In Vivo Antileukemic Effect and Induction of Leukemia-Derived Dendritic Cells with Immunomodulatory agents in a rat AML model
Monica Cusan*,1, Michael Atzler1,2,†, Daniel Christoph Amberger1,2,†, Tobias Baudrexler1,2,†, Alexander Rabe1,2, Marit Inngjerdingen3, Christoph Schmid2,4 and Helga Maria Schmetzer*,1,2
1Medical Department III, Munich University Hospital LMU Munich, Germany
2Bavarian Cancer Research Center (BZKF), Munich, Germany
3Department of Immunology, Oslo University, Oslo, Norway
4Department of Hematology and Oncology, Section for Stem Cell Transplantation, Augsburg University Hospital and Medical Faculty, Augsburg, Germany
*Corresponding Authors: Monica Cusan, Medical Department III, Munich University Hospital LMU Munich, Germany.
Helga Maria Schmetzer, Medical Department III, Munich University Hospital LMU Munich, Germany.
†M.A., T.B. and D.C.A. contributed equally as second authors
Received: 19 February 2026; Accepted: 24 February 2026; Published: 30 March 2026
Article Information
Citation: Monica Cusan, Michael Atzler, Daniel Christoph Amberger, Tobias Baudrexler, Alexander Rabe, Marit Inngjerdingen, Christoph Schmid and Helga Maria Schmetzer. In Vivo Antileukemic Effect and Induction of Leukemia-Derived Dendritic Cells with Immunomodulatory agents in a rat AML model. Fortune Journal of Health Sciences. 9 (2026): 162-172.
View / Download Pdf Share at FacebookAbstract
Purpose: Effective therapies for patients with acute myeloid leukemia (AML) remain limited. Here we compare the immune-modulatory potential and anti-leukemic efficacy of the different combinations of responsemodifiers (rat GM-CSF with Prostaglandin E1 (Kit M), Picibanil (Kit I), and Prostaglandin E2 (Kit K)) in a rat leukemia model to convert leukemic myeloid blasts into leukemia-derived dendritic cells (DCleu), re-engaging both innate and adaptive immunity against leukemia and promoting immunological memory in vitro and in vivo, where Kit M was safely used to treat AML patients.
Methods: DC-generation and effect in mixed lymphocyte culture (MLC)-assays upon different treatments were performed. Brown Norway rats with leukemia were treated twice with Kit I, K, or M and compared to untreated controls. Nine days after treatment, blood and spleen samples were analyzed for blasts and immune cell populations.
Results: Kit treatment of leukemic rat-Whole blood (WB) generated DCleu; Kit-pretreated rat-WB with healthy rat T-Lymphocytes in MLC enhanced the antileukemic cytotoxicity. In vivo Kit-treatments were well tolerated. Kit M and Kit I(-high) resulted in a significant reduction of leukemic blasts. Kit I and Kit K displayed similar efficacy in inducing DCleu, memory-like T cells alongside with a reduction in regulatory T cells in all Kits, while Kit M uniquely promoted increased frequencies of memory-like CD8+CD62L++ T cells indicating induction of immunological memory.
Conclusion: DCleu-inducing in vivo Kits-treatment is safe and induces comparable effects, with Kit M being the most promising candidate for initiating targeted immune responses in AML as previously suggested.
Keywords
Leukemia-derived dendritic cells; GM-CSF; PGE1; immunomodulation; AML; antileukemic therapy; Kit M; rat model
Article Details
Introduction
Acute myeloid leukemia (AML)
Despite advances in treatment, the prognosis for patients with acute myeloid leukemia (AML) remains poor, as the relapse rate after initial therapy still stands at 70–80%. The outlook is especially grim for patients who do not respond to standard treatments or are unable to tolerate them (Tamamyan et al., 2018). The current standard of care consists of high-dose induction chemotherapy with cytarabine (possibly in combination with an anthracycline), followed by allogeneic hematopoietic stem cell transplantation (HSCT), which remains the only potentially curative option—primarily for younger patients with fewer comorbidities (Döhner et al., 2022; O'Donnell et al., 2017). For less fit patients, low-dose cytarabine or hypomethylating agents represent possible alternative strategies (Gil-Perez & Montalban-Bravo, 2019; Burnett et al., 2018). New immunotherapeutic approaches aim to reactivate the dysfunctional immune response against leukemic blasts including leukemia-derived dendritic cell induction, antibody-drug conjugates or T cell-recruiting antibody constructs (Ansprenger et al., 2020; Lichtenegger et al., 2017).
