Targeting Oncogenic Signaling and Anabolic Metabolism in Cancer Therapy
Mohd Javed Akhtar*,1, Maqusood Ahamed2, Sudhir Panwar3
1Associate Professor, King Saud University, Riyadh, Saudi Arabia
2King Abdullah Institute for Nanotechnology, King Saud University, Riyadh-11451, Saudi Arabia
3Department of Zoology, University of Lucknow, Lucknow-226001, UP, India
*Corresponding author: Mohd Javed Akhtar. Associate Professor, Department of Zoology, King Saud University, Riyadh-11451, Saudi Arabia.
Received: 17 March 2026; Accepted: 24 March 2026; Published: 05 April 2026
Article Information
Citation: Mohd Javed Akhtar, Maqusood Ahamed, Sudhir Panwar. Targeting oncogenic signaling and anabolic metabolism in cancer therapy. Journal of Pharmacy and Pharmacology Research. 10 (2026): 49-59.
View / Download Pdf Share at FacebookAbstract
Normal cell proliferation, which is essential for tissue and organ repair, is tightly regulated. Cancer is a deregulated metabolic disease. This deregulation allows cancer cells to become independent of the control mechanisms that regulate cell division and the cell cycle. Cells with few oncologic properties are gradually selected for this independence, which can become aggressive as more growth-promoting factors are added, leading to malignant transformation after a long benign phase. Mutations or epigenetic deregulation cause reduced control over cell proliferation, often ignoring mitogenic signals, and result in metabolic reprogramming that boosts nutrient uptake, anabolic reductive reactions, and redox balance. As a result, cancer therapy mainly targets blocking growth-promoting pathways used in normal cell growth, but this often causes systemic toxicity due to disruption of organismal homeostasis. Despite major advances in understanding cancer biology and how anticancer drugs function, targeting growth pathways such as nucleotide, protein, and lipid synthesis while minimizing side effects remains challenging. This review provides an update on cancer biology and the targetable pathways in cancer therapy, with selected examples of inhibitor molecules.
Keywords
Anti-mitogenic molecules; cell growth inhibitors; anabolic targets; redox homeostasis; cancer therapy
Article Details
Introduction
Cancer is an uncontrolled cell division that results in malignancy, impairing the quality of life for affected individuals and society, making cancer a global challenge. Cancer mortality continues to decline through 2021, preventing over 4 million deaths since 1991 due to reductions in smoking, earlier detection for some cancers, and improved treatment options in both benign and metastatic settings [1]. However, the progress made is threatened by rising rates of six of the top ten cancers. Between 2015 and 2019, incidence rates grew annually by 0.6%-1% for breast, pancreas, and uterine corpus cancers. Meanwhile, prostate, female liver, kidney, HPV-related oral cancers, and melanoma saw increases of 2%-3% each year [1]. Additionally, in young adults, cervical cancer (ages 30-44) and colorectal cancer (under 55) incidence rates increased by 1%-2% annually[1]. Cancer is characterized by deregulated intermediary and anabolic metabolism, involving significant changes in energy production and biosynthesis. Tumor cells exhibit the Warburg effect, preferring glycolysis over oxidative phosphorylation (OXPHOS) despite the presence of oxygen, leading to increased glucose intake and lactate production [2-5]. This metabolic shift supports rapid cell growth by funneling glycolytic intermediates into the hexosamine pathway, the pentose phosphate pathway, and one-carbon metabolism, all essential for producing nucleotides, amino acids, and lipids needed for new cellular components. Additionally, cancer cells show enhanced glutaminolysis to maintain redox balance through NADPH generation. Abnormalities in signaling pathways like PI3K/AKT/mTOR drive these anabolic processes, while oncogenes such as MYC reprogram metabolism by increasing overall processes, from nutrient uptake to enhanced glycolysis, OXPHOS, and macromolecular synthesis [6]. Figure 1 provides an overview of how cell growth is promoted following mitogenic stimulation. As a result, this metabolic deregulation creates targets within specific metabolic pathways vital for tumor growth and survival, offering potential opportunities to interfere with tumor bioenergetics without significantly damaging normal tissues. This article reviews the major growth-promoting pathways, their interconnections, and identifies key vulnerable targets in oncogenic and anabolic metabolism for cancer therapy.
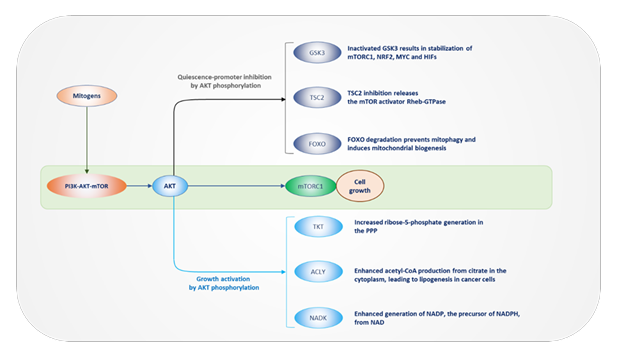
Figure 1: An overview of how cell growth is promoted following mitogenic stimulation. Tumor suppressors such as FOXO, TSC2, and GSK3 are often inactivated in cancer. Growth-promoting kinases and enzymes, including PI3K, AKT, ACLY, and NADPH production, are frequently increased in cancer. The figure offers a general view of cell growth-promoting pathways, as discussed in the text, and highlights potential targets for cancer therapy. This article mainly focuses on growth stimulation and possible targets within the cancer-related PI3K-AKT-mTOR pathway, shown in the light green area. This pathway is explained in more detail in the next figure.
