Endoscopic Evacuation of Deep Intracerebral Haemorrhage Versus Medical Treatment – A Retrospective Cohort Study
Kasper Riis Jepsen1*#, Malthe Thuesen Stephansen2#, Stefanie Binzer1, Sune Munthe1, Frantz Rom Poulsen1, Christian Bonde Pedersen1
1Odense University Hospital, Odense, Denmark
2Aarhus University Hospital, Aarhus, Denmark
#Authors contributed equally to the publication
*Corresponding Authors: Kasper Riis Jepsen, Odense University Hospital, Odense, Denmark
Received: 18 February 2026; Accepted: 24 February 2026; Published: 16 April 2026
Article Information
Citation: Kasper Riis Jepsen, Malthe Thuesen Stephansen, Stefanie Binzer, Sune Munthe, Frantz Rom Poulsen, Christian Bonde Pedersen. Endoscopic evacuation of deep intracerebral haemorrhage versus medical treatment – A retrospective cohort study. Journal of Surgery and Research. 9 (2026): 158-164.
View / Download Pdf Share at FacebookAbstract
Purpose: Spontaneous deep supratentorial haemorrhage is associated with high morbidity and mortality. There is limited evidence on surgical treatment possibilities. This study evaluates the effect of acute minimally invasive surgery (MIS) using the Artemis neuro evacuation device versus medical management alone in patients with spontaneous deep intracerebral haemorrhage (ICH).
Methods: Eleven patients with spontaneous ICH who underwent acute endoscopic surgery with the Artemis system were matched 1:1 with nonsurgical patients, resulting in a total of 22 patients in the study.
Results: The 30-day mortality was 18% in the surgical group and 36% in the non-surgical group (RR=0.5; [95%CI = 0.11;2.19]). Overall mortality was 18% for the surgical group and 45% for the non-surgical group (RR=0.4; [95%CI = 0.10;1.64]). The mean modified Rankin Scale score was 3.82 for the surgical group and 4.45 for the non-surgical group (P=0.3761).
Conclusion: Surgery using the Artemis neuro evacuation device showed a tendency to reduce 30-day mortality in patients with spontaneous basal ganglia intracerebral haemorrhage. These results support the need for a randomized controlled trial of MIS using the Artemis system compared to medical management alone.
Keywords
Intracerebral haemorrhage, Endoscopic surgery, Minimal invasive surgery, Mortality; Neurology, Neurosurgery
Article Details
Introduction
Intracerebral haemorrhage (ICH) accounts for 15% of all strokes [1], and over five million ICH events occur every year worldwide [2]. As 30-day mortality is more than 40% and only few of the surviving patients gain functional independence, it is a disease with a poor prognosis and a high socioeconomic impact [3,4].
Despite research in the field, evidence on medical and surgical treatment of ICH is limited. Outcome is improved by care in a stroke unit [5,6] and early antihypertensive drugs [6-9], but other treatments have not proven effective.
The medical treatment for Danish patients with spontaneous ICH includes early intensive blood pressure control to achieve systolic blood pressure below 140 mmHg and reversal of anticoagulants. The surgical approach aims to reduce mortality and prevent further permanent tissue damage due to brain ischaemia. Deep intracerebral haemorrhages localized in the basal ganglia or thalamus are a surgical challenge as craniotomy for evacuation of the haematoma often causes iatrogenic damage to healthy brain tissue [10]. No surgical approach has yet shown significant improvement in clinical outcome in patients with a deep supratentorial ICH, but surgery is still frequently used [11,12].
The haemorrhage of an ICH causes cytotoxic and mechanical irritation of the surrounding tissue [13]. This inflammation leads in turn to oedema, which elevates the intracranial pressure [14] and increases the risk of permanent cerebral damage. The conventional surgical approach for deep ICH does not alter the cytotoxic impact but increases the mechanical damage, leading to prolonged intensive care treatment after surgery.
Studies indicate that minimally invasive surgery (MIS) improves patients’ functional outcome compared to craniotomy [15,16], and it is a safe and effective treatment for spontaneous ICH located in the basal ganglia [15]. The Artemis neuro evacuation device, which combines endoscopy and vibrating aspiration, is a minimally invasive endoscopic tool for removing haematomas. It has the potential to reduce the mechanical impact to healthy brain tissue while removing the haemorrhage, hence reducing the length of intensive care treatment and enabling earlier neurorehabilitation.
