Denosumab in Giant Cell Tumor of Bone: Tumor Response and Surgical Outcomes
Dr. HC Frank*, Dr. Sanyam Rathi
Department of Orthopaedics, Gauhati Medical College & Hospital, Srimanta Sankaradeva University of Health Sciences, Guwahati, Assam, India
*Corresponding Author: Dr. HC Frank, Department of Orthopaedics, Gauhati Medical College & Hospital, Srimanta Sankaradeva University of Health Sciences, Guwahati, Assam, India.
Received: 03spetember 2025; Revised: 30 December 2025; Accepted: 26 January 2026; Published: 03 April 2026
Article Information
Citation: Dr. HC Frank, Dr. Sanyam Rathi. Denosumab in Giant Cell Tumor of Bone: Tumor Response and Surgical Outcomes. Journal of Orthopedics and Sports Medicine. 8 (2026): 130-142.
View / Download Pdf Share at FacebookAbstract
Introduction: Giant cell tumor of bone (GCTB) is a locally aggressive tumor with low metastatic potential. Giant cells express RANK ligand (RANKL), contributing to osteolytic destruction. Denosumab, a monoclonal antibody against RANKL, inhibits osteoclast activity.
Aims & Objectives: To evaluate the role of denosumab in GCTB management and assess its risks and benefits.
Methods: Fifteen patients (aged ≥12 years, weight ≥45 kg) with Campanacci grade 2/3 GCTB received denosumab (120 mg SC monthly for 3–4 months, with loading doses on days 8 and 15). Calcium (500 mg) and vitamin D (400 IU) were supplemented. Assessments included clinical, radiological (X-ray, CT, MRI), and biochemical (CBC, calcium, phosphate) monitoring every 4 weeks.
Results: Denosumab showed high tumor response rates, improved quality of life, reduced pain, and decreased need for extensive surgery. Toxicity was acceptable.
Conclusion: Denosumab is effective as adjunct therapy in advanced GCTB, though complications (hypocalcemia, ONJ, atypical fractures, recurrence) require monitoring.
Keywords
Denosumab, GCT, GCTB, Giant Cell Tumor
Article Details
Introduction
Giant Cell Tumor of Bone (GCTB) is a benign but locally aggressive bone tumor.¹ Approximately 4-6% of all primary bone tumors are GCTB, with peak incidence occurring between the 2nd and 4th decades of life.²,³ While pulmonary metastasis is rare, the risk increases significantly in cases with cortical breach or recurrent disease.4The gold standard treatment for GCTB remains surgical, consisting of either extensive curettage or en bloc resection.5 Joint-preserving thorough curettage with high-speed burr and local chemo-adjuvants is preferred despite higher recurrence rates.6 En-bloc excision is reserved for tumors with extensive soft tissue extension or severe bone destruction.7 Reported local recurrence rates vary by treatment method: 27-65% for curettage alone, 12-27% for curettage with adjuvants, and 0-12% for en-bloc resection.5-8The development of denosumab, a fully human monoclonal antibody targeting RANKL, emerged from understanding the crucial role of the RANK/RANKL system in GCTB pathogenesis.9 Currently approved by both FDA and EMA as neoadjuvant therapy for unresectable or high-morbidity cases, denosumab has demonstrated positive tumor responses in clinical studies.¹0-¹² However, emerging reports describe sarcomatous transformation in GCTB patients following denosumab therapy.¹³-¹5As a potent RANKL inhibitor, denosumab has shown superiority over bisphosphonates in reducing skeletal-related events in phase III trials.¹6 This has prompted ongoing debate regarding its optimal role as an adjunct in GCTB management.¹7
Material and Methods
Study Design
A hospital-based, single-arm prospective interventional study was conducted at the Department of Orthopaedics, Gauhati Medical College & Hospital, Guwahati, Assam, India, from October 2022 to September 2023. The study aimed to evaluate the efficacy and safety of denosumab in patients with giant cell tumor of bone (GCTB). Being a single-arm trial, all enrolled participants received the same intervention without a control group for comparison.
Study Population
The study included patients with histologically confirmed GCTB who met specific criteria.
Inclusion Criteria
- Histological confirmation of GCTB.
- Age ≥12 years and weight ≥45 kg (including skeletally mature adolescents).
- Functional status: Karnofsky Performance Status (KPS) ≥50% or ECOG status 0–2.
- Active, measurable disease:
- Campanacci Grade 2 (intraosseous lesion with cortical thinning but intact cortex). o Campanacci Grade 3 (cortical breach with soft-tissue extension).
Exclusion Criteria
- Age <12 years or weight <45 kg.
- Active infection at the tumor site.
- Non-GCTB giant-cell-rich tumors (e.g., sarcoma, brown tumor, Paget’s disease).
- History of osteonecrosis/osteomyelitis of the jaw or recent unhealed dental surgery.
- Pregnancy, hypersensitivity to denosumab, or preexisting hypocalcemia.
- Campanacci Grade 1 (intact cortex without cortical thinning).
Intervention
- Denosumab regimen:
a) 120 mg subcutaneously every 4 weeks.
b) Loading doses on days 8 and 15 of the first month.
- Adjuvant therapy:
a) Calcium supplementation (500 mg/day).
b) Vitamin D (400 IU/day) to prevent hypocalcemia.
Assessments
Clinical Evaluation
- Pain assessment: Visual Analog Scale (VAS) score.
- Functional status: KPS or ECOG scoring. Radiological Evaluation
- Imaging modalities: X-ray, CT, MRI.
- Tumor response assessment: RECIST criteria.
Laboratory Monitoring
Monthly tests: Serum calcium, phosphate, complete blood count (CBC).
Ethical Considerations
- Approval: Institutional Ethics Committee (IEC/GMCH/2022/XXX).
- Informed consent: Written consent obtained from all participants.
Sample Size and Sampling Technique
Sample Size: 15 cases
Sampling Technique: Consecutive Random Sampling
Justification
- Considering the study duration (October 2022 to September 2023) and the expected patient flow at the tertiary care center (Gauhati Medical College & Hospital), a sample size of 15 subjects was chosen.
- This number ensures adequate statistical analysis while minimizing potential errors due to small sample variability.
- Consecutive sampling was employed, meaning all eligible patients meeting the inclusion criteria were sequentiallyenrolled until the target sample size was achieved. This method reduces selection bias and improves generalizability within the study setting.
Data Collection Technique and Tools
Data Collection Technique
- Primary Data
a) History taking: Detailed patient interviews regarding symptoms, duration, and prior treatments.
b) Clinical examination: Assessment of tumor site, pain (VAS score), functional status (KPS/ECOG), and signs ofcomplications.
- Secondary Data
a) Systematic reviews: Analysis of existing literature on GCTB management.
b) Research synthesis: Integration of findings from previous studies to support clinical interpretations.