Dendritic cell-based immunotherapy
Dendritic cells (DCs) are crucial mediators between the innate and adaptive immune systems (Wan & Dupasquier, 2005; Palucka & Banchereau, 1999). They can be generated ex vivo from CD14+ monocytes and pulsed with leukemia-associated antigens (LAA), peptides, or mRNA. Alternatively, DCs can be directly derived from leukemic blasts, eliminating the need for antigen loading. These so-called leukemia-derived dendritic cells (DCleu) present the full spectrum of the patient’s individual leukemic antigens, including previously unidentified ones (Kremser et al., 2014), and can be reintroduced as a "vaccine" to stimulate the immune system (Van Acker et al., 2019). Another approach is to generate such DCleu directly in vivo using so-called "DCleu-inducing Kits," which are combinations of GM-CSF and a second response modifier (e.g., Picibanil, PGE1, or PGE2) that promote DC differentiation and maturation (Amberger et al., 2019; Schwepcke et al., 2022). To mimic the immune microenvironment in vitro, whole blood (WB) obtained from leukemia patients can be used for in vitro testing, since it contains both inhibitory and activating immunological components allowing a realistic model of the in vivo situation (Hirn Lopez et al., 2019). The composition and mechanism of action of the different Kits used in the present study are outlined in Table 1. In combination GM-CSF and PGE1 or PGE2 have proven to be potent ex vivo differentiation agents for DCs, as they induce danger signaling and enhance both the maturation and migratory capacity of these cells (Okamoto & Sato, 2003; Conejo-Garcia et al., 2016; Amberger et al., 2019; Schwepcke et al., 2022, Filippini Velazquez et al., 2025). From previous studies in vitro we investigated the efficacy and antiproliferative potential of different concentrations of GM-CSF, PGE1, OK-432/Picibanil and PGE2 by treating cells in WB from donors and leukemic patients, where we observed that all the combinations, but not the single compounds efficiently induced in a dose-dependent manner, the generation of dendritic cells in vitro from blood cells from healthy donors as well as from leukemia patients (Rejeski, HA et al 2025). In a further study, Kit M (GM-CSF + PGE1) was tested in experimental rats displaying high efficacy in generating DCs in vitro from healthy and leukemic samples WB resulting increased activated and memory T- cells from MLC in vitro and consistent antileukemic effect in cytotoxicity assay. Treatment of refractory AML human patients showed good tolerability and partial responses (Atzler et. al., 2024, Filippini Velazquez et al 2025).
Table 1: The different immunomodulatory combinations and their concentrations for the in vitro experiments with rat’s blood are listed in the table below.
|
Kit |
Composition |
Concentration |
Mode of Action |
Culture Time |
Reference |
|
Ilow |
rat GM-CSF + OK-432 |
800 U/ml (=1 mg/ml) 1 mg/ml |
(rat) GM-CSF: induction of myeloid (dendritic cell) differentiation |
7-8 days |
(Amberger et al., 2019) |
|
Ihigh |
rat GM-CSF + OK-432 |
800 U/ml (=1 mg/ml) 10 mg/ml |
OK-432: lysis product from Streptococcus pyogenes; stimulation of dendritic cell differentiation |
7-8 days |
|
|
K |
rat GM-CSF + PGE2 |
800 U/ml (=1 mg/ml) 1 mg/ml |
PGE1: Increase of CD197 expression and enhancement of dendritic cell migration |
7-8 days |
|
|
M |
rat GM-CSF + PGE1 |
800 U/ml (=1 mg/ml) 1 mg/ml |
7-8 days |
The aim of the present study was to further investigate better combinations of these promising Kits in in vitro and in an in vivo rat model to: (1) assess antitumoral and (2) immunomodulatory activity in vivo of different Kits by treating leukemic rats with different immunomodulatory Kits.
Materials and Methods
Leukemia model in rats
The leukemia model in rats Brown Norway Myeloid Leukemia (BNML) was used as previously described (Atzler et al., 2024). Briefly, seven-week-old, inbred BN/OrlRj rats were employed. This model is considered suitable for representing the promyelocytic subtype of AML in humans (Martens et al., 1990; Nestvold et al., 2008). Additionally, PVG.1N rats— a strain with the same MHC background as BN rats, but in which AML cannot be induced—were used as control for safety analyses of the compounds used for treatments. The experiments were approved by the Norwegian Animal Research Authority (NARA) under license numbers 12.4196 (in vitro) and 6060 (in vivo). BNML cell suspensions were extracted from the spleens of leukemia-bearing rats, washed, and cryopreserved at −80°C. Leukemia was induced in male rats via intravenous injection of approximately 8 million BNML cells into the penile vein. Within 23 days, the leukemic blasts had infiltrated various organs, most notably the bone marrow (94%), liver, spleen (88%), and peripheral blood (65%), replacing normal hematopoiesis. Leukemic cells were detectable in whole blood between days 15 and 17 after injection. Clinical symptoms included enlarged spleen and liver, weight loss, lethargy, and a rough coat. For blood sampling, rats were anesthetized using isoflurane inhalation. Weekly blood samples were taken from the tail vein. After 23 days, at the time of manifest leukemia, the rats were euthanized via CO2 inhalation, and cardiac blood and spleen cells were collected for flow cytometric analysis. All procedures—including leukemia induction, Kit administration, and sample collection—were carried out simultaneously across all animals.
Drug combinations
In order to exploit possible better combinations of the aforementioned drugs, following drugs combinations for in vitro testing were used: Kit I = GM-CSF + OK-432/Picibanil (using two different dosages of OK-432), Kit M = GM-CSF + PGE1 (established, Atzler et al. 2024), Kit K: = GM-CSF + PGE2. The in vitro concentrations of the drugs in the different Kits are indicated in Table 1.
In vitro immunomodulatory assays
Generation of DC/DCleu
Three different Kits (see Table 1) were used to generate DC/Dcleu from WB cells from leukemic rats ex vivo. Containing Approximatly 5 million (PBMNCs) in 12-well plates, diluted 1:1 with X-Vivo medium, and treated with one of the Kits mentioned above (Kit I, Kit M, or Kit K). Since Picibanil could induce strong reactions in vivo, different doses were used to assess efficacy and tolerability for the later use in vivo in rats. Cultures were incubated for 7–9 days at 37°C with 5% CO2. On day 3, the same concentrations of the respective substances were added again. Cells were harvested after 7 days and used for follow-up experiments (Schwepcke et al., 2022).
