Correlation Between MRI and Ultrasound for the Diagnosis of Lower Limb Bone Stress Injuries
Ryan C. Kruse MD1*, Elena Volfson BA2, Brennan J. Boettcher DO3, Shelby E. Johnson MD4, Jennifer Ferden MS, ATC5, Krit Petrachaianan MS6
1Assistant Professor, Department of Orthopedics and Rehabilitation, University of Iowa Sports Medicine, Iowa City, IA, USA
2Medical Student, Carver College of Medicine, University of Iowa, Iowa City, IA, USA
3Assistant Professor, Departments of Physical Medicine and Rehabilitation and Orthopedic Surgery, Division of Sports Medicine, Mayo Clinic, Rochester, MN, USA
4Assistant Professor, Departments of Physical Medicine and Rehabilitation and Orthopedic Surgery, Division of Sports Medicine, Mayo Clinic, Minneapolis, MN, USA
5Athletic Trainer, Department of Orthopedics and Rehabilitation, University of Iowa Sports Medicine, Iowa City, IA, USA
6Research Scientist, Department of Orthopedics and Rehabilitation, University of Iowa, Iowa City, IA, USA
*Corresponding Author: Ryan C. Kruse, Assistant Professor, Department of Orthopedics and Rehabilitation, University of Iowa Sports Medicine, Iowa City, IA, USA.
Received: 29 March 2026; Accepted: 06 April 2026; Published: 13 April 2026
Article Information
Citation: Ryan C. Kruse, Elena Volfson, Brennan J. Boettcher, Shelby E. Johnson, Jennifer Ferden, Krit Petrachaianan. Correlation Between MRI and Ultrasound for the Diagnosis of Lower Limb Bone Stress Injuries. Journal of Orthopedics and Sports Medicine. 8 (2026): 148-153.
View / Download Pdf Share at FacebookAbstract
Bone stress injuries (BSIs) are a common lower limb injury amongst active individuals. While magnetic resonance imaging (MRI) has historically been the imaging modality of choice, there is increasing evidence that ultrasound (US) can be a useful diagnostic tool for these injuries. The aim of this prospective, observational study was to evaluate the correlation between MRI and US findings in the diagnosis of lower extremity BSIs and to characterize the sonographic features 25 patients age 19-62 years old diagnosed with an MRI-confirmed BSI to the metatarsals or tibia underwent a diagnostic US of the affected area as well as the correlative, asymptomatic region on the contralateral limb. The primary outcome was whether or not an association existed between sonographic findings and the MRI grade of the stress injuries. Median age was 35 years, and 84% were females. The most common injury grade was a grade 1 (40%). On US evaluation, periosteal edema was most commonly seen (80% of patients). Subcutaneous hyperemia and callous formation demonstrated a significant correlation with grade 4 BSIs (p=0.03). US and MRI demonstrated variable agreement on imaging findings, with as high as 100% agreement on the presence of a cortical break and callous formation, and as low as 64% for periosteal thickening. In conclusion, this study demonstrated a relatively strong agreement between US and MRI findings seen with a BSI. However, US did not consistently predict injury grade. Therefore, our findings suggest that US may be considered as a screening tool for tibial and metatarsal BSIs.
Keywords
Lower Limb; Bone, Injury, MRI, Bone stress injuries (BSIs); Skeletal; Healthcare
Article Details
1. Introduction
Bone stress injuries (BSIs) are common injuries among physically active individuals and can result in prolonged time away from sport [1-3]. Although BSIs can occur in various skeletal regions, the lower extremities are disproportionately affected, comprising up to 95% of cases [4]. This distribution is largely attributed to the repetitive mechanical loading associated with weight-bearing activity. A timely and accurate diagnosis is critical to optimize patient outcomes, reduce risk of complications such as a completed fracture, and minimize overall healthcare burden [5].
The pathogenesis of BSIs is multifactorial due to the interplay between mechanical loading and intrinsic biological factors. An abrupt increase in training intensity, frequency, or duration without sufficient recovery can disrupt the equilibrium between bone resorption and formation. This results in accumulation of microdamage and can result in stress fracture [6]. Given the functional impact of these injuries on active individuals, reliable and accessible diagnostic modalities are essential [6,7].
