The Development, Efficacy and Safety Profiles of Bispecific Antibodies for Solid Tumors: Amivantamab for EGFR-mutant NSCLC and Tebentafusp for Uveal Melanoma
Priya Hays
Hays Documentation Specialists LLC, San Mateo, CA, USA
* Corresponding author: Priya Hays, Hays Documentation Specialists LLC, San Mateo, CA, USA
Received: 20 February 2026; Accepted: 26 February 2026; Published: 01 April 2026
Article Information
Citation: Priya Hays. The Development, Efficacy and Safety Profiles of Bispecific Antibodies for Solid Tumors: Amivantamab for EGFR-mutant NSCLC and Tebentafusp for Uveal Melanoma. Archives of Microbiology and Immunology. 9 (2026): 24-35.
View / Download Pdf Share at FacebookAbstract
Background: This review will describe the unique mechanisms of action of Amivantamab (EGFR NSCLC) and Tebentafusp (uveal melanoma), two bispecific antibodies approved for solid tumors. It will trace their paths to FDA approval, from preclinical development through first-in-human and pivotal clinical trials, and examine the distinct safety profiles of each agent to provide insights into their therapeutic potential and clinical integration.
Methods: A PubMed search was conducted with the search terms “Bispecific Antibodies” AND “Solid Tumors” AND “EGFR-mutant NSCLC” AND “Uveal Melanoma” AND “Amivantamab” AND “Tebentafusp.”
Results: Amivantamab demonstrated efficacy in wild-type EGFR NSCLC tumors and is linked to immune cell-mediated killing and inhibition of binding to both EGFR and Met receptors. Preclinical studies of Ba/F3 cell lines containing EGFR exon20ins have found that amivantamab decreased EGFR and MET receptor expression. The phase I CHRYSALIS trial evaluated amivantamab in a dose escalation/dose expansion study. The phase 3 MARIPOSA trials investigated amivantamab in combination with chemotherapy and the tyrosine kinase inhibitor Lazertinib. Median progression-free survival was 8.3 months (95% CI: 6.5–10.9) and median overall survival was 22.8 months (95% CI: 14.6-NR). Grade 3 and higher AEs include dyspnea (7%), infusion-related reactions (5%), and hypoalbuminemia (4%). Tebentafusp is a bispecific protein fused to an anti-CD3 single-chain variable fragment that targets glycoprotein 100-positive cells, and was evaluated in two, open label phase 3 trials for uveal melanoma resulting in an overall survival of 73% in the tebentafusp group and 59% in the control group in one trial; and a median overall survival of 21.6 months in the tebentafusp group and 16.9 months in the control group (HR 0.68; 95% CI, 0.54 to 0.87) in the subsequent trial. The most common treatment-related adverse events in the tebentafusp group were cytokine-mediated events (due to T-cell activation) and skin-related events (due to glycoprotein 100–positive melanocytes), including rash (83%), pyrexia (76%), and pruritus (69%).
Conclusion: There is ample evidence for the clinical use of amivantamab and tebentafusp in non-small cell lung cancer and uveal melanoma with demonstrated efficacy and favorable safety profiles. Ongoing studies are focused on applying liquid biopsy to monitoring of treatment and prognosis.
Keywords
Bispecific T cell engagers, solid tumors, amivantamab, EGFR-mutant NSCLC, tebentafusp, uveal melanoma
Article Details
Introduction
Non-small cell lung cancer or NSCLC is typically treated with tyrosine kinase inhibitors and third-line TKIs such as Osimertinib that have been found to overcome resistance to first- and second-generation inhibitors. First-line treatment includes neo-adjuvant and adjuvant administration of immune checkpoint inhibitors ipilimumab, nivolumab and pembrolizumab with or without chemotherapy. EGFR exon20ins mutations are defined by insertions and duplications near the EGFR kinase domain and contain a modified active site that hinders the binding of EGFR TKI molecules leading to less efficacy, with a median OS of 16 months, highlighting the need to find efficient targets with activity against EGFR exon20ins mutations. [1] EGFR TKIs can also induce MET receptor amplification causing signaling downstream to promote cancer cell proliferation and resistance to SOC treatments. The discovery of amivantamab (formerly JNJ-61186372), a bispecific antibody targeting EGFR and MET, was conducted in a series of experiments and assays by Neijssen et al. NSCLC driven by EGFR exon 20 ins has a worse prognosis and worse OS than classic sensitizing EGFR mutations such as exon 19 del and L858R, and there is resistance to first second and third generation EGFR TKIs. MET activation is a primary oncogenic driver whose pathway dysregulation leads to proliferation, invasion and metastasis of tumor cells. MET amplification bypasses EGFR signaling pathways and confers resistance to Osimertinib. EGFR and MET, mesenchymal-epithelial transition, are co-expressed in 70% of EGFR mutated cancer. In contrast, healthy cells, almost never concomitantly express both receptors. [2] Amivantamab demonstrated efficacy in wild-type EGFR NSCLC tumors and is linked to immune cell-mediated killing and inhibition of binding to both EGFR and Met receptors. A novel mechanism of action, it targets both the domains of EGFR and MET. Fc-independent MOA block EGFR and MET signaling via ligand binding. The Fc dependent MOA leads to ADCC by the activation of natural killer cells, macrophages and monocytes along with cytokine and chemokine release. [1] Amivantamab has been shown to bind the EGFR and MET receptors extracellularly with binding affinity (KD) of 1.43 and 0.04 nM, respectively in preclinical studies. “Amivantamab binds human EGFR and MET with EC50 values of 0.38 nM 0.27 nM, respectively…Data from in vivo trials showed at doses between 350 and 1750 mg, amivantamab exposure increased proportionally. Steady state was achieved by the ninth infusion.” The RP2D was 168 ug/mL [2].
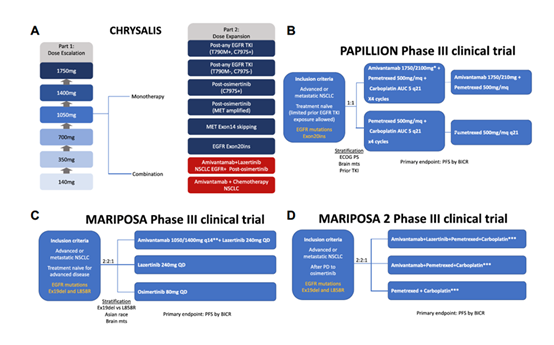
The phase I CHRYSALIS trial evaluated amivantamab in a dose escalation/dose expansion study. The phase 3 MARIPOSA trials investigated amivantamab in combination with chemotherapy and the tyrosine kinase inhibitor Lazertinib. Median progression-free survival was 8.3 months (95% CI: 6.5-10.9) and median overall survival was 22.8 months (95% CI: 14.6-NR). Grade 3 and higher AEs include dyspnea (7%), infusion-related reactions (5%), and hypoalbuminemia (4%). “The side effects are associated with the unique properties of this drug as rash, paronychia, stomatitis, pruritis, and diarrhea resulting from EGFR inhibition while MET inhibition is associated with hypoalbuminemia and peripheral edema.” The initial findings demonstrate an overall tumor response benefit and an acceptable safety profile [1].