In this retrospective cohort study, we aimed to evaluate the effect of minimally invasive surgery using the Artemis neuro evacuation device in patients with deep spontaneous intracerebral haemorrhage, compared to medical management alone.
Methods
Study design
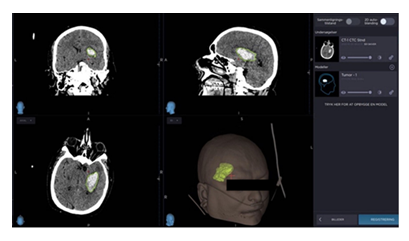
In this retrospective cohort study, we investigated the role of image-guided endoscopic evacuation with the Artemis neuro evacuation device in conjunction with neuro navigation in patients with deep supratentorial ICH with a blood volume of minimum 15 ml.
Twenty-two patients were enrolled in the study, with 11 patients in the surgical group and 11 in the non-surgical group.
Patient selection
Surgical group
All patients who had undergone minimal invasive surgery with the Artemis system at the neurosurgical department at Odense University Hospital between 29 April 2020 and 11 April 2021and met the inclusion criteria were retrospectively enrolled in the study.
Inclusion criteria were patients ≤80 years old who had arrived at the hospital within 8 hours of ictus with a National Institutes of Health Stroke Scale (NIHSS) score ≥2, a Glasgow Coma Scale (GCS) score ≥8, and a haemorrhage of minimum 15 ml that was located ≥2 cm from the cortical surface.
Exclusion criteria were vascular malformations (such as arteriovenous malformation, aneurysms, and dural arteriovenous malformation), any other previous neurosurgical treatment than minimally invasive surgery, and malignant brain tumours or other malignant diseases.
Non-surgical group
All patients admitted to the neurological department at Kolding Hospital from August 2018 to September 2021 with a primary diagnosis of ICH (using the International Classification of Diseases-10 (ICD10) coding system (DI610 – DI619)) and a confirmed ICH on computed tomography (CT) or magnetic resonance imaging (MRI) were assessed for eligibility according to the inclusion and exclusion criteria. Patients who met the inclusion criteria were enrolled into the non-surgical group.
Variables
We reviewed the patients’ hospital notes for data on age, sex, body mass index (BMI), comorbidities, GCS at admission, alcohol intake, and smoking status. Data were measured on ICH localization, midline-shift (in mm), intraventricular extension, length, height, width, and volume measured by 3D-rending using a Medtronic Stealth device. An example of a 3D model is shown in figure 1.
Matching and data management
The patients in the surgical and non-surgical groups were matched 1:1 using GCS, ICH volume, intraventricular extension, and age. In the case of more than one possible match, we used the 'nearest neighbour’ approach where the patients were secondarily matched on BMI, gender, smoking status, alcohol intake, and date of ICH onset. All data were stored in SharePoint.
Medical and surgical treatment
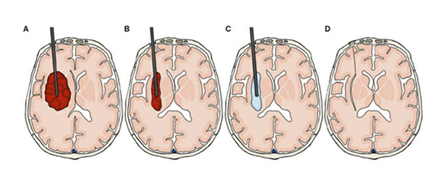
All patients received early intensive medication to lower the systolic blood pressure to ≤140 mmHg, according to established treatment regimens [17]. Patients in the surgical group were furthermore treated with the Artemis neuro evacuation device. These surgeries were performed by the same neurosurgeon at Odense University Hospital. The procedure was performed under general anaesthesia, and the patients were admitted to a neurointensive care unit after surgery. Figure 2 illustrates the endoscopic procedure step by step.
The procedure is performed by drilling a burr hole in the frontal area of the skull for insertion of the endoscope. As shown in figure 2A, the endoscope is then inserted in the brain via the burr hole under neuronavigation. When the endoscope has reached the posterior part of the haematoma, the surgeon starts a vibrating aspiration of the haematoma to relieve the pressure from the surrounding tissue (Figure 2B). When part of the haematoma has been aspirated and the initial pressure is relieved, the cavity is rinsed in sterile salt water under simultaneous aspiration (Figure 2C). The endoscope is retracted when the haematoma has been removed or when satisfactory volume reduction has been achieved and the water is aspirated (Figure 2D). Finally, the burr hole is covered with a small titanium net for cosmetic reasons.