Overview of growth-promoting signaling and cell mass duplication during cell proliferation
Normal cell proliferation, which is crucial for tissue and organ repair, is closely controlled. During cancer development, cells undergo a transformation that gives them independence from external regulation, and over time, these cells adopt autonomous growth modes by ignoring mitogens or signaling to stop cell division. NADPH oxidases (NOXs) are vital activators of cell growth by stimulating the PI3K-AKT-mTOR pathway [7-9]. This pathway is a highly conserved growth-promoting route from yeast to mammals, and when activated by mitogens, it increases anabolic processes like the synthesis of nucleotides, proteins, and lipids. ROS produced by NOXs or mitochondria inhibit lipid phosphatase and tensin homologue (PTEN). NOX defects lead to regenerative issues in the brain and impair sperm production in mice [7-9]. NOXs also play a crucial role in macrophage function, where ROS produced by NOX in specialized cellular compartments, the phagosomes, are used to eliminate ingested pathogens [10,11]. When PTEN is inhibited, it cannot dephosphorylate phosphatidylinositol 3-4,5-bisphosphate (PIP3 or PtdIns (3,4,5)P3) to phosphatidylinositol 4,5-bisphosphate (PIP2 or PtdIns (4,5)P2) sites in cell membranes. PIP3, generated from PIP2 by mitogen-activated phosphoinositide 3 kinase (PI3K), serves as the docking and activation site for AKT (also known as PKB) activators phosphoinositide-dependent kinase (PDPK1, also known as PDK1) and mechanistic target of rapamycin kinase in complex 2 (mTORC2) [6]. Unsurprisingly, the PI3K-AKT axis is the most frequently upregulated growth-promoting pathway in cells [6]. Genetic or epigenetic changes have been the main drivers of PI3K-AKT deregulation. Mutations in the tumor suppressor PTEN, which lead to constitutive activation of AKT signaling, rank as the second most deregulated oncogenic mechanism in cancers [6]. AKT stabilizes MYC, HIF, and NRF2 by inhibiting their degrader GSK3 [6]. MYC activates almost all glycolytic enzymes, promotes OXPHOS by stimulating mitochondrial biogenesis, glutamine metabolism, and nucleotide synthesis through upregulating key enzymes and nutrient transporters [12,13]. MYC and NRF2 upregulate the oxidative PPP enzyme G6PD and the non-oxidative PPP enzyme transketolase (TKT) to produce ribose-5-phosphate, the precursor for nucleotide sugars [14].
NRF2 promotes de novo GSH synthesis [15,16]. Additionally, NRF2 enhances the production of GSH-dependent antioxidant enzymes such as glutathione reductase (GR), glutathione peroxidase (GPX), peroxiredoxins 1 and 3 (PRDX1, PRDX3), thioredoxin reductase 1 (Txnrd 1), and xenobiotic-metabolizing glutathione S-transferases (GSTs) [15,16]. Increased NADPH and GSH are hallmarks of many cancers and resist cancer therapy [17]. HIF enhances glucose uptake and promotes cell growth by increasing aerobic glycolysis and decreasing OXPHOS in hypoxia, particularly in the inner cells of solid tumors, where oxygen delivery is very limited due to increased distance from a nearby capillary. HIF targeting has gained increasing attention in recent times as it promotes cancer stemness and, therefore, cancer recurrence after treatment [18,19]. A meta-analysis of 40 studies in 2025 found that 39.6% of breast cancer patients exhibited HIF1α overexpression [20]. AKT signaling is temporarily necessary for normal growth and is unnecessary when growth is not needed. However, if growth signaling is activated without a need for growth, it can lead to a homeostatic disaster, that is, cancer. As a result, targeting growth signaling pathways has been one of the main focuses of oncologists in treating or controlling cancer. Metabolic reprogramming is a characteristic feature of malignant tumors. Nucleotide synthesis takes place only in proliferating cells, which can have, on average, more than 10 times higher NTP levels compared to resting, non-dividing cells [21]. The AKT-MYC axis increases the expression of phosphoribosyl pyrophosphate synthetase 2 (PRPS2), which produces phosphoribosyl pyrophosphate used for de novo purine synthesis [13]. Notably, PRPS2 is highly active in MYC-driven tumors and promotes colorectal cancer metastasis [13]. Inosine monophosphate (IMP) dehydrogenase (IMPDH, IMPDH1 and IMPDH2), which produces IMP, the precursor for purines (adenine and guanine), CAD2 (carbamoyl-phosphate synthetase 2, aspartate transcarbamylase, and dihydroorotase) that produces dihydroorotate and DHODH, which produces uridine monophosphate (UMP), the precursor for pyrimidines (cytosine and thymine), is upregulated by MYC [13]. Furthermore, AKT phosphorylation of ribose-5-phosphate-generating TKT and NADPH-generating NAD kinase (NADK) enhances their activity and ensures aligned production of growth precursors, that is, NADPH for reduction reactions and ribose-5-phosphate for forming sugar phosphate backbone in nucleic acids (RNA and DNA) [6]. Findings demonstrate that PRPS1/2-mediated purine biosynthesis is essential for maintaining the stemness of highly proliferative pluripotent stem cells (PSCs) in culture [22]. AKT-stabilized SREBP that transcriptionally upregulates enzymes, including the enzymes of the PPP (G6PD and TKT), and cytosolic and mitochondrial isocitrate dehydrogenases (IDHs) and malic enzymes (MEs) that lead to increases in cellular NADPH that support reductive and raise antioxidants in cells. Compared to most non-transformed cells that rely on glucose, MYC-driven cancer cells depend primarily on glutamine metabolism. MYC reduces the amount of glucose carbon entering the TCA cycle and diminishes glucose contribution in mitochondrial-dependent phospholipid synthesis [23]. Figure 2 illustrates the growth-activating potential of the PI3K-AKT-mTOR axis, particularly in increasing cell mass through the regulated synthesis of nucleotides, proteins, and lipids. It also highlights the inhibitors of this pathway as potential cancer therapies, which are the focus of this review, along with a brief discussion of the advantages and disadvantages of targeting them.
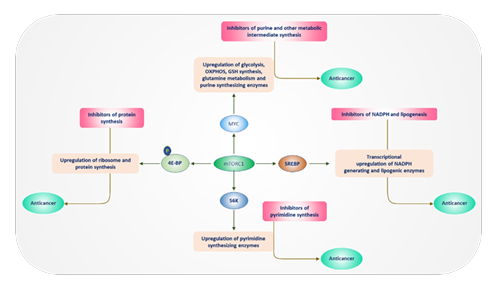
Figure 2: The PI3K-AKT-mTOR pathway promotes anabolic processes such as nucleotide, protein, and lipid synthesis. 4E-binding proteins (4E-BPs) bind to and inhibit the initiation factor 4E (IF-4E), which is necessary for protein synthesis. mTORC1 phosphorylation inhibits 4E-BPs, releasing IF-4E and triggering the start of protein synthesis during cell growth. mTORC1 also activates the sterol regulatory element-binding protein (SREBP), a transcription factor that increases the expression of enzymes involved in the synthesis of ribose-5-phosphate, NADPH, and lipogenic enzymes. NADPH is essential in supporting reductive and antioxidative reactions. AKT-stabilized MYC is a key activator of glycolytic enzymes, mitochondrial biogenesis, glutamine metabolism, and nucleotide production. mTORC1 phosphorylation activates p70S6 kinase 1 (S6K1, or S6K, as written here), which promotes nucleotide and protein synthesis. This article explores anticancer molecules that inhibit the PI3K-AKT-mTOR pathway and also block subsequent nucleotide, protein, and lipid synthesis to control cancer growth.