Uveal melanoma is the most common intraocular cancer in adults with metastases occurring in over half of patients. It has distinct molecular drivers and patterns than cutaneous melanoma, as well as a different tumor microenvironment. Thus UM contributes poorly to systemic treatment including immune checkpoint inhibitors. Prognosis is poor with a mOS of about a year. Tebentafusp is a bispecific protein fused to an anti-CD3 single-chain variable fragment that targets glycoprotein 100-positive cells that treats uveal melanoma, and was evaluated in two, open label phase 3 trials resulting in an overall survival of 73% in the tebentafusp group and 59% in the control group in one trial; and a median overall survival of 21.6 months in the tebentafusp group and 16.9 months in the control group (HR 0.68; 95% CI, 0.54 to 0.87) in the subsequent trial. The most common treatment-related adverse events in the tebentafusp group were cytokine-mediated events (due to T-cell activation) and skin-related events (due to glycoprotein 100-positive melanocytes), including rash (83%), pyrexia (76%), and pruritus (69%). [3]
Amivantamab for the Treatment of EGFR-Mutant NSCLC
Amivantamab, is a bispecific antibody engineered against NSCLC with MET and EGFR mutations on those who have progressed on TKI therapies. It is a novel BsAb that combines the mechanism of action for EGFR and MET antibodies without inducing receptor dimerization and activation and contains one monovalent arm for EGFR and one for MET. The binding of amivantamab ensures structurally that it leads to novel functional in vivo antitumor activity. The BsAb was isolated through a combinatorial approach in which molecules were screened for EGFR and MET binding on tumor cell lines and lack of agonistic activity toward MET. The need to develop amivantamab was due to resistance of NSCLC toward TKIs as well as the need to overcome this resistance for clinical benefit. [4,5,6]. The process of generating amivantamab was through parental antibody selection criteria and the procedure for generating BsAbs. The anti-gp120 antibody b12 was included as a nonbinding arm with either an EGFR or MET binding arm to generate monovalent control antibodies. In two selection phases involving a binding assay and MET phosphorylation assay, it ensured that the binding properties of the antibodies would bind both targets with a robust EC50(<1ug/ml) and ensured that the bivalent EGFR parent mAbs induced MET phosphorylation. “The remaining EGFR×MET BsAb panel was further tested for (1) inhibition of HGF-driven KP4 cell proliferation expressing an autocrine HGF-MET loop and (2) inhibition of H1975 cell proliferation driven by the EGFR signaling pathway.” All BsAbs except for two showed dose-dependent inhibition of proliferation. The final selection involved choosing the BsAb that contained a specific EGFR arm that inhibited cell proliferation and phosphorylation. “Altogether, the MET B×EGFR H BsAb showed the most optimal properties and with growth in a proprietary cell line for production of parental mAbs with low fucose Fc production, the BsAb became amivantamab.” This antibody was found to specifically interact with MET through paratope-epitope interaction mediated by a high level of specificity. “Amivantamab antagonized HGF-induced signaling of MET by blocking binding of the β-chain of HGF to the MET Sema domain.” After demonstrating EGFR epitope binding, the investigators showed in aa EGFR mutant xenograft model that amivantamab was more effective than EGFR and MET TKIs using a HCC827 cell line that endogenously expresses high levels of EGFR with exon 19 deletion and high levels of MET. Amivantamab demonstrated potent antitumor activity with tumor growth inhibition of 99.8%, proving superior to crizotinib, erlotinib and the combination therapy. The authors concluded that the lead bispecific antibody chosen, MET B X EGFR H, had multiple mechanisms of antitumor activity including ADCC, receptor downmodulation and trogocytosis and that amivantamab may provide significant benefit to patients with NSCLC with associated aberrant EGFR and MET signaling [7].
Amivantamab is an epidermal growth factor receptor (EGFR) and mesenchymal-epithelial transition factor (MET) bispecific antibody designed to enable multiple mechanisms of action and broad clinical applications with a favorable safety profile. Amivantamab was approved in 2021 for mNSCLC with EGFR exon 20 insertions. The binding of amivantamab’s binds to EGFR and MET through its Fab arms preventing their binding to respective receptors. This ligand blocking reduces receptor activation through decreased receptor phosphorylation and downstream signaling. As shown in cell lines with T790M and L858R mutations the potential antitumor activity in EGFR mutated cancers was shown early on. [8-10] The immune effects of amivantamab are through binding of the Fc region to FcyRs on immune cells leading to ADCC and trogocytosis in which surface proteins from the tumor cell membrane are removed by immune effector cells, such as monocytes, macrophages, and neutrophils. “Amivantamab led to monocyte- and macrophage-dependent downmodulation of EGFR and MET through trogocytosis in cell culture and xenograft mouse models.” These immune effects are independent of the bispecific antibody binding to EGFR and MET with synergistic antitumor efficacy. Despite the harsh TME, amivantamab leads to broad spectrum tumor death due to its multiple mechanism of actions. Amivanatamb binds to MET with higher affinity (KD 40 picomolar/liter) but enables EGFR to become closer into proximity and bind to its target with weaker affinity (1.4 nanomolar/liter). This dual-arm binding leads to targeting of cancer cells with great potency while sparing healthy cells since these cells lack high levels of these receptors. According to Cho et al, “this precision target binding design of amivantamab intrinsically favors cancer-specific simultaneous binding of EGFR and MET, leading to concomitant inhibition of these interacting signaling pathways.” [11,12]
Amivantamab was evaluated in in EGFR exon 20 insertion-mutated NSCLC progressing on platinum chemotherapy in initial results from the CHRYSALIS phase I study. In the dose escalation dose expansion CHRYSALIS study, the antitumor activity was amivantamab was assessed for patients with EGFR exon 20 insertion. According to the authors, amivantamab is the first biologic to demonstrate efficacy in this patient population after progression on chemotherapy. [13] The recommended phase II dose was determined. As alluded to by the investigators that isolated amivantamab, by the agent’s binding to the EXC domain it can inhibit ligand binding, promote endocytosis of the receptor antibody complex as well as its degradation and induce Fc-dependent trogocytosis and ADCC. ORR and DLT were primary endpoints while DOR, CBR, PFS and OS served as secondary endpoints. Treatment continued until disease progression. Of 362 patients, 258 were treated the RP2D pf 1050 mg based on safety, PK and PD data. “The RP2D of 1,050 mg provided saturation of circulating serum EGFR and MET targets and coverage of the preclinically established target concentration of 168 mg/mL.” Rash occurred in 98 patients or 86%, pruritis in 19 or 17%, stomatitis in 24 or 21%, diarrhea in 14 or 12% and paronychia in 51 or 45%. Peripheral edema (21,18%) and hypoalbuminemia (31,27%) were associated with MET inhibition.