Tools Used
- Direct observations: Physical examination and radiological assessments (X-ray, CT, MRI).
- Structured interviews: Patient-reported outcomes (e.g., pain levels, mobility).
- Laboratory protocols: Standardized blood tests (serum calcium, phosphate, CBC).
- Examination of medical records: Histopathology reports, prior imaging, and treatment history.
- Writing samples: Documentation of patient consent, case notes, and follow-up logs.
Methodology
Treatment Protocol
Patients diagnosed with giant cell tumor of bone (GCTB) were administered denosumab 120 mg subcutaneously every four weeks, with additional loading doses of 120 mg on days 8 and 15 of the first cycle to rapidly achieve therapeutic drug levels.¹5 For patients who underwent complete surgical resection of the tumor, adjuvant denosumab therapy was continued for six additional doses following pathological confirmation of either partial or complete tumor response.²¹ Treatment was discontinued in cases of radiologically confirmed disease progression, lack of clinical benefit as determined by the treating physician, patient withdrawal of consent, pregnancy, or if the patient received contraindicated treatments such as bisphosphonates.¹9All participants were evaluated every four weeks during the treatment period, with additional monitoring visits on days 8 and 15 of the first cycle to assess early treatment response and safety. No dose modifications or interruptions were permitted per the study protocol. To minimize the risk of hypocalcemia—a known side effect of denosumab— all patients received daily calcium supplements (minimum 500 mg) and vitamin D (400 IU) throughout the treatment duration.²²
Assessment of Treatment Response