Inhibiting mitogenic signaling and growth promoters in circumventing cancer
Mechanisms that promote cell growth are therefore obvious targets in cancer therapy. Cell metabolism is a broad topic, so here we focus on well-studied biological processes and targetable components in anticancer strategies, beginning from the growth-promoting signals and anabolic metabolism essential in duplicating cell mass.
NOX inhibition in cancer therapy via modulating the PTEN-PI3K axis
A deranged ROS signaling often results in tumorigenesis [24,25]. Due to their growth-promoting effects via the opposing PTEN and PI3K signaling, NOX enzymes, particularly NOX1, NOX2, and NOX4, are often overexpressed in various cancers, including colon, lung, prostate, and breast cancer, where they promote tumor growth, blood vessel formation, cancer spread, and resistance to therapy [26]. ROS generated by NOXs at inappropriate times is implicated in PTEN inhibition, promoting many cancers of the thyroid, colon, lung, breast, and blood [27]. Dual inhibition of NOX1/4 impairs the pro-tumorigenic effects of transforming growth factor-β (TGF-β) in cholangiocarcinoma-associated fibroblasts [28]. Silencing NOX4 expression by 80%-90% with siRNA extended the doubling time of tumor cells in human neuroblastoma cells SH-SY5Y, indicating that NOX inhibition a viable cancer treatment approach [29]. NOX4 inhibition suppressed glycolysis-related proteins (HIF-1α, LDHA, and PDK1), reducing glucose uptake, lactate production, and ROS generation, while increasing mitochondrial membrane potential [29]. NOX inhibition in cancer treatment is the subject of intense research [26]. The World Health Organization recently introduced the term “xib” to define a new therapeutic category for NOXs inhibitor, further highlighting their potential therapeutic applications [26]. Notably, Setanaxibs (GKT136901 and GKT137831) emerged as specific dual inhibitors of NOX1 and NOX4, with GKT137831 being the only NOX4-specific inhibitor tested in clinical trials. The lack of selective inhibitors for NOX subtypes, however, has restricted their use in clinical trials [26]. NOX-5-specific inhibitor has been reported recently, which remains untested in patients [30]. Because of the essential roles of NOXs in innate immunity, NOX inhibitors used in cancer therapy can significantly weaken the immunity and well-being of cancer patients [31]. Further research is necessary to develop NOX-specific inhibitors and investigate their role in reducing AKT signaling and cancer.
PI3K-mTOR pathway inhibitors in cancer
The landscape of clinical trials is increasingly focused on inhibitors that target PI3K isoforms functioning upstream of mTOR. These inhibitors target all four class I PI3K isoforms (α, β, δ, γ). Additionally, isoform-specific inhibitors are being developed to target particular PI3K isoforms [32,33]. Besides specific inhibitors, several dual inhibitors that target both mTOR and PI3K components have been developed and are in various phases of clinical trials. Dual mTOR/PI3K inhibitors, including dactolisib (NVP-BEZ235), gedatolisib (PKI-587), omipalisib (GSK2126458), apitolisib (GDC-0980), bimiralisib (PQR309), and voxtalisib (XL765), have advanced to Phase I and Phase II clinical trials [33]. Rapamycin, discovered from a soil bacterium (Streptomyces hygroscopicus), as an anti-fungal agent, in the 1970s, along with its analogs, everolimus and temsirolimus, was among the first compounds approved to treat cancer by inhibiting mTOR, the so-called mechanistic target of rapamycin [32,33].
Nucleotide synthesis inhibitors
Targeting nucleotide synthesis has been one of the initial strategies in fighting malignancy [21]. The initial drugs, proven effective in cancer treatment, turned out to be inhibitors of folic acid synthesis in one-carbon metabolism, which is necessary for supporting nucleotide synthesis. Aminopterin, which is a structural analogue of folic acid, was the first antifolate drug found to induce temporary remissions in children with acute lymphocytic leukemia in 1948 [34]. Since then, interventions in nucleotide synthesis and DNA replication have been one of the most effective treatments in cancer therapy. Inosine monophosphate dehydrogenases (IMPDH) are crucial in the final steps of purine synthesis, which generate IPM for the synthesis of adenine and guanine nucleotides. IMPDHs are crucial targets in cancer [35]. Similarly, inhibiting DHODH inhibits cancer growth [36]. DHODH enhances cell growth signaling and provides precursors that support anabolism, making it a crucial focus of cancer therapy research [37-39]. Inhibiting DHODH in any way causes cell proliferation to stop (review) [36]. Interestingly, DHODH activity is linked to respiration, as QH2 oxidation is necessary to sustain its activity [21]. DHODH keeps ferroptosis inhibited, supports cell proliferation, and stimulates ROS generation for signaling [40]. Although DHODH inhibitors alone have shown less effect than they once did, researchers have proposed the use of other drugs to repurpose the DHODH-based therapy in pre-clinical models. A combination of DHODH inhibitors, leflunomide, teriflunomide, brequinar, and the equilibrative nucleoside transporter (ENT) inhibitor dipyridamole offers a potential anti-cancer strategy for certain tumors [41,42]. PRPS2 homologous isoenzyme, PRPS1, demonstrates significant resistance to thiopurine treatment in patients with Burkitt's lymphoma (BL) [43]. The 8q24 chromosomal translocation involving the c-Myc gene, a crucial molecular marker of BL, is linked to upregulated activity of PRPS1/2 in BL patients [43]. High-dose methotrexate (MTX) and cytarabine (ARA-C) significantly improve outcomes in high-risk pediatric patients with BL, except for a subgroup with more than four times the normal activity of lactate dehydrogenase [43]. The enzyme ribonucleotide reductase (RNR), activated by the Rb-E2F axis during the S-phase, converts NTPs into deoxynucleotide triphosphates (dNTPs) for DNA replication and is a promising target in cancer therapy [21,44]. RNR is not present in non-dividing cells and in the G1 phase of the cell cycle. CDK1/2-mediated phosphorylation of RNR at Thr33 of the PRM2 subunit promotes its degradation via cyclin F (i.e., SCFcyclinF ligase) [45]. Stabilizing Cyclin F by blocking its ubiquitination and degradation via APC/C and SCF complexes has been shown to promote cell death [45]. Most cancer drugs targeting RNR are nucleoside analogs that mimic the chemical structure of nucleosides. Hydroxyurea (HU), one of the earliest recognized RNR inhibitors, acts as a radical scavenger with low inhibitory activity against RNR and exhibits various off-target effects through its radical-quenching ability [46]. Several therapeutic RNR inhibitors, including gemcitabine, clofarabine, and hydroxyurea, are used clinically to treat a variety of cancers [46]. Gemcitabine (2′,2′-difluorodeoxycytidine) is one of the few FDA-approved RNR inhibitors approved for the clinical treatment of pancreatic cancer [46]. A major challenge to the current effectiveness of RNR inhibitors is resistance, primarily caused by high levels of RNR expression, which allows tumor cells to repair DNA damage caused by chemotherapy. The exploration of the full range of RNR inhibitors and the main challenges ahead is reviewed here [47,48].
