significantly reduced PLT (median 189 vs 302.5 ×109/L), PCT (0.156% vs 0.245%), PLCR (17.9% vs 22.2%), and PLCC (34.1 vs 66.8 ×109/L) (all p < 0.001) compared to controls. MPV was significantly lower in malaria cases (p < 0.001); however, it increased with rising parasitemia (ρ = 0.21, p = 0.008), suggesting a compensatory platelet response. PLT showed a strong inverse correlation with parasitemia (ρ = −0.53, p < 0.001). Additionally, Plasmodium vivax infection was associated with greater platelet depletion than P. falciparum infection.
Conclusion: Malaria is marked by pronounced thrombocytopenia and diminished platelet mass, with strong inverse correlations between platelet parameters and parasite load. Despite a general reduction in MPV in malaria cases, it is elevated with increased parasitemia, indicating a compensatory platelet response. Platelet indices, particularly PLT, PCT, and MPV, may serve as accessible biomarkers for assessing disease activity and severity.
Keywords
Malaria; Platelet indices; Thrombocytopenia; Mean platelet volume; Parasitemia; Sudan
Article Details
Introduction
Malaria is still a major cause of illness and death worldwide, especially in sub-Saharan Africa [1]. Despite advances in diagnosis and treatment, early assessment of illness severity remains a challenge, particularly in low-resource settings. Laboratory parameters provide valuable information for diagnosis and risk evaluation [1,2]. Hematological anomalies are among the most reliable observations in malaria. Thrombocytopenia is commonly observed in infections caused by Plasmodium falciparum and Plasmodium vivax and is often utilized as a supportive diagnostic indicator [1]. Nonetheless, the role of platelets in malaria extends beyond mere quantitative diminution. Emerging data indicate that platelets contribute to host defense by binding directly to parasitized erythrocytes to help limit parasite replication and by modulating immune responses through the release of signaling molecules involved in inflammation control [3,4]. Routine use of automated hematology analyzers provides access to several platelet indices, including mean platelet volume (MPV), plateletcrit (PCT), and platelet distribution width (PDW). These metrics offer information on platelet synthesis, activation, and turnover [5]. Despite their availability, their clinical relevance in malaria remains inadequately investigated, and they are infrequently included in standard clinical assessments [1,6]. Numerous studies have predominantly concentrated on platelet count, frequently emphasizing thrombocytopenia as a defining characteristic of malaria [7,8]. This method, however, neglects the dynamic characteristics of platelet physiology. Variations in platelet size and distribution may indicate underlying mechanisms such as peripheral destruction, splenic sequestration, and compensatory bone marrow activity. These modifications may provide supplementary insights into illness progression and severity. Hematological responses differ based on parasite type, parasite load, and clinical profile. Understanding these differences is crucial to improving the use of standard test values in clinical practice. This study assesses platelet indices in malaria and their correlation with parasitemia, clinical severity, and demographics. It aims to show the value of platelet indices as functional indicators in malaria, beyond simple platelet counts.
Materials and Methods
Study Design and Setting
This was a cross-sectional analytical study conducted at Kassala Teaching Hospital, Kassala State, Sudan. The research was conducted over 18 months, from June 2023 to December 2024. The laboratory functions as a referral facility for standard hematological assessments, including complete blood count (CBC) analysis for patients suspected of infectious and hematological conditions.
Study Population
The study had 217 volunteers, divided into Malaria-positive cases (n = 167) and Malaria-negative controls (n = 50). Malaria diagnosis was confirmed via standard parasitological techniques, including microscopy and/or laboratory verification in accordance with institutional protocols. The control group included 50 individuals who tested negative for malaria during the study period and were selected from the same laboratory environment as the patients. Controls were delineated as those who: Did not show parasitological evidence of malaria infection upon microscopic examination of blood films. Were submitted for standard laboratory tests or presented with non-infectious health conditions. Had no clinical suspicion of malaria at the time of testing. Had no recent history of receiving antimalarial medication. To ensure comparability, controls were selected from the same population and time frame as malaria patients.
Inclusion and Exclusion Criteria
Participants were eligible if they satisfied the following criteria: Confirmed malaria infection (Plasmodium falciparum or Plasmodium vivax). Provision of comprehensive hematological data, encompassing platelet indicators. All demographics and both genders. Patients who presented throughout the study period. Regarding the control group: Individuals devoid of any indication of malaria infection. Typical or non-malaria-associated clinical manifestation. Participants were disqualified if they possessed: Recognized hematological illnesses (e.g., thrombocytopenia from various etiologies, leukemia). Chronic systemic conditions influencing hematological parameters (e.g., hepatic illness, renal insufficiency), recent blood transfusion, and Insufficient laboratory or clinical data.