Current diagnostic evaluation of BSIs begins with a comprehensive clinical assessment, including history, risk factor analysis, and focused physical examination. Localized, mechanically induced pain remains a hallmark finding [8]. Radiographs are typically obtained first. However, the sensitivity of radiographs is only 12-56% [4]. Additional imaging is often obtained if clinical suspicion for a BSI is high despite normal radiographs. Magnetic resonance imaging (MRI), which is often obtained to confirm clinical suspicion, offers a high sensitivity and specificity but can be limited by cost and accessibility [9]. Bone scintigraphy, while sensitive, lacks specificity and is more invasive and exposes the patient to radiation [9]. These limitations have prompted growing interest in alternative imaging techniques that are more accessible, cost-effective, and non-invasive [10].
Preliminary evidence suggests that ultrasound (US) may serve as a useful adjunct in the early detection of BSIs [11,12]. Readily available, low-cost, and non-invasive, US has the potential to supplement or even partially replace traditional imaging in certain clinical settings [13]. However, there remains inconclusivity regarding the role of US in the diagnosis of BSIs. A recent Dephi consensus paper by Hoenig et al showed high variability amongst expert opinions on the use of US for the diagnosis of BSIs, and concluded that MRI remains the gold standard amongst their group [14]. In this study, we aim to address the lack of consensus in the literature by evaluating the correlation between MRI and US findings in the diagnosis of lower extremity BSIs and to characterize the sonographic features associated with these injuries. Through this investigation, we seek to help define the role of US in the diagnostic algorithm for BSIs.
2. Methods and materials
This prospective observational study was performed at our sports medicine clinic after approval by our local institutional review board. Informed consent was obtained for all included patients. Inclusion criteria included patients age 18-80 years old diagnosed with an MRI-confirmed BSI to the metatarsals or tibia within 14 days prior to presenting to the senior author’s sports medicine clinic. Patients were excluded if they had a prior history of a BSI to that specific bone. Full inclusion and exclusion are outlined in Table 1.
|
Inclusion Criteria |
Exclusion Criteria |
|
Age 18-80 |
Age outside of inclusion range |
|
MRI-confirmed stress injury of tibia or metatarsal |
History of stress injury in the affected bone |
|
History of fracture in the affected bone |
|
|
Concurrent stress injury in the contralateral bone |
|
|
Inability to undergo diagnostic US within 14 days of MRI |
MRI = magnetic resonance imaging; US = ultrasound
Table 1: Inclusion and Exclusion Criteria.
Patients were referred by physicians within our sports medicine clinic as well as local primary care physicians. Patients who were referred for possible inclusion in the study had their charts reviewed by a study team member other than the primary author (RCK) to confirm the presence of an MRI-confirmed BSI. This was done to keep the primary author blinded to the location and grade of the BSI. After confirmation of the BSI, the patient was seen in clinic within 14 days of the MRI for a diagnostic US of the affected region. This was performed by the primary author who was blinded to the grade and location of the BSI. The patient was asked to point to their region of maximal pain to help guide the US evaluation.
US evaluation was performed with a 4-18MHz, linear array transducer (Samsung RS85 Prestige Ultrasound System, Samsung Co., San Jose, CA). The region of maximal pain, as well as the correlative region on the asymptomatic contralateral limb, were evaluated in both a longitudinal and transverse plane. The absence or presence of following sonographic findings were recorded: periosteal edema, periosteal thickening, periosteal hyperemia, subcutaneous edema, subcutaneous hyperemia, cortical break, and callous formation (Figure 1) [15]. The primary author performed all of the US evaluations and recorded the US findings. Deidentified US images for each patient were sent to a second author to review and record their findings. The second author was blinded to the assessments of the first author. If there were discrepancies between the two authors (e.g. disagreement on the presence of subcutaneous edema), those specific images were then sent to a third author for review. The review by the third author ultimately decided the findings for those images. All authors who reviewed the US images had between 4-7 years of independently performing musculoskeletal US examinations, and were also Registered is Musculoskeletal sonography (RMSK) certified. Of note, there was agreement between the first two reviewing authors on 88% of the ultrasound findings, with the highest degree of agreement being on the presence of callous formation (100%) and the least agreement on the presence of periosteal edema (76%).