Mixed Lymphocyte Culture (MLC)
Healthy rats (n = 3) served as T-cell donors. CD3+ T cells were isolated using Lymphoprep (purity ~84%) and co-cultured with WB-derived cells from untreated or Kit-treated (DC/Dcleu containing) leukemic rats in a T cell enriched MLC. Following two stimulations with IL-2 (50 U/ml), immune cells were harvested after 6–8 days, and their cytotoxic activity was analyzed (see section 2.5).
In Vivo Experiments with Rats
Safety Analysis: Single Components in Healthy Rats
To investigate potential side effects of the individual components, three healthy PVG.1N rats were injected intravenously with single dose of the single drugs Picibanil (0.175 µg), PGE1 (1.2 µg), or PGE2 (1.2 µg). Each drug was prepared from a stock solution and diluted in 500 µl of PBS. A control group received only PBS. Rats were monitored for adverse effects (e.g., weight changes, skin reactions, breathing, behavior) at 2, 24, and 72 hours post-injection, and were then euthanized using CO2 for further analysis.
Treatment of Leukemic Rats
To assess the antileukemic effect of these drug combinations in vivo in the rat AML model, 14 days after AML induction diseased rats were divided into five groups (n = 3 rats per group) and treated using following details:
- Group 1: Kit I-low (1 µg ratGM-CSF + 0.175 µg Picibanil)
- Group 2: Kit I-high (1 µg ratGM-CSF + 0.35 µg Picibanil)
- Group 3: Kit M (1 µg ratGM-CSF + 1.2 µg PGE1)
- Group 4: Kit K (1 µg ratGM-CSF + 1.2 µg PGE2)
- Group 5: Control group (no treatment)
The injections were administered intravenously in 500 µl PBS, with a second dose given four days later. On day 24, the animals were euthanized. Blood and spleen samples were collected and analyzed by flow cytometry using standard procedures. The composition of the Kits is described in Table 1.
Flow Cytometry
After red blood cell lysis, WB samples were centrifuged and incubated with rat-specific monoclonal antibodies for 15 minutes on ice in the dark. Analysis was conducted using FACS instruments (Canto™, LSR Fortessa™). Gating strategies in the FACS analyses to define all the subpopulations of leukocytes and dendritic cells analyzed in the present work were performed as previously described (Atzler et al 2024). To verify successful DC/Dcleu generation, markers such as RM124, CD86, CD103, and MHC class II were used. T-, NK-, and NKT-cell subpopulations were also quantified (see Supplemental Table 1).
Cytotoxicity Assays
To assess the capacity of effector cells—T cell-enriched populations derived from healthy rat donors stimulated with or without Kits-treated WB following MLC to lyse target leukemic cells a cytotoxicity fluorolysis assay was performed. The lytic activity was determined by calculating the proportion of viable target cells remaining after co-culture with effector cells for 3 h and 24 h, relative to the control condition (Amberger et al., 2019). Alternatively, for the chromium release assay standard 4-hour 51Cr-release was used to assess cytotoxic activity as previously described (Kim et al., 2007).
Results
Efficient Ex Vivo Generation of DC/Dcleu from Leukemic Blood cells in rats
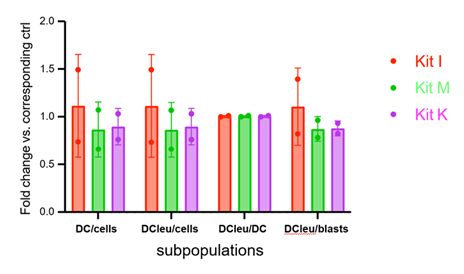
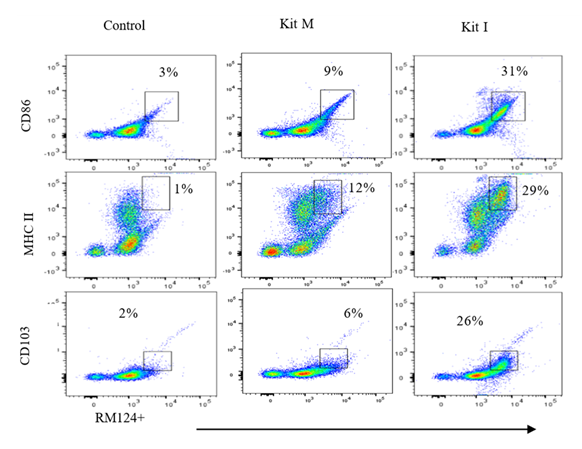
Appearance of a population of blasts in the peripheral blood of rats was observed from 2 weeks after transplantation (Supplemental Figure 1). Heparinized WB from leukemic Brown Norway rats (BNML model) from this time point was cultured ex vivo with Kits I, M or K. The gating strategies to identify the WBC subpopulations from WB of experimental rats by Flow Cytometrics was performed as previously described (Atzler et al. 2024). All tested Kits induced the formation of dendritic cells (DC) and leukemia-derived dendritic cells (Dcleu), with Dcleu frequencies ranging from 42.9-49.2%, of blood while in untreated controls the generation of DC was probably due to spontaneous generation (Figure 1A). Dcleu were identified by co-expression of the blast marker RM124 and DC markers such as CD86, MHC II and CD103 (Figure 1B). These findings demonstrate the efficient conversion of leukemic blasts into immunostimulatory Dcleu in vitro under stimulation with GM-CSF combined with different immune modulators with comparable proportions of modified cells with all Kits.