Data Collection
Data were gathered using a systematic data extraction form and encompassed the Following Demographic variables: Age, Gender (Male/Female), and Location (Urban/Rural). Clinical parameters: Type of malaria (P. falciparum, P. vivax), Clinical presentation (Uncomplicated/Severe). Laboratory parameters: Parameters of complete blood count (CBC), primarily platelet indices and parasitemia levels.
Platelet Indices Assessment
Venous blood specimens (3 ml) were drawn under aseptic conditions into EDTA-anticoagulated tubes. They were then examined using a semi-automated hematology analyzer (Mindray BC30s, China).
The subsequent platelet indices were assessed:
Platelet Count (PLT) – denoted as ×109/L
Mean Platelet Volume (MPV) – measured in femtoliters (fL)
Plateletcrit (PCT) - the proportion of total blood volume comprised of platelets (%)
Platelet Large Cell Ratio (PLCR) - the ratio of big platelets (%)
Platelet Large Cell Count (PLCC) - the absolute quantification of large platelets (×109/L)
These parameters served as measures of platelet formation, activation, and turnover.
Assessment of Parasitemia
Parasitemia grading was performed according to the WHO-recommended microscopy standards [9] as follows:
1 (+) = low parasitemia
2 (++) = mild parasitemia
3 (+++) = moderate parasitemia
4 (++++) = high parasitemia
Parasite density was employed to evaluate the correlation between platelet indicators and illness severity.
Statistical Analysis
Data were entered and analyzed using appropriate statistical software (SPSS, version 25; IBM, Chicago, USA). Before analysis, the distributions of continuous variables were evaluated using the Shapiro–Wilk test, which indicated that most variables were not normally distributed.
Continuous variables were presented as median and IQR, or as mean ± SD when appropriate.
Comparisons between groups used the Mann–Whitney U test for continuous variables and the Chi-square test for categorical variables, which were summarized as frequencies and percentages.
Spearman’s rank correlation coefficient (ρ) was used to assess the relationship between platelet indices and parasitemia. All statistical tests were two-tailed, with p < 0.05 considered significant.
Ethical Approval
Ethical approval was obtained from the relevant Ministry of Health, Health Research Ethics Committee, Kassala State (19/2/2022- 18). Data confidentiality was rigorously respected. Patient identifiers were excluded. Written informed permission was acquired from each participant.
Results
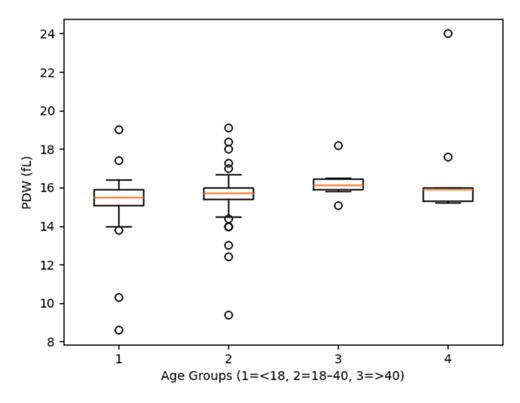
This study recruited 217 participants: 167 malaria-positive patients and 50 malaria-negative controls. The fundamental characteristics of the study population are encapsulated in Table 1. Among malaria-positive patients, Plasmodium falciparum accounted for 158/167 (94.6%), whereas Plasmodium vivax accounted for 9/167 (5.4%). Among P. falciparum cases, 81/158 (51.3%) were male, and 77/158 (48.7%) were female. Among P. vivax cases, 4/9 (44.4%) were male, and 5/9 (55.6%) were female. In the control group, 17/50 (34.0%) were male, and 33/50 (66.0%) were female. Overall, the mean age was 28.0 ± 14.2 years in malaria-positive patients and 25.1 ± 10.5 years in controls, with no significant difference (p = 0.446). More males were among the malaria cases, but this difference was not statistically significant (p = 0.053). In addition, platelet indices were compared across age groups. PLT did not differ significantly across age groups (Kruskal–Wallis, p = 0.362). However, PDW showed a statistically significant difference (Kruskal–Wallis, p = 0.014) (Figure 1), whereas PCT, MPV, PLCR, and PLCC did not demonstrate significant variation (PCT: p = 0.420; MPV: p = 0.209; PLCR: p = 0.650; PLCC: p = 0.621). Analysis of residence showed 130 malaria cases (77.8%) were from urban regions, compared to 46 controls (92.0%). In rural areas, 37 malaria cases (22.2%) and 4 controls (8.0%) were identified. The difference was statistically significant (p = 0.042), indicating a higher malaria burden in rural areas.