Outcome measures
The primary outcome of this study was 30-day mortality. Secondary outcomes were overall mortality and modified Rankin scale (mRS) score. Follow-up was performed between 30 November 2021 and 3 December 2021. Mortality data was obtained in the patients’ hospital notes. The mRS is a single-item scale to measure the degree of a patient’s disability or dependence in daily activities. The mRS scores range from 0 (no disability) to 5 (severe disability) and 6 (death). To obtain the mRS at follow-up, the surviving patients were contacted by telephone, and the clinician scored the mRS based on the patient’s feedback. Mean follow-up time (from ICH to mRS assessment) was 382 days (234 to 581) for the surgical group and 672 days (202 to 1146) for the non-surgical group.
Statistical analyses
Binary data, including mortality data, are presented as percentages. Continuous data are presented as means and standard deviations (±SD).
Differences in mortality between surgical and non-surgical groups were assessed using chi-squared test and evaluated using relative risk (RR) with 95% confidence intervals (CIs).
Numeric data were measured using student’s t-test.
An mRS analysis was made for all patients included in the study. A separate analysis of the mRs scores was made for surviving patients only, to investigate the patients’ chance of gaining independence. A p-value ≤0.05 was considered significant. Statistical analyses were performed with Microsoft Excel v. 16.54.
Results
Trial population
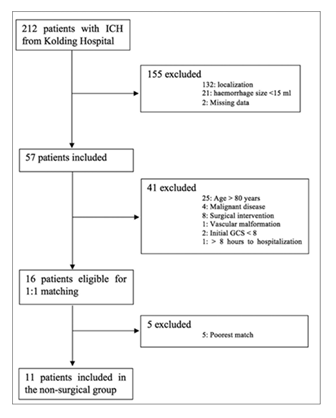
Thirteen patients underwent MIS using the Artemis system in Odense from April 2020 to August 2021. One patient was excluded due to vascular malformation, and one patient was excluded due to initial GCS <8, giving 11 patients in the surgical group.
Of the 212 patients who were admitted with ICH and received medical treatment, 155 were initially excluded because the haemorrhage was too superficial or ICH volume was <15 ml or due to missing data. A further 57 patients were excluded due to age, other conditions, initial GCS <8, or >8 hours from ICH onset to hospitalization. This left 16 patients eligible for matching. The procedure is shown in figure 3.
We matched the 11 surgical patients with the 16 non-surgical patients 1:1 to give 11 patients. The patients’ demographic and clinical data are shown in table 1.
|
Surgical group (N=11) |
Non-surgical group (N=11) |
P-value |
|
|
Age (years), mean (+SD) |
61.8 (11.6) |
66.5 (9.3) |
0.3138 |
|
Male gender (%) |
N=5 (45) |
N=4 (36) |
|
|
Body Mass Index (+SD) |
29.6 (7.5) |
26.3 (6.9) |
0.2933 |
|
Medical history |
|||
|
Smokers (%) |
N=3 (27) |
N=3 (27) |
|
|
Alcoholusers above recommended (%) |
N=2 (18) |
N=1 (9) |
|
|
Hypertension* (%) |
N=7 (64) |
N=10 (91) |
|
|
On antihypertensive drugs (%) |
N=5 (45) |
N=8 (73) |
|
|
Atrial fibrillation (%) |
N=1 (9) |
N=1 (9) |
|
|
Type 2 Diabetes Mellitus (%) |
N=0 (0) |
N=1 (9) |
|
|
Haematoma characteristics |
|||
|
ICH volume in ICU (ml), mean (+SD) |
41.9 (21.1) |
34.2 (35.4) |
0.5407 |
|
Initial GCS, mean (+SD) |
12.0 (1.9) |
12.2 (2.2) |
0.7585 |
|
Midline shift (mm), mean )+SD) |
5.5 (3.4) |
4.5 (3.9) |
0.5301 |
|
Basal ganglia haemorrhage, right-sided (%) |
N=7 (64) |
N=4 (36) |
|
|
Intraventricular extension (%) |
N=6 (55) |
N=7 (64) |
*Patients with diagnosed hypertension before haemorrhage
Table 1: Patients general characteristics and haematoma characteristics, numeric data in the table were measured using student’s t-test.