Figure 1A: Leukemia derived dendritic cells could be generated ex vivo from whole blood (WB) of leukemic rats by performing cultures of blast containing whole blood (WB) from rats with different Kits (Kit M, K and I, n=2). Percentage of DC and DCleu within DC and blast cells in the different conditions are indicated compared to control (ctrl). Mean results ± standard deviation of DC subtype frequencies obtained from each two rats are given.
Figure 1B: Representative measurement of immunophenotyping showing increased frequencies of DC subtypes from whole blood after culture of rats´ blood with Kit M and Kit I compared to Control. RM124 and MCH II / CD103 / CD86 expressing cells define the DC subpopulations. Squares indicate the double positive populations according to the unstained controls. Increased frequencies of DC (subtypes) after culture of rats WB with Kit M and Kit I compared to control are shown.
Enhanced Anti-Leukemic Reactivity After T Cell Priming in vitro
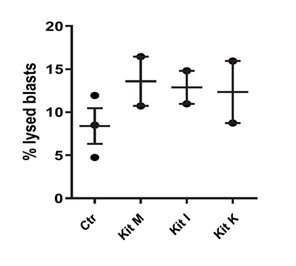
To assess functional immune activation, T cell-enriched MLCs were established using WB from rats pretreated with each Kit and T lymphocytes from healthy rats. All Kit-treated samples elicited enhanced cytotoxic activity against leukemic target cells, as measured by chromium release assays, indicating that Kit-generated DC/DCleu are capable of priming effective anti-leukemic T cell responses. Highest blast lysis was achieved by Kit M pretreated samples (Figure 1C).
Figure 1C: Antileukemic blast lytic activity was demonstrated using a chrome release assay for Kit pretreated rat WB samples upon MLC using T cell from healthy untreated rat donors. Average frequencies ± standard deviation of lysed blasts are given. Abbreviations for cell subtypes are given in Table 3. Immune cells from healthy mice primed with Kit pretreated (vs. untreated) WB achieved higher blast lysis after T cell enriched MLC in comparison to the control.
In Vivo Application of Kits is Safe in Healthy and Leukemic Rats
Healthy PVG.1N rats received single injections of individual Kit components (Picibanil 0.35 mg, PGE1 1mg, or PGE2 1mg), diluted in 500 ml PBS to evaluate safety of the drugs. No adverse effects—such as behavioral changes, weight loss, skin irritation, or altered respiration—were observed in all the rats groups after 2h, 24h and 72 hours post-injection. Similarly, after double treatment of BNML rats (on day 14 and 17 after AML induction with leukemic cells) with Kits I, K, or M, animals did not show adverse events, thereby supporting the tolerability of these combinations even in the presence of disease, as shown before (Atzler et al. 2024).
Kit Treatment In Vivo Reduces Leukemic Burden and Modulates Immune Cell Populations
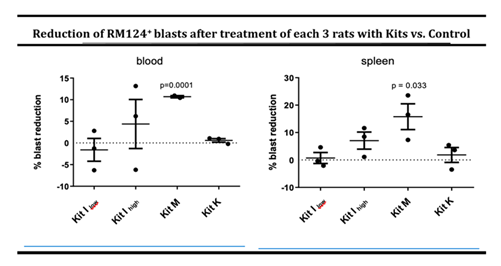
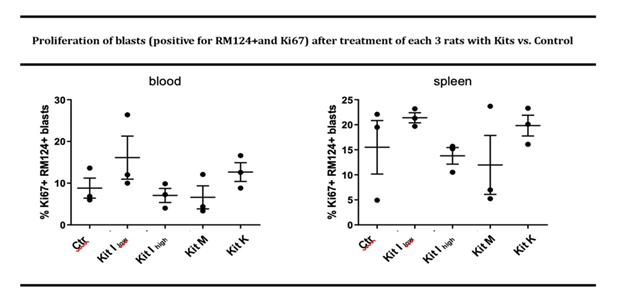
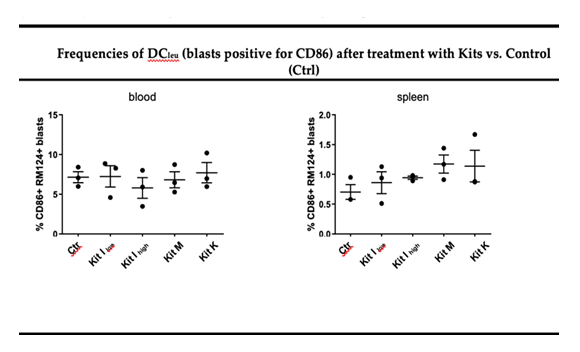
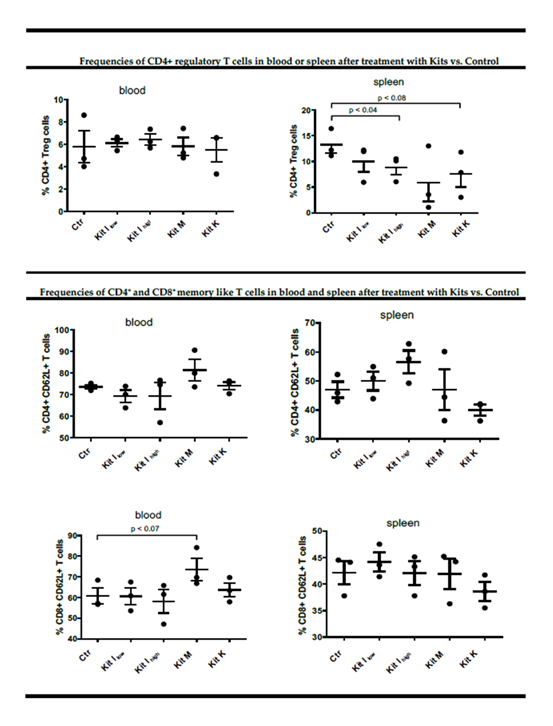
Leukemic BNML rats were treated twice (on day 14 and 17 after AML induction) with Kit I-low, I-high, K, or M, and analyzed after sacrifice on day 23. Control leukemic mice remained untreated (Atzler et al 2024). Kit M and Kit I-high resulted in a significant reduction of leukemic blasts in both peripheral blood (PB) and spleen compared to untreated controls (Kit M: PB: 10.7%, p<0.0001; spleen: 15.8%, p<0.033), while Kit I-low and Kit K did not change frequencies of blasts in comparison to the untreated controls (Figure 2A). Compared to untreated control rats frequences of proliferating (Ki67+) blasts were decreased only upon Kit I-high and Kit M treatment, but not under Kit I-low or Kit K treatment (Figure 2B). Furthermore, treatments increased the frequencies of DCleu measured in the blood especially of the rats treated with Kit M and Kit K, and more clearly in the spleen from the rats treated with all the Kits in comparison to the untreated control (Figure 2C). Next, we analyzed the changes on the endogenous immune cells of the recipient rats. Regulatory T cells (Tregs) were reduced, particularly in the spleen following treatment with Kits I, K, and M (Figure 3, upper panels). Kit M uniquely promoted increased frequencies of memory-like CD4+CD62L+ and CD8+CD62L+ T cells from the blood of treated rats, indicating the potential induction of immunological memory (Figure 3, lower panels). Kit I-high induced an increase in CD4+CD62L+ T cells memory cells in the spleen of treated mice in comparison to the control (Figure 3, lower panels). No significant changes were observed in natural killer (NK) or natural killer T (NKT) cell frequencies (data not shown). Collectively, these results indicate that in vivo Kit application—particularly Kit M and Kit I-high—successfully reduces leukemic burden and fosters an immune-activating environment without inducing blast proliferation. In particular, upon the more effective treatments, the CD4+ regulatory T cells were reduced, while CD4+ and CD8+ memory T cells displayed a trend toward increase upon treatment with Kit I and M, suggesting the induction of immune memory in leukemic animals upon treatments.
Figure 2A: Percentage of reduction of blasts in the blood and spleens of treated rats (each 3 rats per group) in comparison to the untreated control is indicated. The indicated p values are calculated for Kit M in comparison to control. The other treatments were not significant. N=3. Given are mean values ± standard deviation and differences were defined as significant with p values <0.05. Abbreviations for cell subtypes are given in Table S1.
Figure 3: Treatment of leukemic rats with Kits (especially Kit M and I-high) induced DC/DCleu as well as activated immunoreactive and memory-like T cells and reduced regulatory cells compared to control in PB or spleen. The composition of cells in PB and spleen was quantified by flow cytometry. In top 2 plots frequencies of CD4+ regulatory T cells in blood and spleen of treated rats are shown: the strongest reduction was observed upon treatment with Kit M. In the middle and lower plots the proportions of CD4+ and CD8+ memory like T cells, respectively, are shown: Kit M was the strongest in increasing the CD8+ T cells as well as the CD4+ T cells in the blood, while Kit K reduced their frequencies mostly in the spleen. Data were statistically analyzed using a paired t test., n=3. Given are mean values ± standard deviation and differences were defined as significant with p values <0.05. (Abbreviations for cell subtypes are given in Table S1).
Discussion
DC/DCleu Generating and Blast Modulating Protocols (Kits) Lead to Antileukemic Responses in Vivo
Treating AML patients with ex vivo produced DC (loaded with leukemia associated antigens) or with DCleu has been shown to stabilize the disease/remission. These DC/DCleu must be produced under good manufacturing practice (GMP) conditions, followed by an adoptive transfer to patients (Amberger et al., 2020, Van Acker et al., 2019). An elegant solution to improve these DC-based strategies is to directly convert (residual) blasts in the patients in vivo to DCleu resulting in activation of immune cells as previously described (Atzler et al. 2024, Filippini Velazquez et al 2025). We and others could prove that DC/DCleu can be generated ex vivo from blasts containing MNC samples, and also from whole blood and bone marrow without inducing blast proliferation on a regular basis using different DC/DCleu generating protocols and Kits (Schwepcke et al., 2022, Plett et al., 2022, Unterfrauner et al., 2023). The generation process is independent of patients’ characteristics like age, sex, disease/prognostic classifications and have already shown promising results as they are able to induce leukemia-specific, anti-leukemic cells after T cell enriched MLC (Klauer et al., 2021 and 2025). In this manuscript we show that DC/DCleu can also be successfully generated from leukemic rat WB without induction of blasts’ proliferation using different Kits ex vivo. An astonishing finding was, that nearly all the created DC/DCleu were leukemia-derived pointing to a highly efficient leukemic antigen-presentation and leading to leukemia-cytotoxicity of stimulated effector cells. In addition, blast lysis could be improved with all Kit-pretreated WB-samples used as stimulator cells in MLC, as already shown for human AML-samples (Schwepcke et al., 2022, Plett et al., 2022).