Grade 3 AEs were observed in 40 patients (35%), with most frequent being hypokalemia in six (5%) and rash, pulmonary embolism, diarrhea, and neutropenia in four (4%) each. 18 patients (16%) reported treatment-related grade 3 AEs most common included rash in four (4%) and IRR and neutropenia in three (3%) each. Pulmonary embolism and back pain were most frequently reported (3% each) serious AEs which occurred in 34 patients (30%). Single reports each of cellulitis, infected dermal cyst, ILD, pneumonitis, atrial flutter, rash, and toxic epidermal necrolysis were considered treatment related. 15 patients had treatment related dose reductions mainly due to rash (11,10%) with discontinuations in 5 patients (4%) due to rash and IRR and paronychia (2,2,1). 3 confirmed CRs and 29 PRs were observed with an ORR of 40% (95% CI, 29 to 51). ctDNA testing revealed 25 exon20ins variants out of 63 patients with detectable ctDNA, of which “antitumor responses were observed in patients who harbored insertions within the helical, near-loop, and far-loop regions of exon 20.” mPFS was 8.3 months (95% CI, 6.5 to 10.9) by BICR and investigator (95% CI, 5.5 to 10.6) assessments, while mOS was 22.8 months (95% CI, 14.6 to not reached). DP occurred in 47 patients. IRRs were managed by infusion interruption and reduction of infusion rate. Topical steroids and oral antibiotics are recommended for EGFR- inhibitors-mediated rash.As the authors stated, “[a]mivantamab exhibited a tolerable safety profile consistent with on-target inhibition of EGFR and MET pathways. We provide proof of concept that the EGFR can be effectively targeted through the extracellular domain for mutations that are resistant to EGFR tyrosine kinase inhibitors, including EGFR Exon20ins mutations, for which there are no approved therapies.” [14]
Amivantamab plus chemotherapy with and without lazertinib in EGFR-mutant advanced NSCLC after disease progression on Osimertinib were reported from primary results from the phase III MARIPOSA-2 study. In a phase I trial, amivantamab plus chemotherapy with or without Lazertinib showed antitumor activity in refractory EGFR mutated NSCLC. A follow up phase III trial, the MARIPOSA-2 study, was conducted with a total of 657 locally advanced or metastatic patients with either exon 19 deletions or L858R) who progressed on Osimertinib. [15,16] Combining amivantamab and Lazertinib (a highly selective, CNS penetrant third generation TKI) has been shown to provide synergistic benefit with demonstrated clinically meaningful activity on patients who have progressed on Osimertinib by targeting the extracellular and catalytic EGFR domains. [17] The dual endpoints were PFS of amivantamab-chemotherapy and amivantamab-lazertinib-chemotherapy versus chemotherapy randomized in a 2:2:1 ratio. Baseline characteristics including history of brain metastasis were balanced across the three arms. PFS was significantly longer for amivantamab-chemotherapy and amivantamab-lazertinib-chemotherapy versus chemotherapy [ (HR) for disease progression or death 0.48 and 0.44, respectively; for both; median of 6.3 (95% CI 5.6-8.4 months) and 8.3 (95% CI 6.8-9.1 months) versus 4.2 (95% CI 4.0-4.4 months) months, respectively]. PFS was significantly longer in the amivantamab-chemotherapy arm compared to the chemotherapy arm (HR for disease progression or death 0.48, 95% CI 0.36-0.64) and in the amivantamab-lazertinib-chemotherapy arm compared to the chemotherapy arm (HR for disease progression or death 0.44, 95% CI 0.35-0.56). PFS benefit was observed across all subgroups including history of brain metastases, Osimertinib therapy and EGFR mutation type. Objective response rate was significantly higher for amivantamab-chemotherapy and amivantamab-lazertinib-chemotherapy versus chemotherapy 64% (95% CI 55%-72%) and 63% (95% CI 57%-69%) versus 36% 95% CI 30%-42%) with significant improvements versus chemotherapy for amivantamab-chemotherapy (odds ratio 3.10, 95% CI 2.00-4.80) and amivantamab-lazertinib-chemotherapy (odds ratio 2.97, 95% CI 2.08-4.24) respectively. Median intracranial PFS was 12.5 and 12.8 versus 8.3 months for amivantamab-chemotherapy and amivantamab-lazertinib-chemotherapy versus chemotherapy (HR for intracranial disease progression or death 0.55 and 0.58, respectively). Median duration of response was 6.9 months (95% CI 5.5 months-not estimable) for confirmed responders for amivantamab-chemotherapy, 9.4 months (95% CI 6.9 months-not estimable) for amivantamab-lazertinib-chemotherapy, and 5.6 months (95% CI 4.2-9.6 months) for chemotherapy. Interim OS analysis revealed HRs for death of 0.77 (95%CI 0.49-1.21) for amivantamab-chemotherapy versus chemotherapy and 0.96 (95% CI 0.67-1.35) for amivantamab-lazertinib-chemotherapy versus chemotherapy. Grade 3 or higher AEs were hematologic toxicities and occurred in 72% of patients treated with Amivantamab-chemotherapy, in 92% with amivantamab-lazertinib-chemotherapy, and in 48% with chemotherapy, with the most common being neutropenia, thrombocytopenia, anemia, and leukopenia. Serious treatment-emergent AEs were reported in 32% treated with amivantamab-chemotherapy, 52% with amivantamab-lazertinib-chemotherapy, and 20% with chemotherapy, and included thrombocytopenia, neutropenia, and febrile neutropenia. IRRs occurred in 58% and 56% of patients on amivantamab-chemotherapy and amivantamab-lazertinib-chemotherapy, respectively. VTE was reported 10%, 22%, and 5% of patients in the amivantamab-chemotherapy, amivantamab-lazertinib-chemotherapy, and chemotherapy arms, respectively. The most common reason for treatment discontinuation was progressive disease: 41(32%), 68(26%), 152(63%) and hematologic toxicities. Treatment-related AEs leading to death were infrequent in all arms; there were two (2%) deaths in the amivantamab-chemotherapy arm, four (2%) in the amivantamab-lazertinib-chemotherapy arm, and one (0.4%) in the chemotherapy arm. “No clear pattern of specific AEs leading to death was detected.” As the authors conclude, “[a]mivantamab-chemotherapy and amivantamab-lazertinib-chemotherapy significantly improved PFS versus chemotherapy, with a 52% and 56% lower risk of disease progression or death, respectively. Early separation of curves was observed between both amivantamab-chemotherapy and amivantamab-lazertinib-chemotherapy versus chemotherapy. The PFS benefit was consistent across predefined subgroups. Mechanistically, amivantamab has shown efficacy against EGFR C797S, MET amplification, and other EGFR- and/or MET-based alterations after Osimertinib…Together, this combination provides broad coverage of the diverse and polyclonal tumor resistance arising after disease progression on Osimertinib…At this time, however, amivantamab-chemotherapy and amivantamab-lazertinib-chemotherapy are the first regimens to demonstrate efficacy over chemotherapy in the post-osimertinib setting and could represent a new standard of care.”[18]
While MARIPOSA-2 evaluated a heavily pre-treated population, Zhou et al investigated amivantamab versus chemotherapy in advanced NSCLC patients with no systemic treatment. [19-21] In this phase 3 randomized trial, 308 patients with exon 20 insertions were randomized 1:1 to receive amivantamab plus chemotherapy (n=153) versus chemotherapy alone (n=155). Primary endpoint was PFS and cross over was permitted. Progression-free survival was 11.4 months versus 6.7 months, being significantly longer (HR ratio for disease progression or death, 0.40; 95% confidence interval [CI], 0.30 to 0.53; P<0.001). At 18 months, PFS was 31% versus 3% with a CR and PR in 73% versus 47%. Treatment discontinuation was usually the result of PD which occurred in 33% versus 69% [50/151 versus 107/155]. Tumor volume decreased at rate of 53% versus 34%. Ongoing treatment response was 49% versus 17%. 