Building on these findings, Table 2 compares platelet indices between malaria-positive individuals and controls. Malaria patients had a lower PLT: median 189 ×109/L [IQR: 117.5–236.0] versus 302.5 ×109/L [IQR: 252.0–369.8] in controls (p < 0.001). PCT was also lower in malaria cases: 0.156% [IQR: 0.102–0.200] versus 0.245% [IQR: 0.197–0.271] in controls (p < 0.001). Malaria cases had a lower PLCR: median 17.9% [IQR: 16.3–22.3] versus 22.2% [IQR: 18.9–25.5] in controls (p < 0.001). PLCC showed a similar result, with malaria patients at 34.1 ×109/L [IQR: 20.5–49.2] and controls at 66.8 ×109/L [IQR: 48.0–94.3] (p < 0.001). The MPV differed significantly between the groups. Malaria cases had lower median values of 8.9 fL [IQR: 8.3–9.7], while controls had 9.6 fL [IQR: 9.0–10.3] (p < 0.001). This indicates changes in platelet size dynamics. In contrast, PDW showed no significant difference. Malaria cases had a median PDW of 15.7 fL [IQR: 15.3–16.0], and controls had a median PDW of 15.5 fL [IQR: 15.3–15.7] (p = 0.087). Malaria infection was associated with a persistent reduction in platelet count, reduced platelet mass, and altered platelet size characteristics. The distinctions are encapsulated in Table 2.
Species-specific analysis showed that patients with P. vivax had lower platelet indices than those with P. falciparum. The median PLT was 86 ×109/L [IQR: 52–165] in P. vivax compared with 195 ×109/L [IQR: 128–242.5] in P. falciparum (p = 0.012). PCT was also lower in P. vivax, 0.08% [IQR: 0.05–0.13] versus 0.16% [IQR: 0.11–0.20] in P. falciparum (p = 0.012). Likewise, PLCC was significantly lower in P. vivax, 15.1 ×109/L [IQR: 12.0–35.2] compared with 37.0 ×109/L [IQR: 23.8–50.8] in P. falciparum (p = 0.008). In contrast, MPV, PDW, and PLCR did not differ significantly between the two species (MPV: 8.5 fL [IQR: 7.9–9.1] vs 8.3 fL [IQR: 7.8–9.0], p = 0.542; PDW: 15.8 fL [IQR: 15.3–16.2] vs 15.65 fL [IQR: 15.3–16.0], p = 0.774; PLCR: 16.9% [IQR: 16.6–22.9] vs 18.0% [IQR: 16.3–22.2], p = 0.826). Interestingly, this research revealed a substantial inverse correlation between platelet indices, including PLT (ρ = −0.53, p < 0.001), PCT (ρ = −0.51, p < 0.001), and PLCC (p < 0.001) with parasitemia in malaria-positive patients, indicating that increased parasite load contributes to lower platelet levels. Conversely, MPV showed a modest but clear positive link with parasite levels (ρ = 0.21, p = 0.008), indicating that platelet size increased as parasitemia increased. The percentage of PLCR and the range of PDW showed no clear statistical links with parasite levels (PLCR: p = 0.068; PDW: p = 0.100).