Outcome
30-day mortality was 18% (N=2) in the surgical group and 36% (N=4) in the non-surgical group (RR, 0.5; 95% [CI], 0.11 to 2.19; P=0.2433), see table 2.
|
Surgical group (N=11) |
Non-surgical group (N=11) |
||
|
Mean follow-up time (days), min-max (days) |
382 (234-581) |
672 (202-1146) |
|
|
Mean modified Rankin Scale score |
3.82 |
4.45 |
P=0.3761 |
|
30-day mortality (%) |
18 (N=2) |
36 (N=4) |
RR=0.5; 95% [CI],[0.11;2.19] |
|
overall mortality |
18 (N=2) |
45 (N=5) |
RR=04; 95% [CI],[0.10;1.64] |
Table 2: Follow-up time, mean modified Rankin Scale scores, and mortality for the surgical and non-surgical groups, differences in mortality between surgical and non-surgical groups were assessed using chi-squared test.
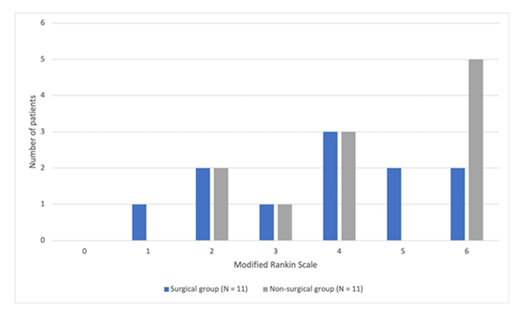
The mean mRS was 3.82 (range 1 to 6) in the surgical group and 4.45 (range 2 to 6) in the non-surgical group, P=0.3761. The distribution of the mRS scores is presented in figure 4.
Among surviving patients at follow-up, the mean mRS was 3.33 in the surgical group (N=9) and 3.17 (P=0.8066) in the non-surgical group (N=6).
Overall mortality was 18% (N=2) in the surgical group and 45% (N=5) in the non-surgical group (RR, 0.4; 95% [CI], 0.10 to 1.64; P=0.1490) (Table 2). The additional patient included in the non-surgical group for analysis of overall mortality had died from unknown cause four months after ICH onset.
Discussion
In this retrospective study of patients with deep spontaneous intracranial haemorrhage, we aimed to evaluate the effect of minimally invasive surgery using the Artemis neuro evacuation device compared to non-surgical treatment. There was a tendency for MIS with the Artemis neuro evacuation device to reduce 30-day mortality, RR 0.5 (0.11-2.19 95% CI) compared to the non-surgical group who received medical treatment alone. The same tendency was found for functional outcome when all 22 matched patients were included (mean mRS score was 3.82 in the surgical group and 4.45 in the non-surgical group; P=0.3761). However, as two patients in the surgical group and five patients in the non-surgical group died before follow-up, we repeated the analysis for the surviving patients only. This resulted in a mean mRS of 3.33 for the surgical group and 3.17 (P=0.8066) for the non-surgical group. Thus, MIS with the Artemis neuro evacuation device did not appear to improve patients’ chance of gaining independence after a spontaneous ICH.
Matching was performed by comparing the patients first on ICH volume and then on GCS at admission, intraventricular extension, and age. This order was used because these criteria have the highest influence on poor prognosis [19]. In more than one possible match was available, the patients were further matched by BMI, gender, smoking status, alcohol intake, and date of onset. This meant that the two groups were very similar (Table 1), but it still tended to favour the non-surgical group as they initially may have had a better prognosis due to smaller ICH volume.
Apart from one patient who had a notably larger ICH volume than any of the other patients in the study, the non-surgical group had smaller ICH volumes than the surgical group. Thus, one could argue there is a baseline bias where the non-surgical group initially have a more favorable prognosis [19,20].
The original intention was to match patients 1:2 to increase the power of the study [21]. This was not possible due to the limited number of ICH patients who met the inclusion and exclusion criteria, however, so we used 1:1 matching instead. This meant that 11 of the 16 non-surgical patients were included in the non-surgical group and 5 non-surgical patients were excluded from the analyses.