Protein synthesis and anticancer targets
The AKT-mTORC1 axis is the primary regulator of protein synthesis, promoting ribosomal biogenesis, the nucleoprotein complex responsible for translating mRNA into proteins. Cell proliferation begins with ribosomal biosynthesis, which remains inhibited in cells not actively growing. Protein synthesis is often upregulated in cancer cells due to impaired sensing mechanisms that normally tightly regulate protein production and its turnover through degradation by autophagy and ubiquitination. AKT activates mTORC1 by phosphorylating and inhibiting its repressor, and autophagy activator tuberous sclerosis complex 2 (TSC2). When TSC2 is inhibited, it releases Rheb GTPase, which then binds to and directly activates mTORC1 [49,50]. mTOR, especially mTORC1, is an activator of protein and nucleotide synthesis through activating ribosomal biogenesis and pyrimidine synthesis via S6K and S and an activator of lipid synthesis through controlling sterol regulatory element binding protein (SREBP). S6K phosphorylation activates CAD for pyrimidine synthesis and inhibits 4E-BP that derepresses mRNA translation [51]. 4E-BP is a protein synthesis repressor that binds to and inhibits eIF4E, an essential mRNA translation initiation factor [49,50]. Elevated S6K activity enhances epithelial-mesenchymal transition (EMT), cancer stemness, and resistance to drugs [52]. Currently, the only S6K inhibitors available for clinical use are rapamycin analogs (rapalogs) that also inhibit the mTOR pathway [53]. Antibiotics that target prokaryotic protein synthesis have been central to treating infectious diseases. Likewise, chemicals that inhibit eukaryotic mRNA translation could significantly expand the anticancer therapy options. Several chemicals have been identified that target key nodes in protein synthesis in curbing cancerous growth. Inhibitors of protein synthesis, such as Silvestrol, Narciclasine, and Plitidepsin, have shown strong anticancer effects by interfering with important steps of translation [54]. These compounds offer new treatment options to slow tumor growth and enhance existing therapies [54]. Tumor cells develop resistance by mutating parts of the protein synthesis machinery, like eIF4A or eIF2α, or by changing drug transport systems. This adaptation lessens the long-term effectiveness of these treatments [54]. Overcoming these resistance mechanisms is crucial to unlocking the full therapeutic potential of protein synthesis inhibitors.
Lipogenesis and anticancer targets
The AKT-SREBP axis upregulates lipogenesis by increasing the expression of citrate-metabolizing enzyme ACLY, acetyl-CoA carboxylase (ACC1), fatty acid synthase (FASN), stearoyl-CoA desaturase (SCD), and glycerol-3-phosphate acyltransferase [55,56]. SREBP-1 and SREBP-2 also enhance the transcription of enzymes such as cytoplasmic acetyl-CoA synthetase (ACSS2), which activates acetate to acetyl-CoA, and acetyl-CoA carboxylase 1 (ACC1), which produces malonyl-CoA by carboxylating acetyl-CoA [57]. Malonyl-CoA is the polymerizing unit in fatty acid synthesis [57]. Cytoplasmic acetyl-CoA is a key source for fatty acid synthesis in proliferating, especially cancer, cells, which have a strong ability for de novo fatty acid production due to often increased upregulation of fatty acid synthesizing enzymes-ACC1, ACSS2, ACLY and FASN [58-60]. Normal proliferating cells primarily rely on lipid uptake from the blood. De novo fatty acid synthesis mainly occurs in adipose tissue, liver, lactating breast, and proliferating fetal cells [57]. Cancer cells from these tissues exhibit enhanced lipogenesis [57,61]. Oxidative stress has been known to activate FASN in hepatoma via SREBP1c activation [62]. SODs decrease oxidative stress by neutralizing superoxide radical, and SOD1 deficiency elevates fatty acid synthesis and lipid droplets in hepatic cancer [63]. Lipid also accumulates in osteoblasts due to oxidative stress and aging. [64,65]. Mitochondrial dysfunction increases lipid synthesis as a generalized response to oxidative stress [66]. Obesity caused by aging and oxidative stress has been a key factor in diseases like cancer and insulin resistance. [67]. With ageing, the number of adipocytes and the amount of adipose tissue increase, while the pool of stem cells and regenerative capacity significantly decline. Interestingly, ACLY is a key enzyme that links glycolytic and lipid metabolism. The expression and activity of ACLY, FASN, and other lipogenic enzymes are often abnormally elevated in various tumors. Inhibiting them pharmacologically or genetically significantly reduces cancer cell growth and triggers programmed cell death, or apoptosis. High fructose intake due to modern lifestyles leads to the production of acetate by the gut microbiome and increased acetyl-CoA synthesis in hepatocytes [68]. Increased fructose signaling also stimulates lipogenic enzymes in these cells [68]. As a result, fructose consumption promotes increased fatty acid synthesis in hepatocytes, partly connecting the modern diet with non-alcoholic fatty liver disease and also increasing the risk of hepatoma [68]. Inhibiting lipogenesis through FASN and ACLY is a promising approach to control cell growth in cancer [69-71]. Cancer cells highly express the ACLY and fatty acid synthase (FASN) enzymes [60]. The majority of cancer cells increase de novo lipid synthesis as well as lipid uptake due to oncogenic signaling [57]. Inhibiting fatty acid synthesis not only depletes lipid stores used to build membranes but also reduces phospholipids crucial for growth signaling, like PI3K signaling [70]. Inhibiting lipogenesis has been a promising strategy to reduce cancer growth, especially in tissues that metabolize and store fatty acids, such as adipose tissue, breast, and pancreas. Recently, combined therapy targeting lipogenesis has been introduced for more effective treatment. Combined inhibition of ACLY by bempedoic acid and G1-S phase transitioning kinase CDK4/6 by palbociclib reduces growth in breast and pancreatic cancer cells [72]. It appears that many cancers, including breast cancer cells, develop resistance to human epidermal growth factor receptor (HER) targeting drugs such as cetuximab and trastuzumab (or Herceptin) by rewiring lipid metabolism [73,74].