Treatment of Leukemically Diseased Rats with All Kits Was Well Tolerated, Reduced Regulatory T Cells and Induced Antileukemic Cells (Kit M and I)
Treatment of leukemically diseased rats’ WB with Kits showed a successful DC/DCleu generation with Kit I, K, M, in line with the improved blast lysis after MLC with rats’ T cells in chrome release assay shown in vitro. These data confirm that combinations of (rat) GM-CSF with a second response modifiers results in an improved (ex vivo) DC/DCleu triggered antileukemic immune response (without induction of blast proliferation) as already shown with leukemic human WB before (Unterfrauner et al., 2023). Therefore, a rat model qualifies for in vivo treatment strategies with Kits as proposed here: Leukemia diseased or healthy rats tolerated treatment with single or combined response modifiers very well (no changes in behavior, respiration, weight, skin, etc.), pointing to tolerable and safe application of Kits to (human) patients. For optimal comparison of results treatment of all rats was started on the same day and all rats were sacrificed on the same day after treatment. Leukemia diseased rats showed improved hematological and immunological effects after treatment with Kits. The most important findings were, that despite of high blast counts in rats’ blood and spleen at start of treatment a significant reduction of blast was seen after only two applications of Kit M (vs. untreated rats). This points to the activation of leukemia-specific effector cells -leading to blast lysis (as seen in rats’ blood and in various human WB models ex vivo) (Rackl et al., 2023). A clear reduction of blasts was also seen after treatment of rats with Kit I-high, but not with Kit I-low. This could point to the need of two response modifiers in ‘sufficient’ dosage (as provided in Kit M or Kit I-high, but not with Kit I-low, that contains only standard GM-CSF and low Picibanil concentrations). This might also confirm earlier clinical data of our group, showing that a treatment of therapy refractory AML patients at relapse after SCT with only GM-CSF did not result in improved outcome of patients compared to control patients without GM-CSF treatment (Schmid et al., 2004). Treating rats with Kit K was not shown to reduce blasts in vivo. Compared to control animals in all Kit treated rats a reduction of regulatory T cells in spleen, but not in blood was seen.
This could point to processes leading to reactivated antileukemic responses in vivo, as already shown with specifically induced reactivations against blasts in leukemic patients’ WB samples ex vivo (Pepeldjiyska et al., 2022). We found nearly significantly increased frequencies of memory like T cells after treatment of rats with Kit M (but not with other Kits). This might point to an induced immunological memory against blasts in vivo, although prove for ‘leukemia specificity’ is missing (due to missing data using a leukemia specific functional assay). With respect to our ex vivo WB data with human WB we however suppose that leukemia specific cells could have been induced (Rackl et al., 2023, Filippini Velazquez et al., 2025).
Conclusions
In summary, we could show that different Kits (containing clinically approved drugs) produce DC/DCleu ex vivo and in vivo in leukemically diseased rats. Compared to controls, we have demonstrated improved anti-leukemic responses ex vivo and in vivo. Kit M (followed by Kit I-high) as rescue therapy was shown to be the most effective and safe treatment option within the combinations used here, where it induced the most significant reduction of blasts in blood and spleen of leukemic rats after treatment. At the same time, immune cells of the adaptive and innate lines were activated and induced to give rise to memory- as well as to leukemia-specific/antileukemic cells. Our results here support the use of the most effective combination of GM-CSF and PGE1for treatment of AML. Although further experiments and treatments (phase I trial is in preparation) are required, this in vivo strategy aiming at the conversion of (residual) blasts to leukemia-derived DC (without need of GMP procedures) appears to be promising, since DC/DCleu can migrate to tissues and prevent (extramedullary) relapses. DC/DCleu-generation is independent of patients’ age, MHC, mutation, cytogenetic risk, transplant, or FAB status and could contribute to stabilization of the disease or of remissions by induction of antileukemic cells and immunological memory.
We show promising results in the field of immunotherapy for AML, that could contribute to stabilize the disease or remission with a well-tolerated therapy. Myeloid blasts can inhibit the efficacy of antileukemic T cells by a variety of mechanisms, e.g. by upregulation of immune checkpoints or inhibition of T cell metabolism (Sauerer et al 2023). Hence the combination with other strategies controlling the leukemic proliferation or the application in situation with less proliferative activity of the leukemia (e.g. in high-risk myelodysplastic syndrome or hypocellular AML) might lead to more sustained clinical responses. Once clinical safety of the approach has been shown in a phase I trial, clinical efficacy will be tested in a larger cohort. Thereby, we hope to make a valuable novel contribution to the armamentarium of treatments for highly aggressive blood cancers.
Acknowledgments: The authors thank technicians for their support with sample materials and experiments. The results presented in this manuscript are part of the doctoral thesis of Atzler Michael, Baudrexler Tobias, Deen Diana and Schutti Olga at the University Hospital of Ludwig-Maximilian-University Munich.
Funding: The whole project was funded by intramural funding of the participant partners and their institutions (University of Oslo, University of Augsburg, University of Munich). The funders had no role in the study design, data collection or analysis, decision to publish or preparation of the manuscript.
Conflicts of Interest: Modiblast Pharma GmbH (Oberhaching, Germany) holds the European Patent 15 801 987.7-1118 and US Patent 15-517627 ‘Use of immunomodulatory effective compositions for the immunotherapeutic treatment of patients suffering from myeloid leukemias’, with whom Schmetzer H. is involved with.
Author Contributions: Cusan M., Atzler M., Baudrexler T., Amberger D. performed DC-, MLC-, CTX-experiments, flowcytometric and statistical analyzes of cell biological data. Rogers N. conducted CSA experiments. Inngjerdingen M. and Rabe A. were responsible for rat experiments. Schmetzer H. designed the study together with Schmid C.. Cusan M., Atzler M., Baudrexler T., Amberger D.C., Schmid C. and Schmetzer H. drafted this manuscript.