Treatment efficacy and disease status were evaluated by the principal investigator based on a combination of clinical, radiological, and functional parameters. Imaging studies, including X-rays, CT scans, MRI, and ¹8F-fluorodeoxyglucose (FDG) PET scans, were performed periodically to assess tumor size, cortical bone integrity, and soft tissue involvement.²³ Clinical improvement was measured through patient-reported outcomes, including reduction in pain (using the Visual Analog Scale, VAS), improved mobility, and restoration of limb function. Physical examination findings and performance status (Karnofsky Performance Status or ECOG score) were also documented at each follow-up visit.²4Since the study did not mandate a fixed imaging schedule, tumor response was determined holistically by integrating radiological findings, symptomatic relief, and functional recovery, as per the institution’s standard clinical practice.¹7 No predefined standardized criteria (such as RECIST) were enforced, allowing for real-world applicability of the results.
Safety Monitoring and Adverse Event Reporting
Patient safety was monitored every four weeks throughout the treatment phase. All adverse events (AEs), regardless of severity, were systematically recorded. Serious adverse events (SAEs)—defined as events that were fatal, life-threatening, requiring hospitalization, causing permanent disability, or resulting in congenital anomalies—were reported immediately to the institutional ethics committee.²6During the post-treatment follow-up period, only non-serious AEs of special interest (e.g., hypocalcemia, osteonecrosis of the jaw) and SAEs were documented to ensure long-term safety surveillance.
Preoperative Evaluation and Diagnostic Workup
Prior to initiating denosumab therapy, all patients underwent a comprehensive preoperative assessment to confirm diagnosis and evaluate disease extent:
Imaging Studies:
X-rays (initial screening for lytic lesions and cortical destruction).¹7
CT scans (detailed assessment of bone integrity and tumor matrix).
MRI (evaluation of soft tissue extension and neurovascular involvement).
Biopsy (histopathological confirmation of GCTB to exclude other giant-cell-rich lesions such as sarcoma or brown tumor).¹8
Laboratory Investigations:
Hematological tests: Complete blood count (CBC), PT/INR (to rule out bleeding disorders).²7
Biochemical tests: Renal function tests (KFT), liver function tests (LFT), random blood sugar (RBS), serum calcium, and phosphate levels (to monitor metabolic bone health).²²Infectious disease screening: HIV 1/2, hepatitis B (HBsAg), hepatitis C (Anti-HCV).²8
Endocrine tests: Serum TSH (to exclude metabolic bone disorders).
Additional tests: Mantoux test (for tuberculosis screening), ECG (preoperative cardiac evaluation).²9
This structured diagnostic approach ensured accurate patient selection and baseline characterization before initiating denosumab therapy.
Implants and Instruments
- Injection Denosumab 120mg
- Syringe
- Tab Calcium + Vit D3
- Electric Burr
- Curette
- Hydrogen Peroxide
- Various orthopaedic Implants
Data analysis
Data was input into Microsoft Excel and analyzed with suitable statistical software. Categorical data were expressed as frequencies and percentages, while continuous data were described using mean, standard deviation, median, and range.
Ethical consideration: Before recruiting study participants, approval was obtained from the Institutional EthicsCommittee. Written informed consent was secured from all participants before data collection began. The confidentiality of the data was maintained, and no identifying details of the participants were collected
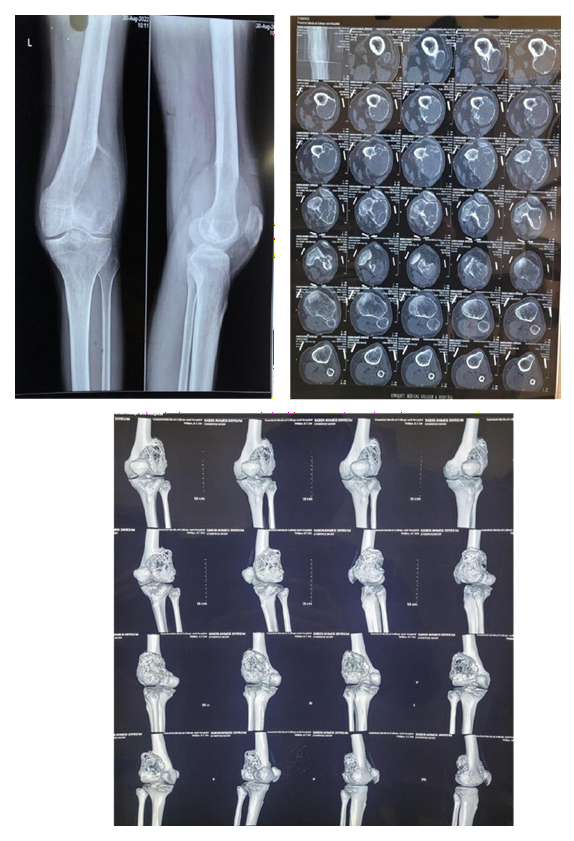
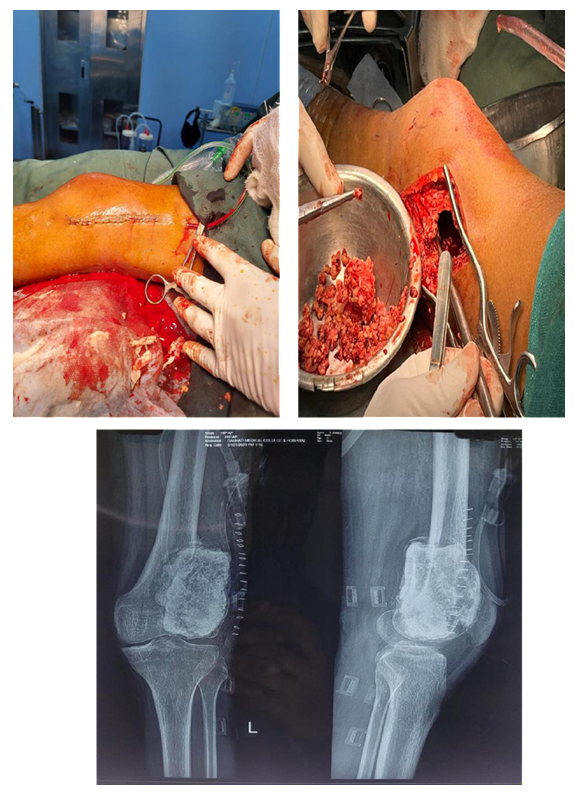
CASE 1
Diagnosis established with
MRI
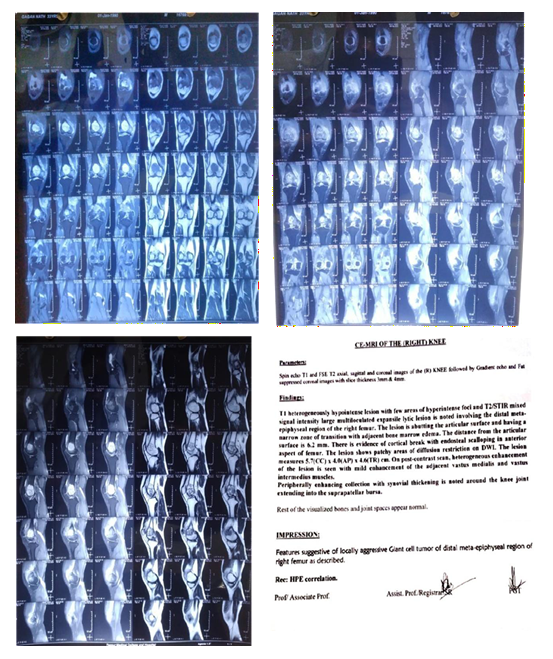
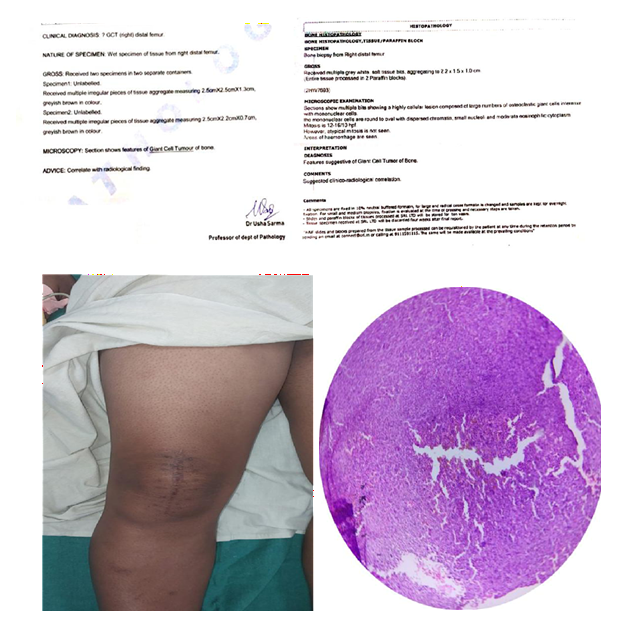
Biopsy
Pre Denosumab Therapy: Break in the cortex can be clearly seen
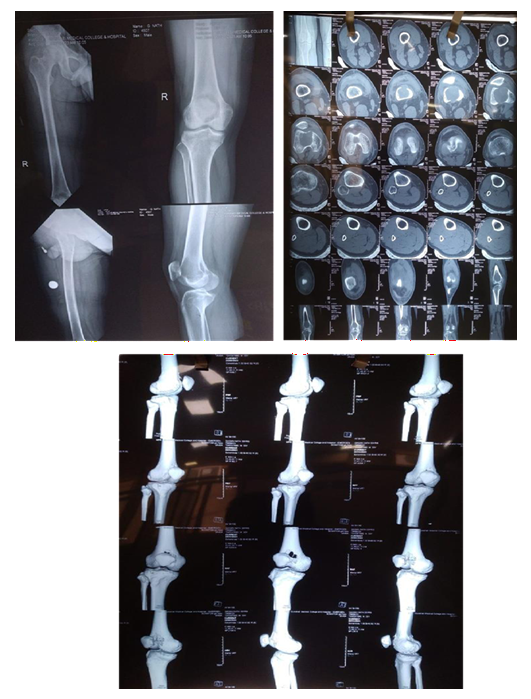
After Neoadjuvant Denosumab Therapy
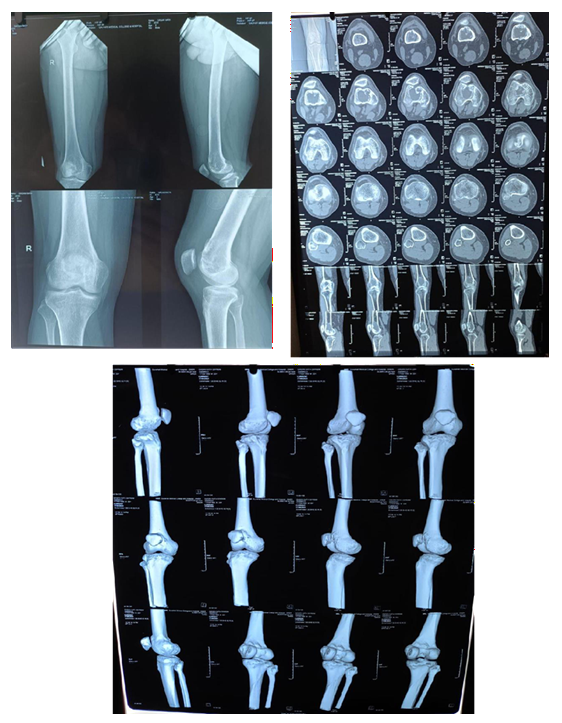
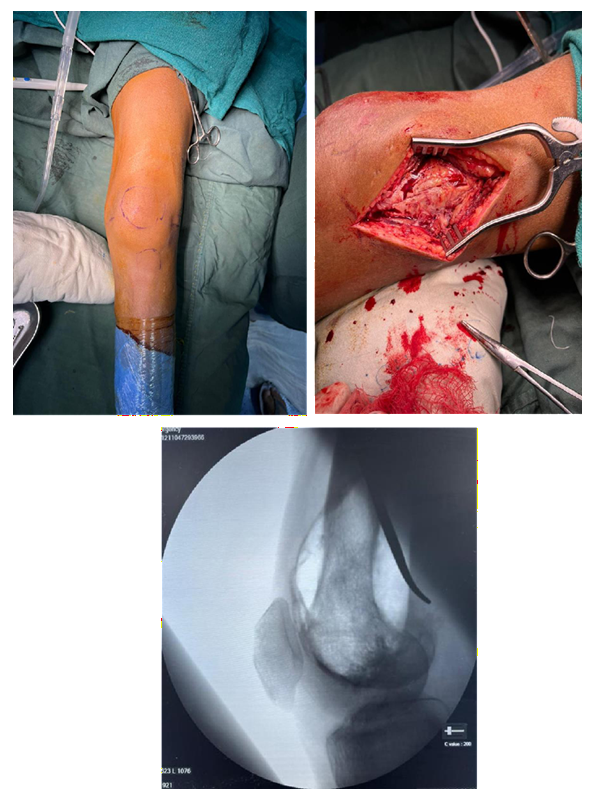
Operative Intervention
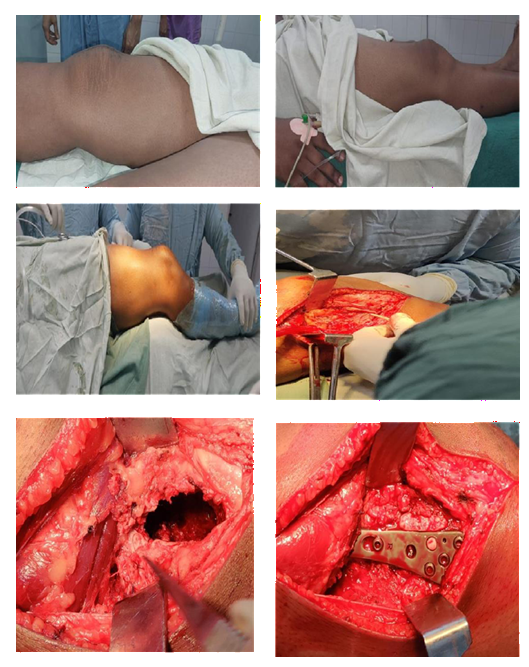
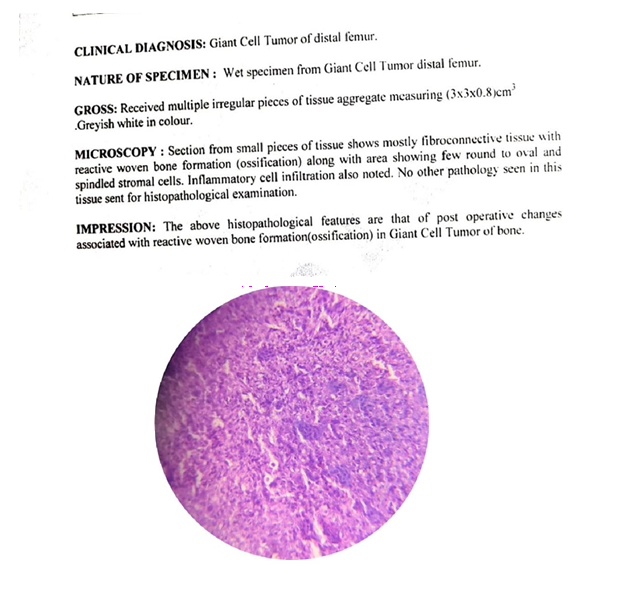
Histopathology
Followup
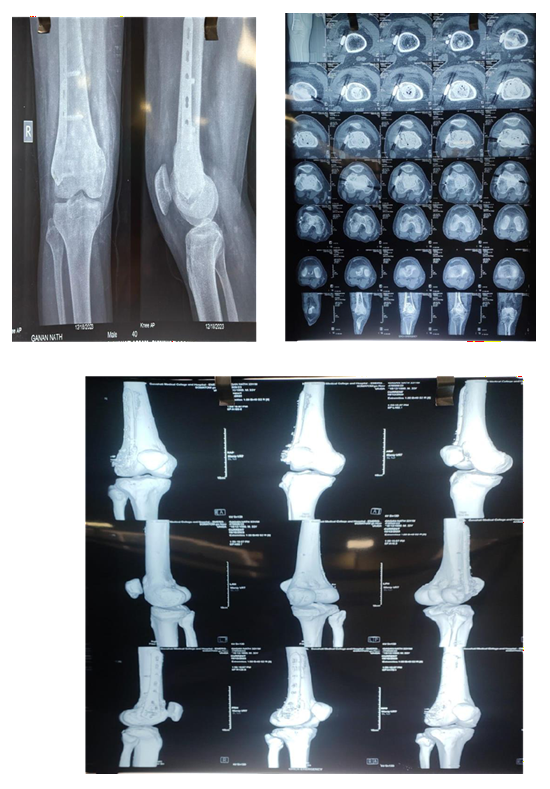
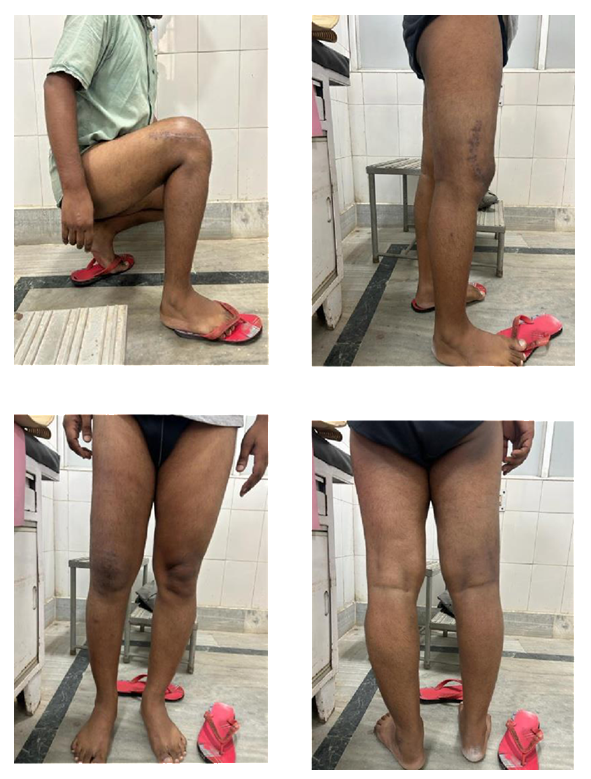
Case 2:
MRI