The primary outcome was whether or not an association existed between sonographic findings and the MRI grade of the stress injuries. Secondary outcomes included whether or not the US findings were consistent with those seen on MRI.
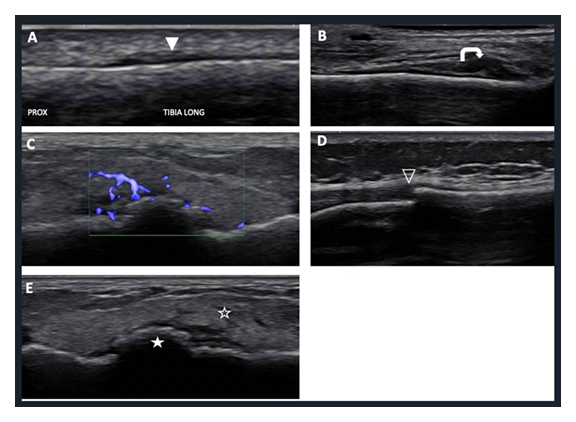
Figure 1: Common sonographic findings seen with a BSI. A. Periosteal thickening, seen as a focal, eccentric increase in the hypoechoic periosteal thickness compared to the adjacent periosteum (white arrow). B. Periosteal edema, seen as asymmetric hypoechogenicity adjacent to the periosteum extending into the subcutaneous tissue (white curved arrow). C. Periosteal hyperemia. D. Cortical break (open arrowhead). E. Callous formation (closed star) and subcutaneous edema (open star), which is seen as relative hyperechogenicity of the subcutaneous tissue, often described as having a “cobblestone” appearance.
DIST = distal; PROX = proximal; LONG = longitudinal
2.1 Data analysis
Patients were grouped according to injury grade (Fredericson classification [16]) and compared with respect to demographics, BSI history, and REDs history to ensure comparability. Kruskal-Wallis rank sum tests were used to compare continuous variables, and Fisher’s exact tests were used for categorical variables.
Findings from MRI and US were cross tabulated with injury grade to assess associations. Fisher's exact test was used to determine statistical significance. Stuart-Kendall’s tau-c was used to measure the strength of each relationship, and each associated p-value was used to determine statistical significance.
Agreement between US and MRI findings was quantified using both percent agreement and Cohen’s kappa.
All analyses were performed using R Statistical Software (v4.1.0; R Core Team 2021).
3. Results
Baseline demographics are outlined in Table 2. A total of 25 patients were included. 16 patients had a BSI involving the tibia, and 9 had a BSI involving the metatarsals. 84 percent were female, with a median age of 35 years (range 19-62). 32 percent had a history of a BSI outside of the anatomic region being evaluated for the study. No patients had a history of BSI in the affected region. The median number of days between symptom onset and MRI was 42 days (range 6-295). There were no significant differences in baseline variables across injury grades with the exception of history of REDs, where only two patients, both with grade 3 stress injuries, had REDs history in our sample.
|
Overall (n=25) |
Grade 1 (n=10) |
Grade 2 (n=6) |
Grade 3 (n=3) |
Grade 4a (n=3) |
Grade 4b (n=3) |
|
|
Age Median Range |
35 19-62 |
27 20-41 |
37 23-62 |
24 19-41 |
41 22-42 |
46 42-47 |
|
Sex Female Male |
21 (84%) 4 (16%) |
9 (90%) 1 (10%) |
5 (83%) 1 (17%) |
3 (100%) 0 (0%) |
2 (67%) 1 (33%) |
2 (67%) 1 (33%) |
|
Height (in) Median Range |
64 61-72 |
65 62-72 |
64 61-68 |
62 61-66 |
64 63-66 |
67 64-70 |
|
Weight (lbs) Median Range |
156 81-262 |
158 134-190 |
180 101-262 |
138 118-150 |
134 118-172 |
178 81-179 |
|
BMI Median Range |
26.1 18.0-38.8 |
25.3 20.8-33.1 |
29.8 18.0-38.8 |
21.6 21.6-28.4 |
22.6 20.7-26.9 |
27.7 25.7-29.6 |
|
Prior stress injury anywhere in body |
8 (32%) |
4 (40%) |
3 (50%) |
1 (33%) |
0 (0%) |
0 (0%) |
|
Prior stress injury at affected site |
0 (0%) |
0 (0%) |
0 (0%) |
0 (0%) |
0 (0%) |
0 (0%) |
|
Prior or current diagnosis of energy deficiency (REDs1) |
2 (8.0%) |
0 (0%) |
0 (0%) |
2 (67%) |
0 (0%) |
0 (0%) |
|
Days between symptom onset and MRI Median Range |
42 6-295 |
62 14-295 |
26 6-82 |
73 42-104 |
47 35-70 |
27 18-71 |
1Relative Energy Deficiency in Sports
Table 2: Baseline demographics.