Ethics declaration: the authors ensure that the present work has not been submitted to other journals, that the work is original, and that the data has not been presente din any other published work. Good practice rules in science, basic research and animal research have been followed according to institutions rules and national law.
References
- DC Amberger, F Doraneh-Gard, C Gunsilius, et al. PGE1-containing protocols generate mature (leukemia-derived) dendritic cells directly from leukemic whole blood. International Journal of Molecular Sciences 20: (2019): 4590
- DC Amberger & HM Schmetzer. Dendritic cells of leukemic origin: Specialized antigen-presenting cells as potential treatment tools for patients with myeloid leukemia. Transfusion Medicine and Hemotherapy 47: (2020) 432–443.
- C Ansprenger, DC Amberger & HM Schmetzer. Potential of immunotherapies in the mediation of antileukemic responses for patients with acute myeloid leukemia (AML) and myelodysplastic syndrome (MDS). Clinical Immunology 217: (2020) 108467.
- M Atzler, T Baudrexler, DC Amberger, et al. In vivo induction of leukemia-specific adaptive and innate immune cells by treatment of AML-diseased rats and therapy-refractory AML patients with blast modulating response modifiers. Int J Mol Sci 25 (2024): 13469.
- AK Burnett, RK Hills, O Nielsen, et al. A comparison of FLAG-Ida and daunorubicin combined with clofarabine in high-risk acute myeloid leukaemia. Leukemia 32 (2018): 2693–2697.
- JR Conejo-Garcia, MR Rutkowski & JR Cubillos-Ruiz. State-of-the-art of regulatory dendritic cells in cancer. Pharmacology & Therapeutics 164 (2016): 97–104.
- G Filippini Velázquez, P Anand, J Abdulmajid, et al. Clinical stabilization of a highly refractory acute myeloid leukaemia under individualized treatment with immune response modifying drugs by in vivo generation of dendritic cells of leukaemic origin (DCleu) and modulation of effector cells and immune escape mechanisms. Biomarker Research 13 (2025): 104.
- H Döhner, AH Wei, FR Appelbaum, et al. Diagnosis and management of AML in adults. Blood 140: (2022): 1345–1377.
- A Gil-Perez & G Montalban-Bravo. Management of myelodysplastic syndromes after failure of response to hypomethylating agents. Therapeutic Advances in Hematology 10 (2019): 2040620719847059.
- A Hirn Lopez, D Deen, Z Fischer, et al. Schmetzer. Role of interferon (IFN)α in cocktails for the generation of leukemia-derived dendritic cells. Journal of Immunotherapy 42 (2019): 143–161.
- GG Kim, VS Donnenberg, AD Donnenberg, et al. A novel multiparametric flow cytometry-based cytotoxicity assay. Journal of Immunological Methods 325 (2007): 51–66.
- LK Klauer, O Schutti, S Ugur, et al. Interferon gamma secretion of adaptive and innate immune cells. Transfusion Medicine and Hemotherapy 49 (2021): 44-61.
- LK Klauer, HA Rejeski, S Ugur, et al. Leukemia-Derived Dendritic Cells Induce Anti-Leukemic Effects Ex Vivo in AML Independently of Patients' Clinical and Biological Features. Int J Mol Sci 26 (2025): 1700.
- A Kremser, J Dreyig, C Grabrücker, et al. Dendritic cells can be successfully generated from leukemic blasts. Journal of Immunotherapy 33 (2010): 185–199.
- FS Lichtenegger, C Krupka, S Haubner, et al. Subklewe. Recent developments in immunotherapy of acute myeloid leukemia. Journal of Hematology & Oncology 10 (2017): 142.
- Martens, D. Van Bekkum & A. Hagenbeek. The BN acute myelocytic leukemia (BNML). Leukemia, 4, (1990): 241–257.
- JM Nestvold, BK Omdal, K-Z Dai, et al. A second prophylactic MHC-mismatched bone marrow transplantation. Transplantation 85 (2008): 102–111.
- MR O'Donnell, MS Tallman, CN Abboud, et al. Coutre. Acute myeloid leukemia, version 3.2017. Journal of the National Comprehensive Cancer Network 15 (2017): 926–957.
- M Okamoto & M Sato. Toll-like receptor signaling in anti-cancer immunity. Journal of Medical Investigation 50 (2003): 9–24.
- K Palucka & J Banchereau. Dendritic cells: A link between innate and adaptive immunity. Journal of Clinical Immunology 19 (1999): 12–25.
- E Pepeldjiyska, L Li, J Gao, CL Seidel, et al. Leukemia-derived dendritic cell mediated immune response. Immunobiology 227 (2022): 152237.
- C Plett, LK Klauer, DC Amberger, et al. Schmetzer. Immunomodulatory kits generating leukemia-derived dendritic cells. Clinical Immunology 242 (2022): 109083.
- E Rackl, L Li, LK Klauer, et al. Dendritic cell-triggered immune activation. International Journal of Molecular Sciences 24 (2023): 463.
- J Rosenblatt, RM Stone, L Uhl, et al. Individualized vaccination of AML patients. Science Translational Medicine 8 (2016): 368ra171.
- T Sauerer, GF Velázquez & C Schmid. Relapse of acute myeloid leukemia after allogeneic stem cell transplantation. Molecular Cancer 22 (2023): 180.
- DA Schillingmann, SB Riese, V Vijayan, et al. Eiz-Vesper. Inhibition of heme oxygenase-1 activity. International Journal of Molecular Sciences 20 (2019): 482.