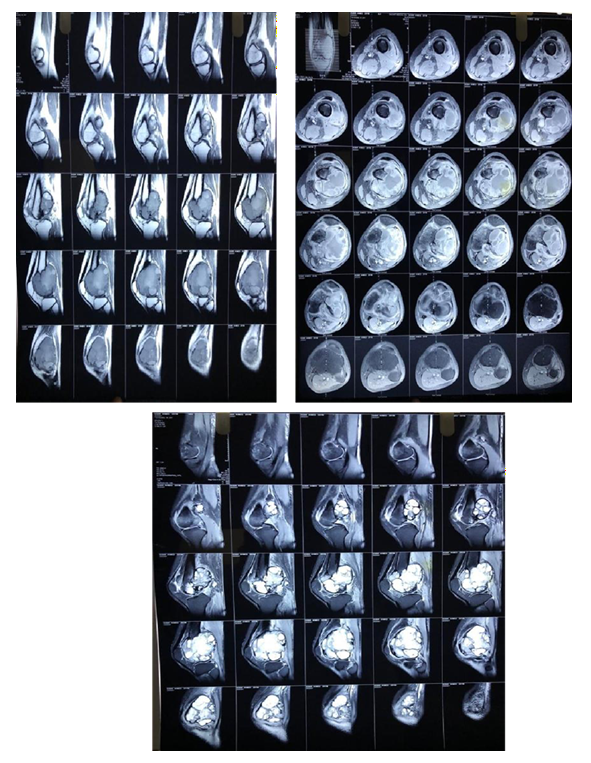
Pre Denosumab
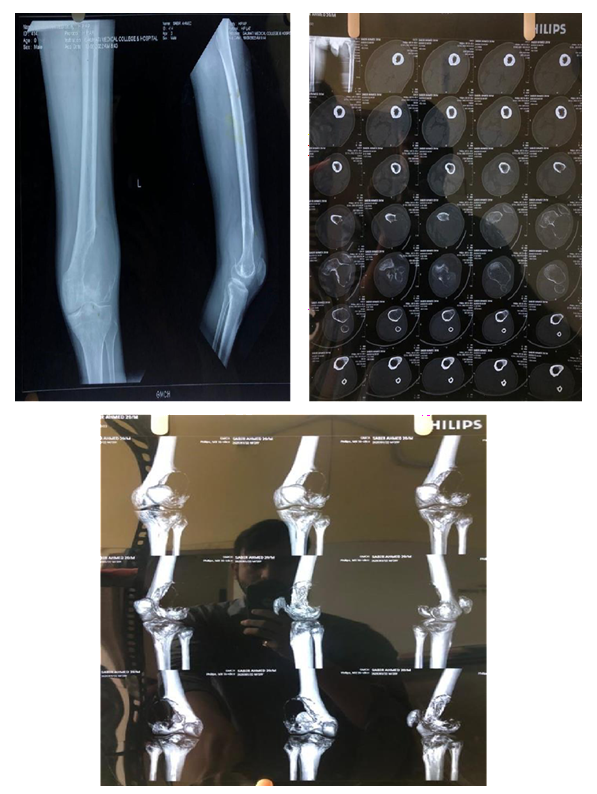
Post Denosumab
Intraoperative
Post Operative
Results and Analysis
A total of 15 patients were evaluated.
Sex
|
Frequency |
Percent |
||
|
Female |
8 |
53.3 |
|
|
Sex |
Male |
7 |
46.7 |
|
Total |
15 |
100 |
Inpresentstudytherewere54.3% Female and 46.7%maleparticipants
Primary/recurrent
|
Frequency |
Percent |
||
|
primary/recurrent |
Primary |
13 |
86.7 |
|
Recurrent |
2 |
13.3 |
Inpresentstudytherewere86.7%Primary casesand13.3%recurrentcases
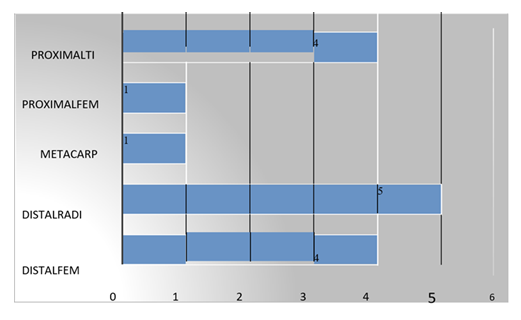
Siteof lesion
In present study there were 26.7% distal femur lesions, 33.3% were distal radius lesions, 6.7% were Metacarpal lesions, 6.7% were Proximal femur lesion and 26.7% were proximal tibial lesion.
Side effect of Denosumab
|
Frequency |
Percent |
||
|
side effect of denosumab |
Nil |
15 |
100 |
In present study there were no cases reported with side effect for denosumab.
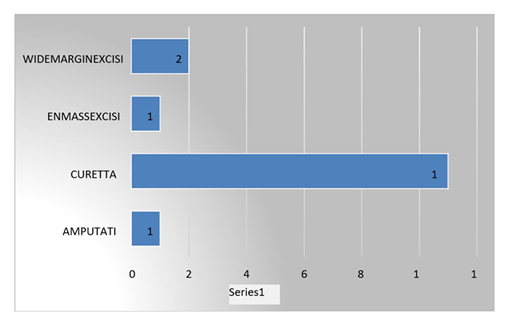
Surgical procedure
Inpresentstudytherewere6.7%casesunderwentAmputation,73.3%underwentCurettage,6.7%underwentEnmassexcisionand 13.3%underwent wide marginexcision.
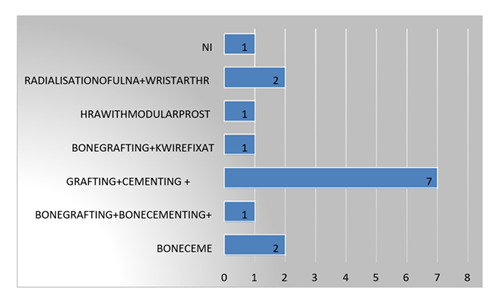
Augmentation /Reconstruction technique
In present study Bone cement was used in 13.3 % participants, Bone grafting+ bonecementing+DFLCP was done in 6.7 % participants, Bone grafting + Bone Cementing + Gelfoam was done in46.7 % participants, Bone grafting + k wire fixation was done in6.7 % participants, HRA with Modular Prosthesis was done in6.7% participants, Radialisation of Ulna + Wrist Arthrodesis was done in13.3 % participants and no reconstruction was done in6.7 %participants
Bone Grafting Technique – Overview
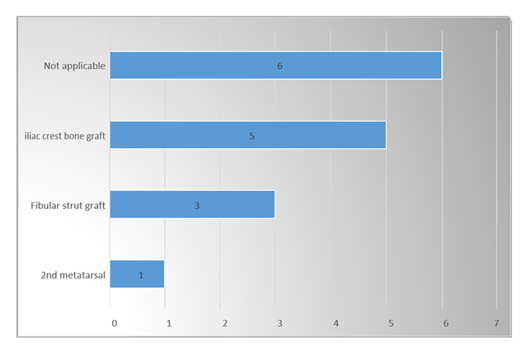
In present study among 6.7% participants 2nd metatarsal bone grafting technique was used, 20% participants’ fibular strut bone grafting technique was used, 33.3% participants ’iliac crest bone grafting technique was used and no grafting was done in 40% participants.