MRI findings and BSI grades can be seen in Table 3. The most common injury grade was grade 1 (40%) followed by grade 2 (24%). On MRI, one-hundred percent of BSI’s demonstrated periosteal edema, which was also the most common finding for grade 1 injuries. Periosteal thickening was seen in all injury grades, but not in all patients within that injury grade. For example, only 50% of grade 2 stress injuries showed periosteal thickening compared to 100% of grade 4a injuries. Cortical breaks and callous formation were only seen in grade 4b injuries; 100% of grade 4b stress injuries showed cortical breaks and 67% showed callous formation.
|
Characteristic |
Overall (n=25) |
Grade 1 (n=10) |
Grade 2 (n=6) |
Grade 3 (n=3) |
Grade 4a (n=3) |
Grade 4b (n=3) |
p-value |
Gamma |
|
Periosteal edema |
25 (100%) |
10 (100%) |
6 (100%) |
3 (100%) |
3 (100% |
3 (100%) |
||
|
Periosteal thickening |
17 (68%) |
5 (50%) |
6 (100%) |
3 (100%) |
3 (100%) |
3 (100%) |
0.2 |
0.706 |
|
Subcutaneous edema |
8 (32%) |
0 (0%) |
0 (0%) |
2 (67%) |
3 (100%) |
3 (100%) |
<0.001 |
1 |
|
Cortical break |
3 (12%) |
0 (0%) |
0 (0%) |
0 (0%) |
0 (0%) |
3 (100%) |
<0.001 |
1 |
|
Callous present |
2 (8.0%) |
0 (0%) |
0 (0%) |
0 (0%) |
0 (0%) |
2 (67%) |
0.03 |
1 |
|
Bone marrow edema |
14 (56%) |
0 (0%) |
6 (100%) |
3 (100%) |
3 (100%) |
3 (100%) |
<0.001 |
1 |
MRI = magnetic resonance imaging
Table 3: MRI findings.
Table 4 demonstrates the sonographic findings. Both callous formation and subcutaneous hyperemia demonstrated statistically significant correlation with grade 4b BSI’s (p=0.03 for both). Periosteal edema was seen in 80% of patients, including all patients with grade 2-4b stress injuries and 50% of those with grade 1 injuries.
|
Characteristic |
Overall (n=25) |
Grade 1 (n=10) |
Grade 2 (n=6) |
Grade 3 (n=3) |
Grade 4a (n=3) |
Grade 4b (n=3) |
Stuart-Kendall Tau-c |
|||
|
Tau-c |
95% CI |
p-value |
||||||||
|
Periosteal edema |
20 (80%) |
5 (50%) |
6 (100%) |
3 (100%) |
3 (100%) |
3 (100%) |
0.48 |
0.173 |
0.79 |
0.002 |
|
Periosteal thickening |
12 (48%) |
3 (30%) |
2 (33%) |
2 (67%) |
2 (67%) |
3 (100%) |
0.48 |
0.104 |
0.86 |
0.012 |
|
Periosteal hyperemia |
6 (24%) |
1 (10%) |
0 (0%) |
2 (67%) |
0 (0%) |
3 (100%) |
0.454 |
0.06 |
0.85 |
0.024 |
|
Subcutaneous edema |
9 (36%) |
0 (0%) |
2 (33%) |
2 (67%) |
2 (67%) |
3 (100%) |
0.768 |
0.518 |
1 |
< 0.001 |
|
Cortical break |
4 (16%) |
0 (0%) |
0 (0%) |
1 (33%) |
0 (0%) |
3 (100%) |
0.486 |
0.12 |
0.85 |
0.009 |
|
Callous present |
2 (8.0%) |
0 (0%) |
0 (0%) |
0 (0%) |
0 (0%) |
2 (67%) |
0.282 |
-0.06 |
0.62 |
0.107 |
US = ultrasound; CI = confidence interval
Table 4: US findings.