- C Schmid, M Schleuning, J Aschan, et al. Low-dose ARAC, donor cells, and GM-CSF. Leukemia 18 (2004): 1430–1433.
- C Schwepcke, LK Klauer, D Deen, et al. Schmetzer. Generation of leukemia-derived dendritic cells. International Journal of Molecular Sciences 23 (2022):
- T Simmet & BA Peskar. Prostaglandin E1 and arterial occlusive disease. European Journal of Clinical Investigation 18 (1988): 549–554.
- K Spiekermann, J Roesler, A Emmendoerffer, et al. Functional features of neutrophils induced by G-CSF and GM-CSF. Leukemia 11 (1997): 466–478.
- CP Stahl, EF Winton, MC Monroe, et al. Evatt. Differential effects of interleukin-3 and GM-CSF (1992).
- G Tamamyan, H Tervonen, L Mendoza, et al. Future of global cancer. Journal of Global Oncology 4 (2018): 74s–74s.
- M Unterfrauner, HA Rejeski, S Bohlscheid, et al. GM-CSF combined with prostaglandin E1. International Journal of Molecular Sciences 24 (2023): 17436.
- HH Van Acker, M Versteven, FS Lichtenegger, et al. Dendritic cell-based immunotherapy of AML. Journal of Clinical Medicine 8 (2019): 579.
- VF Van Tendeloo, A Van De Velde, A Van Driessche, et al. WT1 antigen-targeted dendritic cell vaccination. Proceedings of the National Academy of Sciences 107 (2010): 13824–13829.
- Wan & M. Dupasquier. Dendritic cells in vivo and in vitro. Cellular & Molecular Immunology (2005).
- M Yanada & T Naoe. Acute myeloid leukemia in older adults. International Journal of Hematology 96 (2012): 186–193.
Supplemental Table 1: Subtypes of leukemic blasts, DC and DCleu, adaptive and innate immunity cell subsets in rats’ blood.
|
Cells types |
Name of Subgroups |
Surface Marker |
Referred to |
Abbreviation of cell population |
Reference |
|
|
Blast cells |
Leukemic blasts |
RM124+ |
WB |
Bla/WB |
Martens et al., 1990; Atzler et al., 2024 |
|
|
Proliferating blasts |
Bla+Ki67+ |
WB |
Bla_prol/WB |
Martens et al., 1990; Atzler et al., 2024 |
||
|
Dendritic cells |
Dendritic cells |
DC e.g. CD86+, CD103+ |
WB |
DC/WB |
Plett et al., 2022; Atzler et al., 2024 |
|
|
Leukaemia derived DC |
DC+Bla+ |
WB |
DC_leu/WB |
Plett et al., 2022; Atzler et al., 2024 |
||
|
MHC II |
MHC II |
WB |
MHC+Bla/WB |
Martens et al., 1990; Plett et al., 2022 |
||
|
Adaptive Immune system |
T lymphocytes |
CD3+ pan T cells |
CD3+ |
WB |
CD3+/WB |
Amberger et al., 2019; Atzler et al., 2024 |
|
CD4+ coexpressing T cells |
CD3+CD4+ |
CD3+ |
CD4+CD3+/CD3+ |
Amberger et al., 2019 |
||
|
CD8+ coexpressing T cells |
CD3+CD8+ |
CD3+ |
CD8+T/CD3+ |
Amberger et al., 2019 |
||
|
Memory like T cells |
CD3+CD4+CD62L++ |
CD3+ |
CD4+Tmem/CD3+ |
Waidhauser et al., 2021 |
||
|
Memory like T cells |
CD3+CD4-CD62L++ |
CD3+ |
CD8+Tmem/CD3+ |
Waidhauser et al., 2021 |
||
|
Regulatory T cells |
CD3+CD4+Foxp3+CD25+ |
CD3+ |
CD4+Treg/CD3+ |
Waidhauser et al., 2021 |
||
|
Innate Immune system |
Natural killer cells |
CD3-CD56+ NK cells |
NKR-P1A+ CD3- |
lymphocytes |
NK cell/cells |
Nestvold et al., 2008; Atzler et al., 2024 |
|
Natural killer T cells |
6B11+ NKT cells |
NKR-P1A+ CD3+ |
lymphocytes |
NK/T cell/cells |
Nestvold et al., 2008 |
Abbreviations: WB whole blood; MHC II major histocompatibility complex type II.
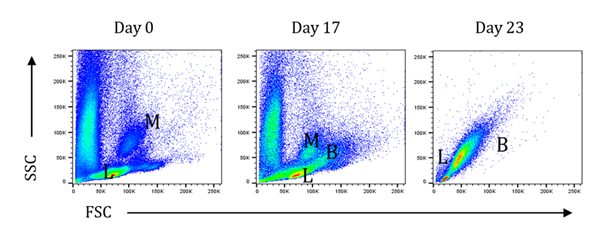
Suppl. Figure 1A: Development of the leukemia in the BNML rats from day 0 to day 23 after injection of leukemic cells. In the right plot displacement of healthy cells (monocytes, granulocytes, and lymphocytes) by an increasing blast population after injection of leukemic cells to rats over 23 days. (B=blasts, M=monocytes, L=lymphocytes). Y axis indicates SSC parameter.


 Impact Factor:
* 6.2
Impact Factor:
* 6.2
 Acceptance Rate:
76.33%
Acceptance Rate:
76.33%
 Time to first decision: 10.4 days
Time to first decision: 10.4 days
 Time from article received to acceptance:
2-3 weeks
Time from article received to acceptance:
2-3 weeks