70 deaths were reported at survival analysis with an increased risk of death for the chemotherapy group (HR 0.67, 95% CI 0.42-1.09). Neutropenia (59% versus 45%), paronychia (56% in the A-C group), rash (54%) in the A-C group and anemia (55%) and nausea (425) in the chemotherapy alone group. Grade 3 or higher events included neutropenia (33%0, leukopenia (11%) and rash (11%) in the AC group and neutropenia (23%), anemia (12%0 and thrombocytopenia (10%) in the chemotherapy group. Serious AEs occurred in 37% of the patients in the amivantamab-chemotherapy group and in 31% of those in the chemotherapy group. Death occurred in 28 patients (18%) in versus 42 patients (27%), with 20 and 30 deaths, respectively, caused by progressive disease. The administration of amivantamab-chemotherapy treatment significantly prolonged PFS as compared (HR for disease progression or death, 0.40; 95% CI, 0.30 to 0.53), with deeper more durable response and higher ORs. There was a clear and early separation of the PFS curves indicting improved longer follow up and rapid disease control. The 18-month PFS was 31% versus 3%. NGS was recommended to test accurately for EGFR exon 20 alterations at the time of metastatic diagnosis. As the authors conclude, “[t]he efficacy results of our trial further support recommendations to identify all oncogenic driver mutations to help select the appropriate first-line therapy before the initiation of chemotherapy-immunotherapy treatments, which do not improve clinical outcomes as compared with chemotherapy alone in patients with EGFR-mutated NSCLC. In our trial, we found that patients with previously untreated, advanced NSCLC with EGFR exon 20 insertions who received amivantamab- chemotherapy had significantly longer progression-free survival than those who received chemotherapy alone. Safety results were consistent with previous adverse-event reports with respect to the individual agents in each treatment. Our results indicate that amivantamab-chemotherapy was an effective first-line treatment in patients with advanced NSCLC with EGFR exon 20 insertions.” [22]
Overall survival with amivantamab-lazertinib in EGFR-mutated advanced NSCLC was reported in a recent study in NEJM wherein patients were randomized to amivantamab-lazertinib, Osimertinib and Lazertinib. [23-25] OS at 3 years was 60% for amivnatamab-lazertinib and 51% for Osimertinib, being significantly longer for amivantamab-lazertinib (median, could not be estimated and 36.7 months with a clinical benefit of more than 12 months. Disease progression was 42% (n=175) versus 60% (n=258) for amivantamab-lazertinib versus Osimertinib. IC PFS was 25.4 months versus 22.2 months (HR for IC progression or death 0.79). Paronychia and rash were related as due to EGFR inhibition. Venous thromboembolism occurred in 40% vs 11%. With a EGFR mutated patient population of locally advanced and metastatic NSCLC, amivantamab-lazertinib led to a significant OS advantage that treatment with Osimertinib [26] (Figure 1). A real world study analyzing databases for patients with EGFR mutant NSCLC receiving amivantamab not on a clinical trial showed that 19 or 45.2% of patients had clinical response and a DCR of 64.3%. 33 patients were in the classical mutation cohort that had a CR of 36.4% (12) and a DCR of 48.5%. 6/7 patients in the atypical mutation cohort had a CR and a DCR of 100%. (Atypical EGFR mutations included EGFR L792R, EGFR exon19 I740_K745dup, EGFR G719S, EGFR G719A, EGFR G719A, EGFR exon19 E746_S752delinsV, and EGFR exon19 E746_T751delinsVA). The exon 2 cohort had a CR and DCR 64.3% (5). The study revealed that amivantamab was an effective treatment option of patients with EGFR mutations outside of exon 20 insertion, particularly when combined with Osimertinib [27, 28].
Tebentafusp for the Treatment of Uveal Melanoma
Tebentafusp is a Bispecific protein treat metastatic uveal positive melanoma for adults who are positive for the HLA-A*02:01 allele that was first approved to treat uveal melanoma. Its MOA is by redirecting T cells to target and kill melanoma cells expressing the protein gp100, by recognizing CD3 on T cells to gp100. Classified as an ImmTAC molecule that combines a soluble T cell receptor domain fused to an effector activating domain in this case an anti-CD3 scFv. The anti-CD3 scFv domain activate sCD3+ T cells while the TCR domain recognizes peptides of interest presented by HLA complexes. Chosen epitopes should be expressed at high level by the tumor but minimally expressed elsewhere to minimize on-target, off tumor toxicity. ImmTACs target intracellular epitopes where the neoantigen pool is. And they are affinity enhanced to increase TCR binding to target. Three challenges for ImmTACs have to be stable, soluble TCR, enhancing ImmTAC affinity to tumor antigens and triggering an antitumor immune response [30-32] By engaging CD3 receptors on T cells the natural immune synapse is replicated, and by binding a specific HLA complex on tumor cell surface, the ImmTAC molecule can recruit CD3+ T cells leading to tumor cell lysis cell lysis regardless of TCR-directed specificity. The release of pro-inflammatory cytokines ad cytolytic agents are induced by ImmTACs activity leading to cell lysis. Epitope spreading further stimulates T-cell activation. Due to HLA restriction, patients must have the HLA-A*02:01 allele. Tebentafusp redirects T cells towards HLA-A*02:01 positive UM cells that present with melanoma associated antigen glycoprotein 100 peptide, a melanocyte specific molecule involved in maturation of melanosomes. PD analysis reveals “increased levels of pro-inflammatory-cytokines including CXCL10, CXCL11, interleukin (IL)-6, interferon gamma (IFN-γ), tumor necrosis factor alpha (TNFα) after treatment. Serum levels of IFN-γ, TNFα, IL-2 and Il-10 are seen to increase to up to 10-fold that of pre-treatment levels in the 8-24 h after drug administration… Tumor tissue studies show increases in CD3+, CD4+ and CD8+ lymphocyte infiltration and increased expression of the cell death marker cleaved caspase 3, suggesting clear evidence of an increase in tumor-infiltrating lymphocytes (TILs) following treatment, with associated tumor cell death.”[33,3]
Overall Survival Benefit with Tebentafusp in Metastatic Uveal Melanoma
One of the earliest studies evaluating tebentafusp was conducted by Nathan et al in an open-label phase 3 trial where HLA*02.01positive patients with metastatic uveal melanoma received tebentafusp versus investigator’s choice of therapy with pembrolizumab, ipilimumab or dacarbazine (control group) stratified according to LDH level. [34] Demographic and clinical characteristics of the two groups were well balanced at baseline. OS was 73% versus 59% HR 0.51 (95% CI 0.37 to 0.71) in favor of tebentafusp group. PFS was 31% versus 19%. Median DoR was 9.9 months versus 9.7 months. CRs PRs and SD at 12 weeks was higher in the tebentafusp group (48% 95% CI 39 to 52) versus 27% (95% CI 20 to 36). Among patients with disease progression as best response, median duration of OS was 15.3 months versus 6.5 months, independent of prognostic variables. OS benefit was seen in patients with no tumor shrinkage. HR for disease progression or death 0.73 (95% CI 0.58 to 0.94). Skin related AEs included rash (83%) pyrexia (76%) and pruritis (69%) as well as in other categories such as chills (47%) hypotension (38%) TRAEs of grade 3 or 4 were reported in 44% pf tebentafusp patients versus 17% in the control group; “No treatment related deaths were reported. In this randomized, phase 3 trial, treatment with tebentafusp, a soluble T-cell receptor and CD3-directed bispecific fusion protein, was associated with longer overall survival than the investigator’s choice of therapy in HLA-A*02:01- positive patients with metastatic uveal melanoma.” [35,36]