The agreement in findings between US and MRI is variable. There was 96% agreement on the presence of a cortical break and 100% agreement on the presence of callous formation. US and MRI agreed 88% of the time on the presence of subcutaneous edema and 80% of the time on periosteal edema. The lowest degree of agreement was on periosteal thickening (64%).
4. Discussion
The primary finding in our study is that there is a correlation between US and MRI findings for BSIs of the metatarsal or tibia. These findings suggest that US may be useful in the diagnosis of an acute BSI.
The most agreed on findings were the presence of a cortical break and callous formation, which US and MRI agreed on 96% and 100% of the time, respectively. These findings are consistent with grade 4b injuries16. Further, the presence of subcutaneous edema, periosteal thickening, periosteal hyperemia, subcutaneous edema, and a cortical break were seen concomitantly in all grade 4b injuries. We can therefore conclude that either the presence of a cortical break and callous formation, or the pentad described above, is highly suggestive of a grade 4b BSI.
Periosteal edema was present on MRI for 100% of patients, while on US it was present for 100% of grade 2-4b injuries and 50% of grade 1 injuries, with an overall agreement between US and MRI of 80%. This suggests that the presence of periosteal edema on US is highly suggestive of a BSI but is not specific for a specific injury grade.
Other than a grade 4b BSI, moderate and even low-grade injuries are difficult to classify on US. No sonographic findings were specific for grade 1-4a injuries, and the findings seen for those grades were not consistently seen sonographically for all patients. The degree of agreement between US and MRI was moderate to high for the three variables with kappa values significantly different from 0, with the lowest being periosteal edema and periosteal thickening with kappa values of 0.00 and 0.29, respectively. Periosteal thickening had the lowest percentage agreement at only 64%, while periosteal edema had the lowest kappa statistic due to the lack of variability in MRI findings. In a prior systematic review evaluating common sonographic findings seen with stress injuries, the most common findings across all studies was cortical disruption (70% of participants) followed by periosteal thickening (66%) [12].
There are limitations with this study which should be mentioned. First, our study contained a rather small sample size as a result of an unexpectedly low enrollment rate. It is possible that this small sample was not large enough to detect a stronger correlation between US findings and BSI grade. A follow up study with a larger sample size would be helpful. Next, the primary author, who also performed all of the US examinations, knew that the patients had a confirmed diagnosis of a BSI. While the primary author was blinded to the location and grade of the injury, it is possible that the knowledge of a confirmed BSI could bias the primary author towards calling pathology on the US images, and thereby artificially increasing the correlation of findings between US and MRI. Additionally, prior studies have evaluated the sensitivity and specificity of US compared to MRI for the diagnosis of a BSI [4,12,13,17,18]. Because the primary author knew the confirmed diagnosis of a BSI, we were not able to reliably calculate sensitivity and specificity. Lastly, this study only included stress injuries of the tibia and metatarsals. It is unclear whether these findings would be seen in stress injuries of other bones.
Our findings suggest that the presence of one or more of the common pathologic sonographic findings seen with a BSI indicate the likely diagnosis of a BSI. However, the absence of these findings does not rule out a BSI, and MRI should be considered if there remains a clinical suspicion for a BSI. Additionally, if a patient has a radiograph-confirmed BSI, US may not provide any additional diagnostic information as it does not appear to provide accurate gradation of the BSI, and further imaging with MRI would be recommended if injury gradation is desired.
Conclusion
Our findings suggest that there is an association between US and MRI for imaging findings consistent with a BSI. However, other than grade 4b injuries, US does not seem to reliably or consistently determine injury grade. Therefore, we recommend that US be considered as a diagnostic tool for the detection of tibial and metatarsal BSIs. This is particularly helpful in low resource areas (limited financial ability to proceed with MRI, limited MRI scanner availability). However, if determination of injury grade is desired, MRI should be considered. Future high-level studies with larger sample sizes are needed to further detect if specific US findings can be helpful with injury gradation.