Table 1: Baseline Characteristics of Study Participants
|
Variable |
Malaria Positive (n=167) |
Control (n=50) |
P-value |
|
Age (years, mean ± SD) |
28.0 ± 14.2 |
25.1 ± 10.5 |
0.446 |
|
Sex |
0.053 |
||
|
Male, n (%) |
85 (50.9%) |
17 (34.0%) |
— |
|
Female, n (%) |
82 (49.1%) |
33 (66.0%) |
|
|
Residence |
0.042 |
||
|
Urban, n (%) |
130 (77.8%) |
46 (92.0%) |
— |
|
Rural, n (%) |
37 (22.2%) |
4 (8.0%) |
|
Table 2: Comparison of Platelet Indices Between Malaria Cases and Controls
|
Platelet Index |
Malaria Cases |
Controls |
P-value |
|
Median (IQR) |
Median (IQR) |
||
|
PLT (×109/L) |
189 (117.5–236.0) |
302.5 (252.0–369.8) |
<0.001 |
|
PCT (%) |
0.156 (0.102–0.200) |
0.245 (0.197–0.271) |
<0.001 |
|
PLCR (%) |
17.9 (16.3–22.3) |
22.2 (18.9–25.5) |
<0.001 |
|
PDW (fL) |
15.7 (15.3–16.0) |
15.5 (15.3–15.7) |
0.087 |
|
MPV (fL) |
8.9 (8.3–9.7) |
9.6 (9.0–10.3) |
<0.001 |
|
PLCC (×109/L) |
34.1 (20.5–49.2) |
66.8 (48.0–94.3) |
<0.001 |
PLT, platelet count; PCT, plateletcrit; PLCR, platelet large cell ratio; PDW, platelet distribution width; MPV, mean platelet volume; PLCC, platelet large cell count
Table 3: Differential Platelet Indices According to Malaria Species (P. falciparum vs P. vivax)
|
Platelet Index |
PF Median (IQR) |
PV Median (IQR) |
P-value |
Interpretation |
|
PLT (×109/L) |
195 (128–242.5) |
86 (52–165) |
0.012 |
Significantly lower in PV |
|
PCT (%) |
0.16 (0.11–0.20) |
0.08 (0.05–0.13) |
0.012 |
Significantly lower in PV |
|
PLCR (%) |
18.0 (16.3–22.2) |
16.9 (16.6–22.9) |
0.826 |
Not significant |
|
PDW (fL) |
15.65 (15.3–16.0) |
15.8 (15.3–16.2) |
0.774 |
Not significant |
|
MPV (fL) |
8.3 (7.8–9.0) |
8.5 (7.9–9.1) |
0.542 |
Not significant |
|
PLCC (×109/L) |
37.0 (23.8–50.8) |
15.1 (12.0–35.2) |
0.008 |
Significantly lower in PV |
PLT, platelet count; PCT, plateletcrit; PLCR, platelet large cell ratio; PDW, platelet distribution width; MPV, mean platelet volume; PLCC, platelet large cell count; PF, plasmodium falciparum; PV, plasmodium vivax; IQR, interquartile range
Figure 1: Platelet distribution width (PDW) Across Age Groups in Malaria Patients. Boxplot illustrating PDW across age groups (<18, 18–40, >40 years). Data are presented as median and interquartile range (IQR), with outliers shown. Differences were assessed using the Kruskal–Wallis test (p = 0.014).
Discussion
This study reveals that malaria infection is linked to substantial alterations in platelet indices, indicating both quantitative and qualitative changes in platelet dynamics. The results indicate a persistent trend of reduced platelet count and platelet mass, along with alterations in platelet morphology. Only specific platelet indices correlated with parasite burden, whereas others showed no significant association. This study found significantly lower platelet counts in patients with malaria than in controls. Median values were 189 ×109/L for patients and 302.5 ×109/L for controls (p < 0.001). This result aligns with previous studies that have identified thrombocytopenia as a common malaria-related blood abnormality [7,8]. Multiple mechanisms likely cause this reduced count, including peripheral destruction, splenic sequestration, immune-mediated lysis, and direct interaction between platelets and infected red blood cells [3,4].
Along with thrombocytopenia, PCT was markedly decreased in patients with malaria, indicating a reduction in the total circulating platelet mass. This study corroborates the notion that platelet changes in malaria extend beyond mere platelet count to encompass a more comprehensive decline in platelet biomass. Previous investigations have yielded similar findings, indicating that PCT may serve as a more comprehensive measure of platelet depletion than platelet count alone [5]. The PLCR was significantly reduced in malaria cases, suggesting an altered platelet size distribution. However, as the association with parasitemia did not reach statistical significance (p = 0.068), PLCR may reflect platelet changes in malaria, but does not reliably indicate parasite burden. PDW similarly showed no significant correlation with parasitemia (p = 0.100), suggesting that platelet size variability is unaffected by disease severity. Therefore, only certain platelet parameters—such as PCT—appear to be informative indicators of malaria severity.