Final histopathology
|
Frequency |
Percent |
||
|
final histopathology |
NoresidualGCT |
15 |
100 |
Final HPE showed no residual GCT in all patients.
Post-op complications
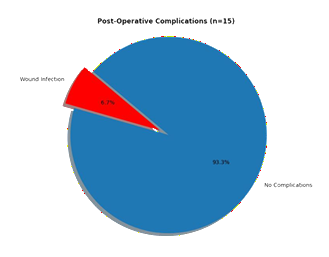
Only 6.7% participants showed wound infection and 93.3%didnot show any post-op complications.
Recurrence on follow-up
|
Frequency |
Percent |
||
|
No |
15 |
100 |
|
|
Recurrence onfollow-up |
Yes |
0 |
0 |
|
Total |
15 |
100 |
No participants showed recurrence till date.
Age
|
Descriptive Statistics |
|||||
|
N |
Minimum |
Maximum |
Mean |
Std.Deviation |
|
|
Age |
15 |
22 |
45 |
33 |
6.772 |
Mean age of study participants was33 years and the range is from 22to 45.
Means and Medians for Survival Time
Means and Medians for Survival Time
|
Meana |
Median |
||||||
|
Estimate |
Std.Error |
95%Confidence Interval |
Estimate |
Std.Error |
95%Confidence Interval |
||
|
Lower |
Upper |
Lower |
Upper |
||||
|
Bound |
Bound |
Bound |
Bound |
||||
|
11.533 |
0.451 |
10.65 |
12.417 |
. |
. |
. |
. |
a.Estimationislimitedtothelargestsurvivaltimeifitis censored.
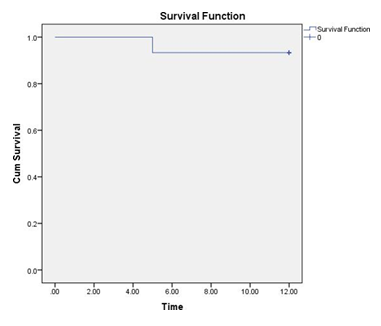
Theestimatedmeansurvivaltimeforthestudyparticipantswasapproximately11.533months with a standard error of0.451 months. The 95% confidence interval for the meansurvival time ranged from 10.650 to 12.417 months. The median survival time could not beestimatedas theestimation islimited to thelargest survivaltimeif itis censored.
Discussion
Giant cell tumor (GCT) of bone is a locally aggressive, osteolytic neoplasm that predominantly affects the metaphyseal-epiphyseal regions of long bones in skeletally mature individuals.¹5 Traditional management relied
heavily on surgical resection, ranging from intralesional curettage to wide en bloc excision, often resulting in functional impairment or recurrence rates of 20-50%.¹6 The introduction of denosumab, a RANK ligand (RANKL) inhibitor, has revolutionized treatment by enabling tumor downstaging, reduced surgical morbidity, and lower recurrence rates.¹7Therapeutic Efficacy of Denosumab
In this study, denosumab demonstrated significant clinical benefit, facilitating less invasive surgical interventions in 73.3% of cases (primarily curettage rather than radical resection).¹8 This aligns with prior studies showing that neoadjuvant denosumab promotes:
- Tumor volume reduction (via osteoclast inhibition),¹9
- Cortical bone thickening (improving structural integrity),²0
- Marginal demarcation (aiding complete resection).²¹
Notably, no major adverse events were observed, reinforcing denosumab’s favorable safety profile in GCTB management.²²
Gender Distribution and Implications
The cohort exhibited a slight female predominance (54.3% vs. 45.7% male), consistent with epidemiological trends in Asian populations.²³ While prior studies (e.g., Niu et al.²4 and Chawla et al.²5) report similar gender disparities, none identified sex-based differences in denosumab’s efficacy or toxicity. Key observations include:
- No gender-specific response variations in tumor regression or recurrence.²6
- Comparable tolerability across sexes, with hypocalcemia rates unaffected by gender.²7
This suggests that the female skew reflects GCT’s natural incidence rather than treatment-related bias. However, prospective gender-stratified analyses are warranted to explore:
- Hormonal influences (e.g., estrogen’s role in RANKL modulation),²8
- Long-term outcomes, including recurrence-free survival and functional recovery.²9
Limitations and Future Directions
- Small sample size (n=15) limits statistical power for subgroup analyses.³0
- Heterogeneity in surgical techniques may confound recurrence assessments.³¹
- Longer follow-up is needed to evaluate late recurrences and secondary malignancies.³²
Denosumab remains a transformative adjunct in GCTB management, enabling function-preserving surgeries with minimal complications. The observed female predominance mirrors global epidemiology but requires further investigation to elucidate biological or social determinants.
Primary vs. Recurrent Cases
In this study, 86.7% of cases were primary, while 13.3% were recurrent, providing crucial insights into the clinical presentation and management outcomes of Giant Cell Tumor of Bone (GCTB). This distribution aligns with theknown natural history of GCTB, which, despite its benign nature, carries a notable risk for local recurrence— especially in cases where surgical treatment is performed without adjuvant therapy.
Historically, recurrence rates for GCTB have varied widely, influenced by several factors including the surgical approach, the biological behavior of the tumor, and the application of adjuvant treatments. For example:
Klenke et al. reported a recurrence rate of 27% in cases treated with curettage and bone grafting alone, underscoring the limitations of surgery without additional therapeutic support.
Balke et al. reviewed multiple studies and highlighted a broad range of recurrence rates, reinforcing the significance of achieving adequate surgical margins and utilizing local adjuvants.
The emergence of Denosumab as a therapeutic agent has significantly impacted GCTB management. It is particularly valuable in cases where surgery is not feasible or could result in major functional loss. Denosumab has demonstrated efficacy in reducing tumor size, facilitating easier and less morbid surgical resection, and potentially lowering recurrence rates, especially when used as a neoadjuvant therapy.