Redox homeostasis disruption and cancer therapy
High NADPH promotes reductive reactions like the synthesis of nucleotides, proteins and lipids as well as redox homeostasis due to high metabolic turnover associated with cell growth. Moreover, cytosolic and mitochondrial pools of NADPH and NADH are separate and are maintained independently. An increase in mitochondrial NADPH supports the reductive reaction in the TCA cycle by converting glutamine-derived α-ketoglutarate to isocitrate and then to citrate, which is a robust precursor for lipogenesis in the cytoplasm of cancer cells. NRF2 increases the transcription of the cystine-glutamate antiporter (also known as xCT and SLC7A11), which enhances the intracellular uptake of cysteine, an essential component of GSH. [15,16]. Paradoxically, this transporter exports glutamate for cysteine import, decreasing GSH substrate glutamate. However, plasma and cells typically have a high concentration of glutamine and can replenish glutamate via the citrate-α-ketoglutarate pathway as well as through increased glutamine uptake by other transporters [75]. In addition to uptake, cysteine can be derived from methionine through the trans-sulfuration pathway. This pathway connects GSH synthesis with one-carbon metabolism, which generates serine, glycine, and S-adenosylmethionine (SAM) [76]. Since AKT phosphorylation of xCT inhibits its transport function, leading to low cellular cysteine levels, malignant tumors with activated or deregulated PI3K-AKT signaling rely heavily on the trans-sulfuration pathway for cysteine supply via methionine, which is used for GSH synthesis. [77]. Methionine restriction inhibits the growth of colon, prostate, and breast cancers [78-80]. GSH depletion is known to increase the sensitivity of cancer cells to many anticancer therapeutic agents [77]. Similarly, KEAP1 dysfunction, which causes constant NRF2 activation, has been associated with tumor development in the ovary, esophagus, gallbladder, prostate, kidney, and non-small cell lung cancer (NSCLC) [81]. Deregulation of metabolism in cancer cells boosts NADPH production via multiple pathways, supporting cancer cell synthesis and antioxidants needed to counteract high ROS generated during various reactions essential for cell growth [17]. In contrast, abrupt or sustained ROS causes stem cell activation, which can exhaust the stem cell pool and lead to tumorigenesis if activated in resting cells, including cells in the liver, pancreas, lung, and central nervous system [82,83]. Endothelial cells activate complex III to produce the necessary ROS for cell migration, independent of OXPHOS-mediated ATP production [84]. Nicotinamide nucleotide transhydrogenase (NNT) deficiency impairs cell proliferation, while excessive activity promotes tumorigenesis, as NNT increases the mitochondrial NADPH pool by converting NADP+ to NADPH through the oxidation of NADH [85,86]. NNT is often regarded as a complex VI because it is situated on the matrix side of the inner mitochondrial membrane and also uses the proton motive force to reduce NADP+. In proliferating cells, PMF is higher than in cells at rest due to increased fuel influx and reduced OXPHOS, and NNT activity promotes tumor growth and metastasis in gastric cancer [86]. NNT also contributes to non-small cell lung carcinoma (NSCLC) by maintaining Fe-S centers in the mitochondrial electron transfer chain [87].
NADK is the only enzyme that converts NAD+ to NADP+ in both the cytosol (NADK1/cNADK) and mitochondria (NADK2/mNADK) [88]. Mitochondrial NADP+ limits NADPH production, as NADK2-deficient mammalian cells show growth defects caused by reduced NADPH and proline synthesis [89]. Proline supplementation enhances cell growth in NADK2-deficient cells, rendering them proline auxotrophs [89]. Removing cytosolic NADK hampers cell growth in human plasma-like medium [90]. Interestingly, NADKs (NADK1 and NADK2) are activated through phosphorylation by various kinases, such as AKT, protein kinase C (PKC), and ERK, which are triggered by mitogenic signals in different proliferating cells under both normal and abnormal signaling conditions [91,92]. NADPH phosphatase, which opposes the action of NADKs, causes cellular NADPH depletion, elevating oxidative stress and leading to ferroptotic cell death [93]. Conversely, gene depletion helps maintain cellular NADPH levels, shielding cells from ferroptosis [93]. The prodrug thionicotinamide (TN) is converted into NADS and NADPS inside cells and acts as a dual inhibitor of NADK and glucose-6-phosphate dehydrogenase (G6PD), which catalyzes NADPH production in cells. Inhibiting NADK and G6PD makes cancer cells more vulnerable to death caused by increased ROS levels [88,94]. Furthermore, NADK inhibitors make cancer cells more responsive to other ROS-inducing treatments, such as gemcitabine and cisplatin and ferroptotic agents [88]. Knockdown of NNT significantly decreases NADPH levels, causes high ROS production, and results in apoptosis in gastric cancer cells under oxidative stress conditions [86]. NNT inhibition through knockdown or other methods has led to a significant decrease in cancer growth. However, known chemicals that can inhibit NNTs are not available, highlighting a future research direction to identify such drugs.
Conclusion and future prospects
Metabolic targeting in cancer therapy offers a promising strategy by exploiting cancer cells' unique metabolic needs. However, current trials encounter obstacles due to tumor heterogeneity and their capacity for metabolic adaptation, which can cause resistance to single therapies. Going forward, integrating metabolomics with genomic profiling could help identify vulnerabilities specific to each patient. Additionally, developing agents that target multiple facets of cancer metabolism may enhance treatment success, diminish resistance, and reduce systemic toxicity by reducing the requirement for additional inhibitors, resulting in improved clinical results.
Acknowledgment- This work received no funding.
Conflict of Interest- The authors declare no conflict of interest.
References
- RL Siegel, AN Giaquinto, A Jemal. Cancer statistics, 2024, CA. Cancer J. Clin. 74 (2024) 12-49.