This investigation shows a strong inverse association between parasitemia and PLT (ρ = −0.53, p < 0.001), and between parasitemia and PCT (ρ = −0.51, p < 0.001). As parasite load rises, PLT drops, suggesting that higher parasitemia may be associated with lower platelet levels. Similar negative correlations have been reported, supporting the hypothesis that elevated parasitemia can result in increased platelet consumption and destruction [6,10]. Species-specific analysis showed clear differences in platelet indices between Plasmodium falciparum and Plasmodium vivax. Patients with P. vivax infection had significantly lower PLT, PCT, and PLCC than those with P. falciparum (p < 0.05), indicating more pronounced platelet depletion in P. vivax infection. In contrast, MPV, PDW, and PLCR did not differ significantly between the species (MPV: p = 0.542; PDW: p = 0.774; PLCR: p = 0.826), suggesting parasite species do not substantially affect platelet size or distribution. These findings align with studies showing that P. vivax malaria often causes significant thrombocytopenia, sometimes matching or surpassing that seen in P. falciparum infections [11,12]. While P. falciparum is usually considered the more severe form of malaria, recent evidence shows that P. vivax also causes marked hematological disturbances, notably platelet depletion [12,13]. Immune-mediated platelet destruction increased splenic sequestration, and enhanced peripheral consumption—each varying by species—may underlie this effect.
Conversely, MPV showed a small but significant positive association with parasitemia (ρ = 0.21, p = 0.008). Notably, MPV was reduced overall in malaria cases compared to controls but increased with rising parasitemia, suggesting a compensatory response. Reduced platelet counts may trigger the bone marrow to release larger, younger platelets as a compensatory response. This phenomenon, known as reactive thrombopoiesis, occurs in inflammatory and infectious diseases such as malaria [14]. The observed increase in MPV with rising parasitemia supports the idea that increased peripheral platelet consumption drives greater platelet turnover.
The trend seen here—lower platelet count and mass with larger platelet size—shows a balance between platelet loss and production. This supports the idea that malaria-induced thrombocytopenia involves not just bone marrow suppression, but also active peripheral processes. The absence of a substantial correlation between PDW and Parasitemia indicates that platelet size variability is not significantly affected by parasite load, whereas the marginal relevance of PLCR may suggest a limited role in disease assessment. These findings underscore the need to assess platelet indicators in conjunction rather than independently.
This study's findings indicate that platelet indices, including PLT, PCT, and MPV, may be valuable supplementary markers for evaluating malaria severity clinically. Since these characteristics are consistently obtainable from automated hematology analyzers, they provide a cost-efficient and accessible resource for enhancing clinical evaluation, particularly in resource-constrained environments. Based on current results, malaria activity appears to cause specific changes in platelet measurements rather than affecting them uniformly. In this study, PLT and PCT decreased as malaria infection increased, while MPV increased slightly. These findings suggest that the pattern of increased consumption is accompanied by compensatory platelet production as parasite burden rises.
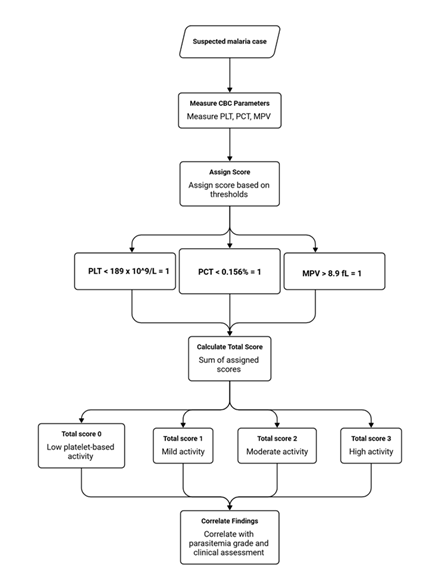
Building on these observations, a simple system for measuring platelet activity can be proposed. Using average values observed in malaria cases, one point is given for each of the following: PLT < 189, PCT < 0.156%, and MPV > 8.9. The total score goes from 0 to 3, making it easier to group patients by their platelet activity. This model system might help healthcare workers classify malaria cases and decide how to monitor or treat patients (Figure 2). Future research should test this model across more diverse patient groups and assess its usefulness in real-world clinical settings. A score of 0 may indicate diminished platelet activity, whereas 1, 2, and 3 may indicate mild, moderate, and high activity, respectively. This pattern illustrates the cumulative impact of thrombocytopenia, diminished platelet mass, and the compensatory secretion of bigger platelets from the bone marrow. The model excluded the platelet large cell ratio and platelet distribution width because they showed no significant association with parasitemia. This proposed model is intended as an exploratory tool. While simple and reliant on commonly accessible laboratory parameters, its purpose is to robustly supplement—rather than replace—recognized clinical severity criteria. Additional prospective studies are essential to confirm its clinical utility and further clarify its predictive value across diverse malaria settings.