A Phase I study of safety, tolerability, and efficacy of tebentafusp using a step-up dosing regimen and expansion in patients with metastatic uveal melanoma showed that 68 ug was the RP2D and TEAEs included pyrexia (91%), rash (83%) and pruritis (83%) along with nausea (74%) and chills (69%). mOS was 25.5 months at a median follow up of 32.4 months with an ORR was 11.9%. The key objective of this study was to demonstrate tebentafusp’s promising activity with greater response observed at the MTD. A step up dosing regimen was developed for higher doses with escalation of the final dose using a standard 3 + 3 design. This study showed that the RP2D exceeded the MTD in the first in human trial and the 1-year survival of 67% and mOS 25.5 months suggested “clinically meaningful antitumor activity as monotherapy.” Using dosing regimens that was below the MTDs for the first two doses may reduce the occurrence of severe toxicity associated with bispecific antibodies. This study was conducted to optimize the dosing by increase in exposure and mitigate treatment toxicity. During the DE portion four dose levels were investigated and of these with three DLTs of hepatic events occurring in one at 64 ug, grade 3 or 4 transaminase elevations at 73 ug. All of these resolved in 1 week. An intermediate dose level 68ug was enrolled that had no DLTs or significant elevations of hepatic transaminases. Grade 3 TEAEs included abdominal pain, hypotension, and hypophosphatemia. SAEs occurred in 16 or 38% of patients which included AST and ALT increase abdominal pain (7%), AST increased (7%), ALT increased (5%), hypotension (5%), hyperbilirubinemia (5%), and hypophosphatemia (5%). “Increases in transaminases from baseline were observed in 9 (21%) patients, including four patients with grade 3 or 4 events; 38 or 90% of patients experienced CRS of any grade.” 5/42 patients had confirmed PR with an ORR of 11.9%. Tumor reduction was archived in 55% of patients. 24-week DCR was 33%. Serum markers such as IL-2, IL-6 IL-10 and CXCL10 and CXCL11 were also analyzed and existed temporally with treatment induced increase. On treatment biopsy samples had a greater presence of CD3+ T cells. [37,38]
A multi-center, open label single arm phase 2 trial conducted by Carvajal et al reported on the clinical and molecular response to tebentafusp in previously treated patients with metastatic uveal melanoma. ORR was the primary endpoint with secondary objective including safety OS, PFS and DCR. ORR was 5% the 1-year OS rate was 62% with mOS of 16.8 months. Dosings were initially 20ug on day 1, 30 ug on day 8 and 68ug on day 15 and then IV once weekly. Best overall response of stable disease at 24 weeks occurred after date of first dose. Duration of response from date of documented disease progression. Imaging endpoints were assessed by BICR. 96% of patients had hepatic involvement with patients having received at least one prior line of therapy in the metastatic setting with two thirds receiving prior ICI with resistance. Disease progression (70%) was the primary cause of treatment discontinuation. Rash (87%), pyrexia (80%), pruritic (67%) and chills (64%) were the primary adverse events. 40% had grade 3 events which were mostly rash, which was successfully managed with antihistamine and topical corticosteroids. 20% and 3% of patient had TRAEs resulting in hospitalization and drug discontinuation. 86% had CRS, mostly grade 1 or 2, with two leading to discontinuation. One grade 3 CRS event was concurrent with atrial fibrillation. CRS usually occurred during step up dosing regimen treated with antipyretics and systemic glucocorticoids. Median DOR was 8.7 months and 45% achieved SD. DCR was 32% at 16 weeks and 23% at 24 weeks. mPFS was 2.8 months. PFS rates were 25% at 6 months and 11% at 12 months. mOS was 16.8 months with a median duration of study follow up of 19.5 months. Longer survival was associated with tumor shrinkage. In subgroup analysis, OS was 51% at greater than 65 years; 45% with elevated LDH. “75%, 60% and 25% in patients with largest target liver metastasis at baseline of &3 cm (M1a), >3 cm to &8 cm (M1b) and >8 cm (M1c), respectively. In patients who had previously relapsed (best overall response of complete response, partial response or stable disease on prior therapy) following immunological checkpoint inhibition, 1 year overall survival was 76% (95% CI: 56-88%) compared with 60% (95% CI: 46-71%) in patients who were refractory (best overall response of progressive disease on prior therapy) to prior checkpoint inhibition… Following the first few doses, treatment-related adverse events tend to decrease in both frequency and severity, enabling the monitoring period to be shortened to 30-60 minutes…tebentafusp demonstrates a promising survival benefit for patients with metastatic disease that has progressed on at least one line of prior therapy.” [39-42]
Hassel reported on the three-year overall survival with tebentafusp in metastatic uveal melanoma. HLA-A*02:01 positive patients were randomly assigned 2:1 to receive either tebentafusp or investigator’ choice therapy with pembrolizumab, ipilumumab or dacarbazine. OS was the primary endpoint. mOS was 21.6 months at a minimum follow up of 36 moths versus 16.9 months (HR 0.68 95% CI 0.54 to 0.87). 27% versus 18% were surviving in 3 years, respectively. No new adverse events emerged during long term administration of the treatment, with low drug discontinuation rates. Baseline characteristics were balanced with both groups having high LDH levels, large metastatic lesion of greater than 3cm and 50% had extrahepatic disease. Patients in the tebentafusp group were treated beyond initial radiographic progression. 72%, 45%, and 27% versus 60%, 30%, and 18% were surviving at 1, 2, and 3 years. TMB was not associated with OS and was superior in the tebentafusp group regardless. mPFS was 3.4 months versus 2.9 months (stratified HR for progression or death 0.76,95% CI 0.60- 0.97. “In a 100-day landmark analysis involving the patients who had a best overall response of progressive disease by day 100 after randomization, postlandmark overall survival was longer in the tebentafusp group than in the control group. The hazard ratio for death in this analysis was 0.62 (95% CI, 0.44 to 0.89) in favor of tebentafusp”. Rash (83%), pyrexia (76%), pruritus
(70%), and hypotension (38%) were the most common adverse events. “Grade 3 or 4 treatment-related adverse events occurred in 116 patients (47%). The most common were rash (19%) and an elevation in the aspartate aminotransferase level (6%).” Most events occurred during the early parts of treatment (4 weeks) during step up dosing and then decreased in frequency and severity with subsequent doses. CRS occurred in 89% of patient during the early weeks of treatment, Most CRS was grade 1. “This updated analysis confirms that the overall survival benefit with tebentafusp, as compared with the investigator’s choice of single-agent pembrolizumab, ipilimumab, or dacarbazine, persisted after a follow-up of at least 3 years. An estimated 27% of the patients who were randomly assigned to receive tebentafusp were alive at the 3-year landmark, as compared with 18% of patients who were assigned to the control group… Our results show greater disease control and more durable responses in the tebentafusp group than in the control group, and the results for progression-free survival remained in favor of tebentafusp, with an estimated 8% of the patients in the tebentafusp group progression-free at 2 years, as compared with 3% of the patients in the control group.” The safety profile is cytokine mediated and skin related because of targeting gp100-expressing melanocytes. [43-46]
Overall survival from tebentafusp versus nivolumab plus ipilimumab in first-line metastatic uveal melanoma: a propensity score-weighted analysis was conducted comparing tebentafusp versus nivo + ipi. Propensity score methods have been widely used in epidemiological settings for treatment comparisons involving nonrandomized observational data that mimics the effect of randomization by creating balance between groups of patients with respect to baseline covariates and allows for more valid statistical comparisons. Access to patient level data is only needed. The authors compared OS on tebentafusp or pembrolizumab to N+I in untreated mUM using propensity scoring methods. Propensity score based inverse probability weighting (IPTW) balancing age sex LDH alkaline phosphatase and ECOG status and time from primary diagnosis to metastasis. OS was assessed using IPT-weighting Kaplan-Meier models. The primary IPTW including 240 patients randomized to tebentafusp from IMCgp100-202 and 45 N+I treated patients from GEM-1402.v. The IPTW OS favored tebentafusp HR 0.52 95% CU 0.35-0.78) The mOS and 1-year OS rate were 21.7 months and 73% for tebentafsup and 12.6 months and 50% for nivolumab and ipilimumab. The analysis revealed that tebentafus resulted in significantly superior OS compared with nivolumab and ipilimumab in patients with previously untreated mUM. N+I confers no distinct survival advantage, possibly due to low TMB and limited immunogenicity and low expression of PD-1 and PD-L1 and lack of TILs. Tebentafushp stimulates a strong polyclonal antitumor response even in absence of activated TILs [47].