- A Stincone, A Prigione, T Cramer, et al. The return of metabolism: Biochemistry and physiology of the pentose phosphate pathway, Biol. Rev. 90 (2015) 927-963.
- AA Cluntun, H Huang, L Dai, et al. The rate of glycolysis quantitatively mediates specific histone acetylation sites, Cancer Metab 3 (2015) 10.
- ÁD Ortega, M Sánchez-Aragó, D Giner-Sánchez, et al. Glucose avidity of carcinomas, Cancer Lett. 276 (2009) 125-135.
- PF Oliveira, AD Martins, AC Moreira, et al. The Warburg Effect Revisited-Lesson from the Sertoli Cell, Med. Res. Rev. 35 (2015) 126-151.
- G Hoxhaj, BD Manning. The PI3K-AKT network at the interface of oncogenic signalling and cancer metabolism, Nat. Rev. Cancer 20 (2020) 74-88.
- VS Adusumilli, TL Walker, RW Overall, et al. ROS Dynamics Delineate Functional States of Hippocampal Neural Stem Cells and Link to Their Activity-Dependent Exit from Quiescence, Cell Stem Cell 28 (2021) 300-314.e6.
- BC Dickinson, J Peltier, D Stone, et al. Nox2 redox signaling maintains essential cell populations in the brain, Nat. Chem. Biol. 7 (2011) 106-112.
- H Morimoto, T Yamamoto, T Miyazaki, et al. An interplay of NOX1-derived ROS and oxygen determines the spermatogonial stem cell self-renewal efficiency under hypoxia, Genes Dev. 35 (2021) 250-260.
- G Aviello, UG Knaus. NADPH oxidases and ROS signaling in the gastrointestinal tract review-article, Mucosal Immunol. 11 (2018) 1011-1023.
- ZM Moghadam, P Henneke, J Kolter. From Flies to Men: ROS and the NADPH Oxidase in Phagocytes, Front. Cell Dev. Biol. 9 (2021) 628991.
- CV Dang. MYC, metabolism, cell growth, and tumorigenesis, Cold Spring Harb. Perspect. Med. 3 (2013).
- JT Cunningham, MV Moreno, A Lodi, et al. Protein and nucleotide biosynthesis are coupled by a single rate-limiting enzyme, PRPS2, to drive cancer, Cell 157 (2014) 1088-1103.
- YC Tang, JR Hsiao, SS Jiang, et al. c-MYC-directed NRF2 drives malignant progression of head and neck cancer via glucose-6-phosphate dehydrogenase and transketolase activation, Theranostics 11 (2021) 5232-5247.
- W Li, AN Kong. Molecular mechanisms of Nrf2-mediated antioxidant response, Mol. Carcinog. 48 (2009) 91-104.
- K Itoh, T Chiba, S Takahashi, et al. An Nrf2/small Maf heterodimer mediates the induction of phase II detoxifying enzyme genes through antioxidant response elements, Biochem. Biophys. Res. Commun. 236 (1997) 313-322.
- GM Rather, AA Pramono, Z Szekely, et al. In cancer, all roads lead to NADPH, Pharmacol. Ther. 226 (2021).
- JJ Loh, S Ma. Hallmarks of cancer stemness, Cell Stem Cell 31 (2024) 617-639.
- G Peng, Y Liu. Hypoxia-inducible factors in cancer stem cells and inflammation, Trends Pharmacol. Sci. 36 (2015) 374-383.
- XD Zheng, HY Li, SY Gao, et al. High hypoxia inducible factor-1α expression is associated with reduced survival in patients with breast cancer: A meta-analysis, World J. Clin. Oncol. 16 (2025) 105691.
- NJ Mullen, PK Singh. Nucleotide metabolism: a pan-cancer metabolic dependency, Nat. Rev. Cancer 23 (2023) 275-294.
- Y Yang, L Song, X Huang, et al. PRPS1-mediated purine biosynthesis is critical for pluripotent stem cell survival and stemness, Aging (Albany. NY). 13 (2021) 4063-4078.
- DR Wise, RJ Deberardinis, A Mancuso, et al. Myc regulates a transcriptional program that stimulates mitochondrial glutaminolysis and leads to glutamine addiction, Proc. Natl. Acad. Sci. U. S. A. 105 (2008) 18782-18787.
- L Zhang, X Wang, R Cueto, et al. Biochemical basis and metabolic interplay of redox regulation, Redox Biol. 26 (2019).
- F Weinberg, NS Chandel. Reactive oxygen species-dependent signaling regulates cancer, Cell. Mol. Life Sci. 66 (2009) 3663-3673.
- B Xiong, Y Zhang, S Liu, et al. NOX Family: Regulators of Reactive Oxygen Species Balance in Tumor Cells, FASEB J. 39 (2025) e70565.
- A Parascandolo, MO Laukkanen. Carcinogenesis and reactive oxygen species signaling: Interaction of the NADPH oxidase NOX1-5 and superoxide dismutase 1-3 signal transduction pathways, Antioxidants Redox Signal. 30 (2019) 443-486.
- J Amengual, E Gonzalez-Sanchez, M Yáñez-Bartolome, et al. NADPH oxidase 1/4 dual inhibition impairs transforming growth factor-beta protumorigenic effects in cholangiocarcinoma cancer-associated fibroblasts, Signal Transduct. Target. Ther. 10 (2025) 257.
- T Yu, L Li, W Liu, et al. Silencing of NADPH oxidase 4 attenuates hypoxia resistance in neuroblastoma cells SH-SY5Y by inhibiting PI3K/Akt-dependent glycolysis, Oncol. Res. 27 (2019) 525-532.
- J Reis, C Gorgulla, M Massari, et al. Targeting ROS production through inhibition of NADPH oxidases, Nat. Chem. Biol. 19 (2023) 1540-1550.
- BA Diebold, SME Smith, Y Li, et al. NOX2 as a target for drug development: Indications, possible complications, and progress, Antioxidants Redox Signal. 23 (2015) 375-405.
- M Laplante, DM Sabatini. MTOR signaling in growth control and disease, Cell 149 (2012) 274-293.
- B Mao, Q Zhang, L Ma, et al. Overview of Research into mTOR Inhibitors, Molecules 27 (2022) 5295.
- O Shuvalov, A Petukhov, A Daks, et al. One-carbon metabolism and nucleotide biosynthesis as attractive targets for anticancer therapy, Oncotarget 8 (2017) 23955-23977.