Funding:
None
Acknowledgments:
None
Disclosures:
Ryan Kruse is a consultant for Lipogems and Apex Biologix. The remaining authors have nothing to disclose.
References
- Waterman BR, Gun B, Bader JO, et al. Epidemiology of Lower Extremity Stress Fractures in the United States Military. Military Medicine 181 (2016): 1308-1313.
- Bratsman A, Wassef A, Wassef CR, et al. Epidemiology of NCAA Bone Stress Injuries: A Comparison of Athletes in Divisions I, II, and III. Orthopaedic Journal of Sports Medicine 9 (2021): 23259671211014496.
- Kelly S, Waring A, Stone B, et al. Epidemiology of bone injuries in elite athletics: A prospective 9-year cohort study. Physical Therapy in Sport 66 (2024): 67-75.
- Wright AA, Hegedus EJ, Lenchik L, et al. Diagnostic Accuracy of Various Imaging Modalities for Suspected Lower Extremity Stress Fractures: A Systematic Review With Evidence-Based Recommendations for Clinical Practice. Am J Sports Med 44 (2016): 255-263.
- Robertson GA, Wood AM. Return to sports after stress fractures of the tibial diaphysis: a systematic review. Br Med Bull 114 (2015): 95-111.
- Boden BP, Osbahr DC, Jimenez C. Low-Risk Stress Fractures. The American Journal of Sports Medicine 29 (2001): 100-111.
- Schneiders AG, Sullivan SJ, Hendrick PA, et al. The ability of clinical tests to diagnose stress fractures: a systematic review and meta-analysis. J Orthop Sports Phys Ther 42 (2012): 760-771.
- Dobrindt O, Hoffmeyer B, Ruf J, et al. Estimation of return-to-sports-time for athletes with stress fracture - an approach combining risk level of fracture site with severity based on imaging. BMC Musculoskelet Disord 13 (2012): 139.
- Jacobson JA. Musculoskeletal ultrasound and MRI: which do I choose? Semin Musculoskelet Radiol 9 (2005): 135-149.
- Bierig SM, Jones A. Accuracy and Cost Comparison of Ultrasound Versus Alternative Imaging Modalities, Including CT, MR, PET, and Angiography. Journal of Diagnostic Medical Sonography 25 (2009): 138-144.
- Bodner G, Stöckl B, Fierlinger A, et al. Sonographic findings in stress fractures of the lower limb: preliminary findings. Eur Radiol 15 (2005): 356-359.
- Schaper M, Harcus J. Preliminary image findings of lower limb stress fractures to aid ultrasonographic diagnoses: A systematic review and narrative synthesis. Ultrasound 29 (2021): 208-217.
- Banal F, Gandjbakhch F, Foltz V, et al. Sensitivity and specificity of ultrasonography in early diagnosis of metatarsal bone stress fractures: a pilot study of 37 patients. J Rheumatol 36 (2009): 1715-1719.
- Hoenig T, Hollander K, Popp KL, et al. International Delphi consensus on bone stress injuries in athletes. British Journal of Sports Medicine 59 (2025): 78.
- Banal F, Etchepare F, Rouhier B, et al. Ultrasound ability in early diagnosis of stress fracture of metatarsal bone. Ann Rheum Dis 65 (2006): 977-978.
- Fredericson M, Bergman AG, Hoffman KL, et al. Tibial stress reaction in runners. Correlation of clinical symptoms and scintigraphy with a new magnetic resonance imaging grading system. Am J Sports Med 23 (1995): 472-481.
- Papalada A, Malliaropoulos N, Tsitas K, et al. Ultrasound as a primary evaluation tool of bone stress injuries in elite track and field athletes. Am J Sports Med 40 (2012): 915-919.
- Rao A, Pimpalwar Y, Sahdev R, et al. Diagnostic Ultrasound: An Effective Tool for Early Detection of Stress Fractures of Tibia. Journal of Archives in Military Medicine (2017): In Press.


 Impact Factor:
* 5.3
Impact Factor:
* 5.3
 Acceptance Rate:
73.64%
Acceptance Rate:
73.64%
 Time to first decision: 10.4 days
Time to first decision: 10.4 days
 Time from article received to acceptance:
2-3 weeks
Time from article received to acceptance:
2-3 weeks