Discussion
Preclinical studies elaborating on the mechanisms of action of these two bispecific antibodies have been reported. One study showed that the in vitro and in vivo antitumor activity of amivantamab was dependent on its dominant mechanism of action of trogocytosis. The downregulation of EGFR and MET was shown to be mediated by peripheral blood mononuclear cells or PBMCs and apoptotic activity require Fc binding to immune cells while inducing ADCC and ADCR. Monocyte composition as opposed to B and T cells and NK cells was the relevant factor. Trogocytosis is an Fc effector function that mediates transfer of cell surface proteins form tumor to effector cells. It was shown that amivantamab induced monocyte dependent trogocytosis leading to EFGR and cMET downmodulation [48].
Tebentafusp patients underwent ctDNA analysis in clinical studies which was found to be associated with overall survival and more sensitive indicator or response when compared to standard imaging. “The potential of ddPCR-based ctDNA monitoring as an economical, pragmatic and informative approach in MUM management, offering valuable insights into treatment response and prognosis. Discrepancies between radiographic response and OS was reported. Therefore, easily accessible, sensitive and specific methods allowing a better monitoring of individuals with metastatic UM (MUM) treated with tebentafusp in real-life are needed.” [42,45,49] cDNA levels at higher levels correlate with poor prognosis and tumor burden. Reductions in ctDNA are also associated with improved outcomes and prolonged OS and PFS, with linear relationships observed. In a phase 2 study, baseline ctDNA levels correlated with tumor burden and two-thirds of patients had some degree with ctDNA reduction that was associated with longer survival. Uveal melanoma genes were detected (GNAQ, GNA11, SF3B1, PLCB4, CYSLTR2) at a variant allelic frequency of >0.3 at baseline and were included in the analyses. baseline ctDNA levels were associated with overall survival. “The subset of patients with below-median levels of ctDNA had longer overall survival compared with the subset with above-median levels of ctDNA at baseline (HR 0.23, 95% CI: 0.13-0.4).” A PR, SD of 7 and PD of 3 was reported in twelve patients had complete ctDNA clearance (undetectable). A PR of 3, SD of 34 and PD 44 was reported in the patients without ctDNA clearance. By week nine improvement in OS was strongly associated with ctDNA reduction. “The 1 year overall survival rate in patients with ctDNA clearance (n = 12) was 100% compared with 52% in those with increased ctDNA (n = 27). These findings suggest that early reductions in ctDNA reflect tebentafusp-related activity in the tumor and may provide a more precise molecular predictor of clinical response to tebentafusp than traditional radiographic response criteria.” ctDNA as an early indicator of benefit and the addition of ctDNA analysis to assess molecular repose to treatment may provide a more sensitive means than imaging identify which patients will benefit from tebentafusp treatment.[42] A similar analysis on the benefit of ctDNA analysis was conducted by Hassel et al). Undetectable ctDNA at baseline were more likely to have smaller lesions and normal LDH levels. 45 or 37% of patients had ctDNA clearance and baseline ctDNA levels were prognostic with undetectable levels correlated with longer OS than detectable ctDNA. OS among 99 patients who had a reduction of at least 50% was longer than the 24 patents who had a reduction of less than 50%, no change, or an increase in the ctDNA level (hazard ratio for death, 0.41; 95% CI, 0.25 to 0.67) [45].
In a study by Rodrigues et al, ddPCR was used for ctDNA analysis on 60 mUM patients with tebentafusp treatment. 61% had detectable ctDNA prior to treatment which was associated with shorter OS (median 12.9 months versus 40.5 months for patients with undetectable ctDNA; p < 0.001). At 12 weeks patients displaying a 90% or greater reduction in levels of ctDNA had a significant increase in (median 21.2 months versus 12.9 months; p=0.02). Clonal, hotspot mutations, predominantly involving GNAQ, GNA11, SF3B1 or PLCB4 and CYSLTR2 to a lesser extent were consistently present. ctDNA positivity was associated with serum LDH levels, best tumor response and diameter of largest metastasis. 97% of patients presented with at least one somatic mutation (67; in GNAQ (n =37), GNA11 (n =29) or SF3B1 (n = 1)), for which 64 were evaluable for plasma sample analysis. 39 of these 64 had ctDNA detected with median of 31 copies/mL of plasma (IQR = 9-557 copies/mL of plasma, which were positively correlated with serum LDH level and diameter or largest metastasis due to its association with tumor burden Paired plasma samples between baseline and 3 weeks 22(36%) showed no ctDNA detection and 36 with baseline ctDNA presence demonstrated reduced ctDNA levels at 3 weeks, with 8 achieving complete ctDNA clearance. At 12 weeks during first tumor assessment, 21/56 patients detectable ctDNA (median timing for ctDNA sampling = 12.0 weeks [IQR: 11.9-12.6]). “Patients with detectable ctDNA before treatment displayed significantly shorter PFS (median 2.5 months versus 10.8 months; HR = 2.8, 95%CI [1.5-5.2]) and OS (median 12.9 months versus 40.5 months; HR = 10.1, 95% CI [3.0-33.8]. Furthermore, ctDNA detection at 3w and 12w were correlated also with poorer prognoses in both PFS
(p < 0.001 for both 3w and 12w) and OS…More precisely, among the 11 patients achieving complete ctDNA clearance at 12w, five (45%) had SD and six had PD.” Regarding survival outcomes, ctDNA-positive patients at baseline who experienced complete clearance at 12 weeks (n = 11) had a significantly longer median OS (12.7months versus 34.6 months; HR = 7.1, 95%CI [1.6-31.4]) and PFS (2.6 months versus 17.1 months; HR = 2.9, 95%CI [1.1-8.0) compared to those without clearance (n =21). Similar trends were observed with different thresholds.