- H Zhu, H Wang, X Li, et al. IMP metabolic mechanisms and IMPDH targeting strategies in tumor metabolic reprogramming and therapy (Review), Int. J. Mol. Med. 57 (2026).
- S Boukalova, S Hubackova, M Milosevic, et al. Dihydroorotate dehydrogenase in oxidative phosphorylation and cancer, Biochim. Biophys. Acta - Mol. Basis Dis. 1866 (2020).
- Y Zhou, L Tao, X Zhou, et al. DHODH and cancer: promising prospects to be explored, Cancer Metab. 9 (2021).
- I Elia, D Broekaert, S Christen, et al. Proline metabolism supports metastasis formation and could be inhibited to selectively target metastasizing cancer cells, Nat. Commun. 8 (2017) 1-11.
- X Xu, G Zhang, Y Chen, et al. Can proline dehydrogenase—a key enzyme involved in proline metabolism—be a novel target for cancer therapy?, Front. Oncol. 13 (2023).
- RJ Mailloux. Proline and dihydroorotate dehydrogenase promote a hyper-proliferative state and dampen ferroptosis in cancer cells by rewiring mitochondrial redox metabolism, Biochim. Biophys. Acta - Mol. Cell Res. 1871 (2024) 119639.
- CR Cuthbertson, H Guo, A Kyani, et al. The Dihydroorotate Dehydrogenase Inhibitor Brequinar Is Synergistic with ENT1/2 Inhibitors, ACS Pharmacol. Transl. Sci. 3 (2020) 1242-1252.
- R Rajamohamed, S Veerappapillai. Identification of novel dihydroorotate dehydrogenase (DHODH) inhibitors for cancer: computational drug repurposing strategy, BMC Pharmacol. Toxicol. 26 (2025).
- T Li, L Song, Y Zhang, et al. Molecular mechanism of c-Myc and PRPS1/2 against thiopurine resistance in Burkitt’s lymphoma, J. Cell. Mol. Med. 24 (2020) 6704-6715.
- A Hofer, M Crona, DT Logan, et al. DNA building blocks: Keeping control of manufacture, Crit. Rev. Biochem. Mol. Biol. 47 (2012) 50-63.
- V D’Angiolella, V Donato, FM Forrester, et al. Cyclin F-mediated degradation of ribonucleotide reductase M2 controls genome integrity and DNA repair, Cell 149 (2012) 1023-1034.
- R Kohnken, KM Kodigepalli, L Wu. Regulation of deoxynucleotide metabolism in cancer: Novel mechanisms and therapeutic implications, Mol. Cancer 14 (2015).
- BL Greene, G Kang, C Cui, et al. Ribonucleotide Reductases: Structure, Chemistry, and Metabolism Suggest New Therapeutic Targets, Annu. Rev. Biochem. 89 (2020) 45-75.
- SE Huff, JM Winter, CG Dealwis. Inhibitors of the Cancer Target Ribonucleotide Reductase, Past and Present, Biomolecules 12 (2022) 815.
- J Kim, KL Guan. mTOR as a central hub of nutrient signalling and cell growth, Nat. Cell Biol. 21 (2019) 63-71.
- KM Hannan, Y Brandenburger, A Jenkins, et al. mTOR-Dependent Regulation of Ribosomal Gene Transcription Requires S6K1 and Is Mediated by Phosphorylation of the Carboxy-Terminal Activation Domain of the Nucleolar Transcription Factor UBF, Mol. Cell. Biol. 23 (2003) 8862-8877.
- I Ben-Sahra, JJ Howell, JM Asara, et al. Stimulation of de novo pyrimidine synthesis by growth signaling through mTOR and S6K1, Science 339 (2013) 1323-1328.
- A Bdzhola, O Malanchuk, S Palchevskyi, et al. Co-expression of the RPS6KB1 and PDPK1 genes for production of activated p70S6K1 using bac-to-bac baculovirus expression system, Mol. Biol. Rep. 52 (2025).
- M Artemenko, SSW Zhong, SKY To, et al. p70 S6 kinase as a therapeutic target in cancers: More than just an mTOR effector, Cancer Lett. 535 (2022).
- L Liu, Z Li, W Wu. Harnessing natural inhibitors of protein synthesis for cancer therapy: A comprehensive review, Pharmacol. Res. 209 (2024) 107449.
- JD Horton, JL Goldstein, MS Brown. SREBPs: activators of the complete program of cholesterol and fatty acid synthesis in the liver, J. Clin. Invest. 109 (2002) 1125-1131.
- RA DeBose-Boyd, J Ye. SREBPs in Lipid Metabolism, Insulin Signaling, and Beyond, Trends Biochem. Sci. 43 (2018) 358-368.
- F Röhrig, A Schulze. The multifaceted roles of fatty acid synthesis in cancer, Nat. Rev. Cancer 16 (2016) 732-749.
- DR Crooks, N Maio, AN Lane, et al. Acute loss of iron-sulfur clusters results in metabolic reprogramming and generation of lipid droplets in mammalian cells, J. Biol. Chem. 293 (2018) 8297-8311.
- L Castro, V Tórtora, S Mansilla, et al. Aconitases: Non-redox Iron-Sulfur Proteins Sensitive to Reactive Species, Acc. Chem. Res. 52 (2019) 2609-2619.
- C Martinez Calejman, S Trefely, SW Entwisle, et al. mTORC2-AKT signaling to ATP-citrate lyase drives brown adipogenesis and de novo lipogenesis, Nat. Commun. 11 (2020) 575.
- JA Menendez, R Lupu. Fatty acid synthase and the lipogenic phenotype in cancer pathogenesis, Nat. Rev. Cancer 7 (2007) 763-777.
- M Sekiya, A Hiraishi, M Touyama, et al. Oxidative stress induced lipid accumulation via SREBP1c activation in HepG2 cells, Biochem. Biophys. Res. Commun. 375 (2008) 602-607.
- J Lee, T Homma, T Kurahashi, et al. Oxidative stress triggers lipid droplet accumulation in primary cultured hepatocytes by activating fatty acid synthesis, Biochem. Biophys. Res. Commun. 464 (2015) 229-235.
- A Nandy, A Richards, S Thapa, et al. Altered Osteoblast Metabolism with Aging Results in Lipid Accumulation and Oxidative Stress Mediated Bone Loss, Aging Dis. 15 (2024) 767-786.
- JJ Cao, TJ Wronski, U Iwaniec, et al. Aging increases stromal/osteoblastic cell-induced osteoclastogenesis and alters the osteoclast precursor pool in the mouse, J. Bone Miner. Res. 20 (2005) 1659-1668.