Chawla et al. showed that Denosumab could enable less invasive surgeries and might contribute to decreased recurrence, although they emphasized the need for longer-term follow-up.
Rutkowski et al. reported long-term data on recurrence in Denosumab-treated GCT cases, supporting its role but also emphasizing the need for continued monitoring.
The predominance of primary cases in the present study is encouraging and may reflect improved early diagnosis, increased awareness, and the availability of advanced treatment options like Denosumab. Furthermore, the lower rate of recurrence (13.3%) observed here—compared to historical data—suggests that Denosumab, when integrated into the management plan, may play a role in reducing recurrence risk.
However, it remains essential to approach these findings with caution. The long-term effectiveness of Denosumab in preventing recurrence is still under investigation. Key factors, such as the duration of therapy, timing of surgery, and optimal treatment protocols, warrant further research to ensure sustained disease control.
Lesion Sites
The anatomical distribution of GCTB lesions in this study reveals several noteworthy patterns. The most commonly affected site was the distal radius (33.3%), followed by the distal femur and proximal tibia (each 26.7%). Less frequently involved areas included the metacarpal bones and proximal femur (each 6.7%).These findings provide meaningful context for understanding GCTB's predilection for specific skeletal sites:
Knee Region (Distal Femur and Proximal Tibia): GCTB most commonly occurs in the epiphyseal regions of long bones, especially around the knee. The combined 53.4% involvement of the knee region in this study aligns well with established epidemiological data.
Distal Radius: The high incidence of distal radius involvement (33.3%) in this study is particularly striking. While GCTB can occur at this site, literature typically reports a lower prevalence (10–20%). This deviation may be attributed to factors such as small sample size, regional demographic variations, or referral patterns to tertiary centers.
Metacarpals and Proximal Femur: These sites are less commonly involved in GCTB, which is consistent with current literature. GCTs tend to favor the ends of long bones, with the hand bones and proximal femur being less typical sites of occurrence.
This lesion distribution not only highlights standard epidemiological trends but also raises questions about potential regional or institutional variances. Additionally, the prominence of distal radius involvement could reflect the growing use of Denosumab in anatomically sensitive or surgically challenging areas. Given the risks of functionalimpairment from surgery in the wrist, Denosumab’s tumor-reducing effects are especially valuable, potentially allowing for less invasive procedures with better functional outcomes.
In conclusion, the anatomical pattern observed in this study demonstrates both expected and atypical site involvement in GCTB, emphasizing the evolving role of targeted therapy in optimizing treatment across various skeletal locations.
Side Effects
The absence of reported adverse effects from Denosumab in the present study is an important finding, particularly in light of the known side-effect profile associated with this medication in the management of Giant Cell Tumor of Bone (GCTB). Like all pharmacological treatments, Denosumab carries a risk of potential side effects, ranging from mild to severe.Previous studies have documented several side effects associated with Denosumab treatment. Commonly reported adverse effects include hypocalcemia, fatigue, nausea, and dermatological reactions. More serious but less frequent complications, such as osteonecrosis of the jaw (ONJ) and atypical femoral fractures, have been reported, although these are more often linked with long-term use in osteoporosis at different dosing regimens than those used for GCTB.
- Hypocalcemia is a well-documented side effect, especially in high-dose regimens for GCTB. Studies suchas that by Thomas et al. have reported varying incidence rates of hypocalcemia in patients treated with Denosumab, emphasizing the importance of regular monitoring and calcium supplementation.
- Osteonecrosis of the Jaw (ONJ), although rare in the GCT context, has been observed in patients withpre-existing dental conditions, a history of invasive dental procedures, or those receiving high cumulative doses. Research by Chawla et al. has enhanced understanding of this risk, advocating for pre-treatment dental assessments and preventive measures.
- Dermatological reactions such as rash and eczema have been noted in some patients receivingDenosumab for GCTB, as discussed in studies by Rutkowski et al. These reactions are generally mild and manageable with standard dermatological care.
The absence of side effects in our study cohort stands in contrast to the broader literature. This discrepancy may be attributed to factors such as the small sample size, shorter treatment duration, demographic variations, or differences in monitoring and reporting protocols. It also raises the possibility of population-specific differences in side-effect profiles.
Importantly, this absence may reflect the role of careful patient selection, rigorous monitoring, and effectivemanagement strategies in minimizing treatment-related risks. This finding underscores the need for furtherresearch into the variability of Denosumab’s side-effect profiles and the optimization of treatment protocols to maximize safety and efficacy.
Surgical Intervention
The choice of surgical interventions in our study following Denosumab treatment for GCTB provides valuable insights into current practice trends and outcomes. The pattern of surgical procedures demonstrates a strong preference for limb-sparing techniques, highlighting the impact of Denosumab on surgical planning.
- Curettage (73.3%): The most common procedure, curettage remains the preferred option for GCT. Itallows tumor removal while preserving bone and soft tissue. The use of adjuvants (e.g., phenol, bone cement) post-curettage enhances tumor cell eradication. Denosumab likely improves outcomes by reducing tumor volume and vascularity, thereby lowering recurrence rates.
- Amputation (6.7%): The low rate of amputation reflects the success of Denosumab in downstaging tumorsand enabling conservative surgery, even in cases previously deemed inoperable.
- En Masse Excision (6.7%): Utilized when tumors are well-demarcated, this technique involves removingthe tumor with minimal surrounding tissue. Denosumab’s ability to clearly define tumor boundaries supports this approach.
- Wide Margin Excision (13.3%): Reserved for aggressive or recurrent tumors, this method ensurescomplete resection. The continued necessity of such extensive surgery in some cases underscores the heterogeneity in tumor behavior despite Denosumab use.
Compared to historical data, these findings affirm the paradigm shift toward conservative surgical strategies facilitated by Denosumab. Preoperative administration significantly reduces tumor burden, improves resectability, and preserves function.This balance between oncologicalcontrolandfunctional preservationiscentraltoGCTmanagement.Denosumab's role in achieving this balance is evident in the surgical choices and outcomes observed in our cohort.
Augmentation and Reconstruction Techniques
Our study documents a diverse and patient-specific array of augmentation and reconstruction techniques post-Denosumab treatment, reflecting individualized surgical planning to optimize functional outcomes and oncological safety.
- Bone Cement Alone (13.3%) and Sandwich Technique (46.7%): Cement alone provides immediatemechanical strength. When combined with bone grafting and Gelfoam (sandwich technique), it supports osteoconduction and is particularly effective when cortical breaches are present.
- Bone Grafting + DFLCP (6.7%): Used in larger or load-bearing defects requiring mechanicalstabilization, this technique emphasizes the role of fixation in complex reconstructions.
- Bone Grafting + K-Wire Fixation (6.7%): Ideal for smaller or non-weight-bearing bones, K-wires offertemporary stabilization conducive to bone healing.