PDW showed significant variation between age groups, though other platelet indices did not. This suggests that platelet size heterogeneity may reflect age-related physiological differences rather than direct effects of malaria. However, without accompanying changes in PLT or PCT, the clinical significance of this finding is uncertain. Further studies are required to clarify whether age-related changes in PDW affect disease progression or reflect age-based differences in platelet turnover. This research possesses certain limitations. The cross-sectional design restricts the capacity to determine causal links. Furthermore, parasitemia was evaluated using a semi-quantitative grading method rather than exact parasite counts per microliter, thereby diminishing the sensitivity of the correlation analysis. The limited number of Plasmodium vivax cases (n = 9) may constrain the statistical power of species-specific comparisons, necessitating cautious interpretation of these findings. Future research employing quantitative parasitemia assessments and longitudinal monitoring is advised.
Conclusion
This study's findings indicate that malaria is associated with substantial changes in platelet indices, suggesting both platelet depletion and compensatory production. PLT and PCT exhibit significant inverse correlations with parasitemia; while MPV, although reduced overall, increases with parasite burden, indicating reactive thrombopoiesis. Notably, Plasmodium vivax infection was associated with greater platelet depletion than P. falciparum infection. These findings highlight the potential of platelet indices as simple, cost-effective biomarkers for assessing malaria severity and disease activity.
Figure 2: A simplified flowchart illustrating the proposed platelet-based activity model derived from the present study. Platelet count (PLT), plateletcrit (PCT), and mean platelet volume (MPV) are obtained from routine complete blood count analysis. One point is assigned for each of the following: PLT < 189 ×109/L, PCT < 0.156%, and MPV > 8.9 fL. The total score (0–3) provides an estimate of platelet-based disease activity, ranging from low to high. This model is exploratory and requires further validation.
References
- World Health Organization. World malaria report 2023. Geneva: WHO (2023).
- White NJ, Turner GDH, Medana IM, et al. The pathology of malaria. In: Manson’s tropical diseases. 23rd ed. Elsevier (2014): 532–600.
- McMorran BJ, Marshall VM, de Graaf C, et al. Platelets kill intraerythrocytic malarial parasites and mediate survival to infection. Science 323 (2009): 797-800.
- Kho S, Barber BE, Johar E, et al. Platelets kill circulating parasites of all major Plasmodiumspecies in human malaria. Blood 132 (2018): 1332-1344.
- Noris P, Melazzini F, Balduini CL. New roles for mean platelet volume measurement in the clinical practice? Platelets 27 (2016): 607-612.
- Maina RN, Walsh D, Gaddy C, et al. Impact of Plasmodium falciparum infection on haematological parameters in children living in Western Kenya. Malar J 9 (2010): S4.
- Lacerda MV, Mourão MP, Coelho HC, et al. Thrombocytopenia in malaria: who cares? Mem Inst Oswaldo Cruz 106 (2011): 52-63.
- Lampah DA, Yeo TW, Malloy M, et al. Severe malarial thrombocytopenia: a risk factor for mortality in Papua, Indonesia. J Infect Dis 211 (2015): 623-34.
- World Health Organization. Basic malaria microscopy – Part I: Learner’s guide. 2nd ed. Geneva: WHO (2010).
- Antwi-Baffour S, Mensah BT, Johnson G, et al. Haematological parameters and their correlation with the degree of malaria parasitaemia among outpatients attending a polyclinic. Malar J 22 (2023): 281.
- Saravu K, Docherla M, Vasudev A, et al. Thrombocytopenia in vivax and falciparum malaria: an observational study of 131 patients in Karnataka, India. Ann Trop Med Parasitol 105 (2011): 593-8.
- Phyo AP, Dahal P, Mayxay M, et al. Clinical impact of vivax malaria: A collection review. PLoS Med 19 (2022): e1003890.
- Rahimi BA, Thakkinstian A, White NJ, et al. Severe vivax malaria: a systematic review and meta-analysis of clinical studies since 1900. Malar J 13 (2014): 481.
- Kotepui M, Phunphuech B, Phiwklam N, et al. Effect of malarial infection on haematological parameters in population near Thailand-Myanmar border. Malar J 13 (2014): 218.


 Impact Factor:
* 3.5
Impact Factor:
* 3.5
 Acceptance Rate:
71.36%
Acceptance Rate:
71.36%
 Time to first decision: 10.4 days
Time to first decision: 10.4 days
 Time from article received to acceptance:
2-3 weeks
Time from article received to acceptance:
2-3 weeks