- SJ Lee, J Zhang, AMK Choi, et al. Mitochondrial dysfunction induces formation of lipid droplets as a generalized response to stress, Oxid. Med. Cell. Longev. (2013).
- P Manna, SK Jain. Obesity, Oxidative Stress, Adipose Tissue Dysfunction, and the Associated Health Risks: Causes and Therapeutic Strategies, Metab. Syndr. Relat. Disord. 13 (2015) 423-444.
- S Zhao, C Jang, J Liu, et al. Dietary fructose feeds hepatic lipogenesis via microbiota-derived acetate, Nature 579 (2020) 586-591.
- R Lupu, J Menendez. Pharmacological Inhibitors of Fatty Acid Synthase (FASN)-Catalyzed Endogenous Fatty Acid Biogenesis: A New Family of Anti-Cancer Agents?, Curr. Pharm. Biotechnol. 7 (2006) 483-494.
- K Tomek, R Wagner, F Varga, et al. Blockade of fatty acid synthase induces ubiquitination and degradation of phosphoinositide-3-kinase signaling proteins in ovarian cancer, Mol. Cancer Res. 9 (2011) 1767-1779.
- A Khwairakpam, M Shyamananda, B Sailo, et al. ATP Citrate Lyase (ACLY): A Promising Target for Cancer Prevention and Treatment, Curr. Drug Targets 16 (2015) 156-163.
- BC Velez, CP Petrella, KH Disalvo, et al. Combined inhibition of ACLY and CDK4/6 reduces cancer cell growth and invasion, Oncol. Rep. 49 (2023) 32.
- WW Feng, M Kurokawa. Lipid metabolic reprogramming as an emerging mechanism of resistance to kinase inhibitors in breast cancer, Cancer Drug Resist. 3 (2020) 1-17.
- KK Ray, HE Bays, AL Catapano, et al. Safety and Efficacy of Bempedoic Acid to Reduce LDL Cholesterol, N. Engl. J. Med. 380 (2019) 1022-1032.
- YD Bhutia, V Ganapathy. Glutamine transporters in mammalian cells and their functions in physiology and cancer, Biochim. Biophys. Acta - Mol. Cell Res. 1863 (2016) 2531-2539.
- J Lewerenz, SJ Hewett, Y Huang, et al. The cystine/glutamate antiporter system xc- in health and disease: From molecular mechanisms to novel therapeutic opportunities, Antioxidants Redox Signal. 18 (2013) 522-555.
- EC Lien, CA Lyssiotis, A Juvekar, et al. Glutathione biosynthesis is a metabolic vulnerability in PI(3)K/Akt-driven breast cancer, Nat. Cell Biol. 18 (2016) 572-578.
- D Komninou, Y Leutzinger, BS Reddy, et al. Methionine restriction inhibits colon carcinogenesis, Nutr. Cancer 54 (2006) 202-208.
- S Lu, DE Epner. Molecular mechanisms of cell cycle block by methionine restriction in human prostate cancer cells, Nutr. Cancer 38 (2000) 123-130.
- EC Lien, L Ghisolfi, RC Geck, et al. Oncogenic PI3K promotes methionine dependency in breast cancer cells through the cystine-glutamate antiporter xCT, Sci. Signal. 10 (2017).
- B Lv, S Xing, Z Wang, et al. NRF2 inhibitors: Recent progress, future design and therapeutic potential, Eur. J. Med. Chem. 279 (2024) 116822.
- P Chaudhari, Z Ye, YY Jang. Roles of reactive oxygen species in the fate of stem cells, Antioxidants Redox Signal. 20 (2014) 1881-1890.
- EC Cheung, KH Vousden. The role of ROS in tumour development and progression, Nat. Rev. Cancer 22 (2022) 280-297.
- LP Diebold, HJ Gil, P Gao, et al. Mitochondrial complex III is necessary for endothelial cell proliferation during angiogenesis, Nat. Metab. 1 (2019) 158-171.
- HY Ho, YT Lin, G Lin, et al. Nicotinamide nucleotide transhydrogenase (NNT) deficiency dysregulates mitochondrial retrograde signaling and impedes proliferation, Redox Biol. 12 (2017) 916-928.
- S Li, Z Zhuang, T Wu, et al. Nicotinamide nucleotide transhydrogenase-mediated redox homeostasis promotes tumor growth and metastasis in gastric cancer, Redox Biol. 18 (2018) 246-255.
- NP Ward, YP Kang, A Falzone, et al. Nicotinamide nucleotide transhydrogenase regulates mitochondrial metabolism in NSCLC through maintenance of Fe-S protein function, J. Exp. Med. 217 (2020).
- AA Pramono, GM Rather, H Herman, et al. NAD- and NADPH-contributing enzymes as therapeutic targets in cancer: An overview, Biomolecules 10 (2020) 358.
- DH Tran, R Kesavan, H Rion, et al. Mitochondrial NADP+ is essential for proline biosynthesis during cell growth, Nat. Metab. 3 (2021) 571-585.
- KM Flickinger, CA Mellado Fritz, KS Huggler, et al. Cytosolic NADK is conditionally essential for folate-dependent nucleotide synthesis, Nat. Metab. 7 (2025) 1150-1167.
- G Hoxhaj, I Ben-Sahra, SE Lockwood, et al. Direct stimulation of NADP+ synthesis through Akt-mediated phosphorylation of NAD kinase, Science 363 (2019) 1088-1092.
- R Rabani, C Cossette, F Graham, et al. Protein kinase C activates NAD kinase in human neutrophils, Free Radic. Biol. Med. 161 (2020) 50-59.
- CKC Ding, J Rose, T Sun, et al. MESH1 is a cytosolic NADPH phosphatase that regulates ferroptosis, Nat. Metab. 2 (2020) 270-277.
- PM Tedeschi, HX Lin, M Gounder, et al. Suppression of Cytosolic NADPH Pool by Thionicotinamide Increases Oxidative Stress and Synergizes with Chemotherapy, Mol. Pharmacol 88 (2015) 720-727.


 Impact Factor:
* 3.3
Impact Factor:
* 3.3
 Acceptance Rate:
74.39%
Acceptance Rate:
74.39%
 Time to first decision: 10.4 days
Time to first decision: 10.4 days
 Time from article received to acceptance:
2-3 weeks
Time from article received to acceptance:
2-3 weeks