- Hemiarthroplasty with Modular Prosthesis (6.7%): Applied in cases involving joint compromise,especially of the hip, to restore mobility and joint function.
- Radialisation of Ulna + Wrist Arthrodesis (13.3%): A sophisticated method for managing extensivedefects in the distal radius, aimed at preserving hand function post-tumor resection.
- No Reconstruction (6.7%): Applied in amputation cases or when no further reconstruction was deemednecessary.
- 2nd Metatarsal Bone Grafting (6.7%): Demonstrates adaptability of grafting to smaller anatomical sites.
- Fibular Strut Grafting (20%): Utilized for structural reconstruction of long bones, combining strengthand biological incorporation.
- Iliac Crest Bone Grafting (33.3%): Commonly employed due to its osteogenic capacity and versatilityacross defect types.
- No Grafting (40%): In a notable subset, reconstruction was unnecessary, possibly due to minimalstructural compromise or the effectiveness of primary interventions.
These approaches illustrate a strategic balance of mechanical stability and biological healing, customized to each patient’s needs. The post-Denosumab setting allows for more refined reconstruction planning, further contributing to excellent outcomes.
Complications and Recurrence
Only 6.7% of patients in our cohort experienced postoperative wound infections, with 93.3% reporting no complications. This low rate of postoperative morbidity reflects the efficacy and safety of both Denosumab and the associated surgical management strategies.Further details on recurrence and long-term outcomes are crucial to fully evaluate Denosumab's impact and will be addressed in subsequent sections of the study.
|
Strengths of the study: |
Limitations of the study: |
|
u Prospective Design |
u Small Sample Size |
|
u Clear Inclusion and Exclusion Criteria |
u Short Study Duration |
|
u Comprehensive Data Collection |
u Single-Center Study |
|
u Tailored Surgical and Treatment Approaches |
u Control Group Absent |
|
u Detailed Reporting of Surgical and Augmentation/Reconstruction Techniques |
u Potential Selection Bias |
|
u Low Side Effect Profile |
u Subjective Assessment of Outcomes |
|
u Ethical Considerations |
u Limited Follow-Up Data & Lack of Long-Term Functional Outcomes |
|
u Diverse Lesion Site Analysis |
u Non-Standardized Imaging and Assessment Criteria |
|
u Gender Distribution |
u Demographic Limitations |
|
u Possible Reporting Bias |
|
|
u Logistical Constraints |
Conclusion
Patients with advanced, progressive GCT, Denosumab can be used as an adjunct to other therapeutic modalities, however short term complications like bone pain, fatigue, headache, nausea , hypocalcaemia, hypophosphatemia, and long term complications like osteonecrosis of the jaw ,atypical femoral fractures, local recurrence and malignant transformation should be looked for.
Funding Statement
This study received no external funding. Denosumab was procured from the Central Pharmacy, Gauhati Medical College and Hospital, Guwahati, Assam, India.
Conflict of Interest
The authors declare no conflicts of interest relevant to this study.
Ethical Approval and Informed Consent
Written and oral informed consent was obtained from all participants prior to enrollment. The study protocol adhered to ethical guidelines and was approved by the Institutional Ethics Committee of Gauhati Medical College and Hospital (if applicable, add IEC reference number).
Data Availability
All data generated or analyzed during this study are included in this manuscript. No additional datasets were created or referenced.
Peer Review
This manuscript underwent external peer review via a double-blind process.
References
- Xu SF, Adams B, Yu XC, Xu M. Denosumab and giant cell tumour of bone—a review and future management considerations. Curr Oncol20 (2013):e442–7.
- Branstetter DG, Nelson SD, Manivel JC, et al. Denosumab induces tumor reduction and bone formation in patients with giant-cell tumor of bone. Clin Cancer Res18 (2012):4415–24.
- Amgen Inc. XGEVA® (denosumab): full prescribing information [Internet]. Thousand Oaks, CA: AmgenInc (2018).
- Thomas D, Henshaw R, Skubitz K, et al. Denosumab in patients with giant-cell tumour of bone: an open-label, phase 2 study. Lancet Oncol11 (2010):275–80.
- Chawla S, Henshaw R, Seeger L, et al. Safety and efficacy of denosumab for adults and skeletally mature adolescents with giant cell tumour of bone: interim analysis of an open-label, parallel-group, phase 2 study. Lancet Oncol 14 (2013):901–8.
- Uday S, Gaston CL, Rogers L, et al. Osteonecrosis of the jaw and rebound hypercalcemia in young people treated with denosumab for giant cell tumor of bone. J Clin Endocrinol Metab103 (2018):596–603.
- Agarwal MG, Gundavda MK, Gupta R, Reddy R. Does denosumab change the giant cell tumor treatment strategy? Lessons learned from early experience. Clin Orthop Relat Res476 (2018):1773–82.
- Palmerini E, Chawla NS, Ferrari S, et al. Denosumab in advanced/unresectable giant-cell tumour of bone (GCTB): for how long? Eur J Cancer76 (2017):118–24.
- Rutkowski P, Gaston L, Borkowska A, et al. Denosumab treatment of inoperable or locally advanced giant cell tumor of bone—multicenter analysis outside clinical trial. Eur J Surg Oncol44 (2018):1384–90.
- Martin-Broto J, Cleeland CS, Glare PA, et al. Effects of denosumab on pain and analgesic use in giant cell tumor of bone: interim results from a phase II study. Acta Oncol53 (2014):1173–9.
- Stopeck AT, Fizazi K, Body JJ, et al. Safety of long-term denosumab therapy: results from the open-label extension phase of two phase 3 studies in patients with metastatic breast and prostate cancer. Support CareCancer 24 (2016):447–55.
- Thomas DM. RANKL, denosumab, and giant cell tumor of bone. Curr Opin Oncol24 (2012):397–403.
- Branstetter DG, Nelson SD, Manivel JC, et al. Denosumab induces tumor reduction and bone formation in patients with giant-cell tumor of bone. Clin Cancer Res18 (2012):4415–24.
- Rutkowski P, Ferrari S, Grimer RJ, et al. Surgical downstaging in an open-label phase II trial of denosumab in patients with giant cell tumor of bone. Ann Surg Oncol(2015).
- Stadler N, Fingernagel T, Hofstaetter SG, Trieb K. A recurrent giant cell tumor of bone treated with denosumab. Clin Pract5 (2015):697.


 Impact Factor:
* 5.3
Impact Factor:
* 5.3
 Acceptance Rate:
73.64%
Acceptance Rate:
73.64%
 Time to first decision: 10.4 days
Time to first decision: 10.4 days
 Time from article received to acceptance:
2-3 weeks
Time from article received to acceptance:
2-3 weeks