“Patients achieving a 90% ctDNA decrease at 12w (n = 13) versus those who did not (n = 19) had a longer median OS (12.9 months versus 21.2months; HR = 3.4, 95%CI [1.1-10.4]) and longer median PFS (2.6 months versus 10.6 months; HR = 2.6, 95%CI [1.1-6.5]). Similar to high LDH levels, detection of ctDNA at baseline was associated with a poor median OS, emphasizing its potential as a prognostic marker. ctDNA levels were positively associated with tumor burden as appraised by TNM in most cases. The role of tebentafusp in patients with MUM as shown by this study advocates for the use of ddPCR-based ctDNA monitoring as a practical, cost-effective approach directly applicable in routine. These findings offer a potential avenue for treatment deescalation (limited course of tebentafusp instead of pursuing it until progression) or intensification (combination studies) based on ctDNA dynamics, paving the way for personalized therapeutic strategies in the management of UM.” [49]
Guc et al explored the tumor microenvironment of uveal melanoma treatment with tebantafusp in the specific context of TAM macrophages, where they identified ImmTAC-mediated M2 to M1 reprogramming of macrophages in vitro. They found that tumor cell elimination by ImmTAC-directed T cells was significantly reduced in the presence of M2 macrophages, wherein this suppression of T cell activation and tumor killing took place in direct contact. This situation was not reversed by checkpoint inhibition since simultaneous blockade of PD-1, LAG-3 and CTLA did not restore IFN-gamma secretion but partially restored ImmTAC-mediated killing. Neutralize TGF beta and Il-10 also did not reverse macrophage suppression of Imm-TAC-redirected T cell activation and tumor killing. Significantly they found that pretreatment of T cells with IL-2 enhances ImmTAC-redirected tumor killing and overcomes tumor macrophage suppression. Pre treatment with IL-2 at 4 days was superior to simultaneous administration. Tumor killing was up to 2 fold higher. “Co-culture with M2 macrophages caused a drastic reprogramming of gene expression in ImmTAC-redirected T cells, significantly upregulating 541 genes and downregulating 314 genes compared to ImmTAC-redirected T cells in the absence of M2 macrophages. In contrast, M2 macrophages had minimal effects on ImmTAC-redirected IL-2 pre-treated T cells as only 30 genes were differentially regulated (14 upregulated, 16 downregulated). High ratio of CD163+TAM in tumor biopsies was associated with short OS on tebentafusp treatment.”
“However, patients with high CD163+ TAM-to-CD3+ T cell ratios (ratio of CD163:CD3 > 2) at baseline had shorter OS compared with those with a low CD163+ TAM-to-CD3+ T cell ratio prior to treatment (HR = 2.09; 95% CI, 1.31-3.33). Tebentafusp treatment induces upregulation of anti-tumoral macrophage gene signatures in patients with mUM. these results demonstrate the effect of tebentafusp on macrophage reprogramming from a pro- to an anti-tumoral phenotype as early as day 16, after 3 doses of tebentafusp, and up to 18months on treatment. Gene expression of inflammatory markers (IL-1B, IFN-γ-inducible chemokines CXCL10, CXCL11, and IFN-γ-induced IRF1 and IFITM1) was significantly upregulated in tumor biopsies collected after 3 doses of tebentafusp treatment (1.3-2.5-fold-change). Conversely, the M2 macrophage-associated genes CA2 and ARG145,46 were significantly downregulated (0.9-1.3 fold-change) in post tebentafusp treatment biopsies.” [50]
Conclusion and Future Directions
This review highlights a specific indication of bispecific T cell engagers for the treatment of solid tumors in the metastatic and resistant settings. Both amivantamab and tebentafusp have efficacy for NSCLC and UM, respectively. Future directions may lie in combination therapies with other classes of cancer immunotherapies such as immune checkpoint inhibitors and antibody drug conjugates, or for other specific indications such as other solid tumors including colorectal cancer, breast cancer and prostate cancer.
Funding
None to Declare
Competing Interests
None to Declare
Ethics Declaration and Consent to Participate: not applicable
Datasets: not applicable
P.H. designed, wrote and edited this manuscript in its entirety.
References
- Shah V, McNatty A, Simpson L, et al. Amivantamab-Vmjw: A Novel Treatment for Patients with NSCLC Harboring EGFR Exon 20 Insertion Mutation after Progression on Platinum-Based Chemotherapy. Biomedicines 11 (2023).
- Brazel D, Nagasaka M. The development of amivantamab for the treatment of non-small cell lung cancer. Respir Res 24 (2023): 256.
- Howlett S, Carter TJ, Shaw HM, Nathan PD. Tebentafusp: a first-in-class treatment for metastatic uveal melanoma. Ther Adv Med Oncol 15 (2023): 17588359231160140.
- Melosky B, Wheatley-Price P, Juergens RA, et al. The rapidly evolving landscape of novel targeted therapies in advanced non-small cell lung cancer. Lung Cancer 160 (2021): 136-151.
- Huang S, van Duijnhoven SMJ, Sijts A, et al. Bispecific antibodies targeting dual tumor-associated antigens in cancer therapy. J Cancer Res Clin Oncol 146 (2020): 3111-3122.
- Meador CB, Sequist LV, Piotrowska Z, et al. Targeting EGFR Exon 20 insertions in non-small cell lung cancer: Recent advances and clinical updates. Cancer Discov 11 (2021): 2145-2157.
- Neijssen J, Cardoso RMF, Chevalier KM, et al. Discovery of amivantamab (JNJ-61186372), a bispecific antibody targeting EGFR and MET. J Biol Chem 296 (2021): 100641.
- Yun J, Lee SH, Kim SY, et al. Antitumor activity of amivantamab-vmjw (JNJ-61186372), an EGFR-MET bispecific antibody, in diverse models of EGFR exon 20 insertion-driven NSCLC. Cancer Discov 10 (2020): 1194-1209.
- Eck MJ, Yun CH. Structural and mechanistic underpinnings of the differential drug sensitivity of EGFR mutations in non-small cell lung cancer. Biochim Biophys Acta 1804 (2010): 559-566.
- Yasuda H, Kobayashi S, Costa DB. EGFR exon 20 insertion mutations in non-small-cell lung cancer: preclinical data and clinical implications. Lancet Oncol 13 (2012): e23-e31.
- Cho BC, Simi A, Sabari J, et al. Amivantamab, an Epidermal Growth Factor Receptor (EGFR) and Mesenchymal-epithelial Transition Factor (MET) Bispecific Antibody, Designed to Enable Multiple Mechanisms of Action and Broad Clinical Applications. Clin Lung Cancer 24 (2023): 89-97.
- Liu X, Yao W, Newton RC, et al. Targeting the c-MET signaling pathway for cancer therapy. Expert Opin Investig Drugs 17 (2008): 997-1011.
- Yu HA, Arcila ME, Rekhtman N, et al. Analysis of tumor specimens at the time of acquired resistance to EGFR-TKI therapy in 155 patients with EGFR-mutant lung cancers. Clin Cancer Res 19 (2013): 2240-2247.
- Park K, Haura EB, Leighl NB, et al. Amivantamab in EGFR Exon 20 Insertion-Mutated Non-Small-Cell Lung Cancer Progressing on Platinum Chemotherapy: Initial Results From the CHRYSALIS Phase I Study. J Clin Oncol 39 (2021): 3391-3402.
- Surati M, Patel P, Peterson A, et al. Role of MetMAb (OA-5D5) in c-MET active lung malignancies. Expert Opin Biol Ther 11 (2011): 1655-1662.
- Zhang YL, Yuan JQ, Wang KF, et al. The prevalence of EGFR mutation in patients with non-small cell lung cancer: a systematic review and meta-analysis. Oncotarget 7 (2016): 78985-78993.
- Westover D, Zugazagoitia J, Cho BC, et al. Mechanisms of acquired resistance to first- and second-generation EGFR tyrosine kinase inhibitors. Ann Oncol 29 (2018): i10-i19.
- Passaro A, Wang J, Wang Y, et al. Amivantamab plus chemotherapy with and without lazertinib in EGFR-mutant advanced NSCLC after disease progression on osimertinib: primary results from the phase III MARIPOSA-2 study. Ann Oncol 35 (2024): 77-90.
- Shu CA, Goto K, Ohe Y, et al. Amivantamab and lazertinib in patients with EGFR-mutant non-small cell lung cancer after progression on osimertinib and platinum-based chemotherapy: Updated results from CHRYSALIS-2. J Clin Oncol 40 (2022): 9006.
- Wu JY, Yu CJ, Shih JY. Effectiveness of treatments for advanced non-small-cell lung cancer with exon 20 insertion epidermal growth factor receptor mutations. Clin Lung Cancer 20 (2019): e620-e630.
- Naidoo J, Sima CS, Rodriguez K, et al. Epidermal growth factor receptor exon 20 insertions in advanced lung adenocarcinomas: Clinical outcomes and response to erlotinib. Cancer 121 (2015): 3212-3220.
- Zhou C, Tang KJ, Cho BC, et al. Amivantamab plus Chemotherapy in NSCLC with EGFR Exon 20 Insertions. N Engl J Med 389 (2023): 2039-2051.
- Yun J, Lee SH, Kim SY, et al. Antitumor activity of amivantamab (JNJ-61186372), an EGFR-MET bispecific antibody, in diverse models of EGFR exon 20 insertion-driven NSCLC. Cancer Discov 10 (2020): 1194-1209.
- Mok TSK, Nakagawa K, Park K, et al. Nivolumab plus chemotherapy versus chemotherapy in patients with EGFR-mutated metastatic non-small cell lung cancer with disease progression after EGFR tyrosine kinase inhibitors in CheckMate 722. Ann Oncol 33 (2022): 1560-1597.
- Moores SL, Chiu ML, Bushey BS, et al. A novel bispecific antibody targeting EGFR and cMet is effective against EGFR inhibitor-resistant lung tumors. Cancer Res 76 (2016): 3942-3953.
- Yang JC, Lu S, Hayashi H, et al. Overall Survival with Amivantamab-Lazertinib in EGFR-Mutated Advanced NSCLC. N Engl J Med 393 (2025): 1681-1693.
- Yun J, Lee SH, Kim SY, et al. Antitumor activity of amivantamab (JNJ-61186372), an EGFR-MET bispecific antibody, in diverse models of EGFR exon 20 insertion-driven NSCLC. Cancer Discov 10 (2020): 1194-1209.
- Wang K, Du R, Myall NJ, et al. Real-World Efficacy and Safety of Amivantamab for EGFR-Mutant NSCLC. J Thorac Oncol 19 (2024): 500-506.
- Petrini I, Giaccone G. Amivantamab in the Treatment of Metastatic NSCLC: Patient Selection and Special Considerations. Onco Targets Ther 15 (2022): 1197-1210.
- Buder K, Gesierich A, Gelbrich G, et al. Systemic treatment of metastatic uveal melanoma: Review of literature and future perspectives. Cancer Med 2 (2013): 674-686.
- Carvajal RD, Schwartz GK, Tezel T, et al. Metastatic disease from uveal melanoma: treatment options and future prospects. Br J Ophthalmol 101 (2017): 38-44.
- Damato BE, Dukes J, Goodall H, et al. Tebentafusp: T Cell Redirection for the Treatment of Metastatic Uveal Melanoma. Cancers (Basel) 11 (2019).
- Middleton MR, McAlpine C, Woodcock VK, et al. Tebentafusp, a TCR/Anti-CD3 bispecific fusion protein targeting gp100, potently activated antitumor immune responses in patients with metastatic melanoma. Clin Cancer Res 26 (2020): 5869-5878.
- Sacco JJ, Carvajal R, Butler MO, et al. A phase II, multi-center study of the safety and efficacy of tebentafusp (IMCgp100) in patients with metastatic uveal melanoma. Ann Oncol 31 (2020): S1442-S1443.
- Nathan P, Hassel JC, Rutkowski P, et al. Overall Survival Benefit with Tebentafusp in Metastatic Uveal Melanoma. N Engl J Med 385 (2021): 1196-1206.
- Carvajal RD, Sato T, Shoushtari AN, et al. Safety, efficacy and biology of the gp100 TCR-based bispecific T cell redirector, IMCgp100 in advanced uveal melanoma in two phase 1 trials. J Immunother Cancer 5 (2017): P208.
- Hassel JC, Rutkowski P, Baurain JF, et al. Co-primary endpoint of overall survival for tebentafusp-induced rash in a phase 3 randomized trial comparing tebentafusp versus investigator’s choice in first-line metastatic uveal melanoma. J Clin Oncol 39 (2021): 9527.
- Carvajal RD, Sacco JJ, Jager MJ, et al. Phase I Study of Safety, Tolerability, and Efficacy of Tebentafusp Using a Step-Up Dosing Regimen and Expansion in Patients With Metastatic Uveal Melanoma. J Clin Oncol 40 (2022): 1939-1948.
- Bossi G, Buisson S, Oates J, et al. ImmTAC-redirected tumour cell killing induces and potentiates antigen cross-presentation by dendritic cells. Cancer Immunol Immunother 63 (2014): 437-448.
- Javed A, et al. PD-L1 expression in tumor metastasis is different between uveal melanoma and cutaneous melanoma. Immunotherapy 9 (2017): 1323-1330.
- Rantala ES, Hernberg M, Kivelä TT. Overall survival after treatment for metastatic uveal melanoma: a systematic review and meta-analysis. Melanoma Res 29 (2019): 561-568.
- Carvajal RD, Butler MO, Shoushtari AN, et al. Clinical and molecular response to tebentafusp in previously treated patients with metastatic uveal melanoma: a phase 2 trial. Nat Med 28 (2022): 2364-2373.
- Khoja L, Atenafu EG, Suciu S, et al. Meta-analysis in metastatic uveal melanoma to determine progression free and overall survival benchmarks: an international rare cancers initiative (IRCI) ocular melanoma study. Ann Oncol 30 (2019): 1370-1380.
- Rantala ES, Hernberg M, Kivelä TT. Overall survival after treatment for metastatic uveal melanoma: a systematic review and meta-analysis. Melanoma Res 29 (2019): 561-568.
- Hassel JC, Piperno-Neumann S, Rutkowski P, et al. Three-Year Overall Survival with Tebentafusp in Metastatic Uveal Melanoma. N Engl J Med 389 (2023): 2256-2266.
- Carvajal RD, Sacco JJ, Jager MJ, et al. Advances in the clinical management of uveal melanoma. Nat Rev Clin Oncol 20 (2023): 99-115.
- Piulats JM, Watkins C, Costa-Garcia M, et al. Overall survival from tebentafusp versus nivolumab plus ipilimumab in first-line metastatic uveal melanoma: a propensity score-weighted analysis. Ann Oncol 35 (2024): 317-326.
- Vijayaraghavan S, Lipfert L, Chevalier K, et al. Amivantamab (JNJ-61186372), an Fc Enhanced EGFR/cMet Bispecific Antibody, Induces Receptor Downmodulation and Antitumor Activity by Monocyte/Macrophage Trogocytosis. Mol Cancer Ther 19 (2020): 2044-2056.
- Rodrigues M, Ramtohul T, Rampanou A, et al. Prospective assessment of circulating tumor DNA in patients with metastatic uveal melanoma treated with tebentafusp. Nat Commun 15 (2024): 8851.
- Guc E, Treveil A, Leach E, et al. Tebentafusp, a T cell engager, promotes macrophage reprogramming and in combination with IL-2 overcomes macrophage immunosuppression in cancer. Nat Commun 16 (2025): 2374.


 Impact Factor:
* 3.5
Impact Factor:
* 3.5
 Acceptance Rate:
71.36%
Acceptance Rate:
71.36%
 Time to first decision: 10.4 days
Time to first decision: 10.4 days
 Time from article received to acceptance:
2-3 weeks
Time from article received to acceptance:
2-3 weeks