3D-rendering with a neuro navigation workstation was used to measure the ICH volume for all included patients in the study. The 3D-rendering technique provides a more accurate measure of the actual ICH volume compared to the frequently used ABC/2 method [22]. Use of the 3D method allowed us to better compare the ICH volumes in the two groups.
Mean follow-up time was 382 days for the surgical group and 672 days for the non-surgical group. This difference in follow-up time is considerable and may have led to favouring of one of the two groups. There is no certainty about the time for recovery in patients who have had an ICH [23]. All the study patients were offered rehabilitation services after discharge, giving the non-surgical group an advantage at follow-up as they had had more time for recovery. A second follow-up assessment of the surgical group could be performed at a later stage to make the follow-up times more similar.
While the notable difference in follow-up time leads to inconclusive results for the mRS scores due to risk of bias, it does not change the validity of the results on 30-day mortality. This study demonstrated a tendency toward reduced mortality for the surgical group, but a larger sample size is needed to show statistical significance. Our results are supported by the meta-analysis by Scaggiante et al. who found significantly reduced odds ratios for death comparing MIS with other treatments [24].
The MISTIE III trial found that all-cause mortality in patients receiving MIS with thrombolysis was similar to that for standard medical care alone [25]. This technique differs from MIS using the Artemis procedure by using thrombolytics to induce passive drainage. This requires a longer time to drain the haematoma and also requires a large number of follow-up CT scans [25]. The Artemis procedure may have an advantage over the MISTIE technique due to the shorter time needed to drain the haematoma, fewer follow-up CT scans, and no requirement for thrombolytics.
We believe this is the first comparative study of the effect of the Artemis neuro evacuation device versus standard medical care alone. Other studies have demonstrated the safety and effect of the Apollo-system (first generation of Artemis) in a case series on cadaver models [26] demonstrating the technique and in human case series [27,28]. Spiotta et al. reported an in-hospital mortality of 13.8% in 29 patients with ICH. Of their 14 patients with basal ganglia haemorrhage, two died [28]. A comparative case control study demonstrated a preliminary trend indicating an absolute risk reduction of 28% in in-hospital mortality in patients treated with the Apollo system compared to patients receiving standard medical management alone [29]. These findings are comparable to those in the current study.
A US randomized control trial is currently recruiting patients and intend to examine the effect of the Artemis procedure performed within 72 hours of ictus in patients with supratentorial ICH (ClinicalTrials.gov identifier: NCT03342664). In comparison, the current study evaluated patients treated within 8 hours of ictus. A Dutch multicentre interventional pilot study is also underway in preparation for a phase III study to evaluate the effect of surgery performed with the Apollo and Artemis systems within 8 hours of onset in patients with supratentorial ICH (ClinicalTrials.gov identifier: NCT03608423).
The current study involved 22 patients with spontaneous deep ICH, where 11 patients were in the surgical group and 11 patients were in the non-surgical group. The small sample size may have led to chance findings and the risk of type 2 error, and by coincidence may have favoured one of the two study groups. Thus, a larger sample size is needed.
Conclusion
In conclusion, this retrospective study of 22 matched patients with deep supratentorial ICH found a non-significant tendency for minimally invasive surgery using the Artemis neuro evacuation device in conjunction with neuro navigation to reduce 30-day mortality when compared to medical treatment alone. A larger randomized controlled trial is needed to demonstrate the full potential of this new haematoma evacuation technique.
Acknowledgments
A special thanks to Sonia Holmegaard Brutti, an indispensable scientific secretary at neurosurgical department in Odense University Hospital. You have been a great support and we applaud your work in providing sufficient data for the research.
Declarations
Ethical approval
This study was approved by the regional ethical committee and data protection authority (j.nr: 21/49624).
Competing interests
The authors have no competing interests to declare that are relevant to the content of this article.
The authors declare that they have no conflict of interest.
Authors’ contribution
The authors confirm contribution to the paper as follows: study conception and design: Kasper R. Jepsen, Malthe T. Stephansen, Christian Bonde, Frantz R. Poulsen, Sune Munthe, Stephanie Binzer; data collection: Kasper R. Jepsen, Malthe T. Stephansen; analysis and interpretation of results: Kasper R. Jepsen, Malthe T. Stephansen; draft manuscript preparation: Kasper R. Jepsen, Malthe T. Stephansen Surgical procedures; Sune Munthe.
All authors reviewed the results and approved the final version of the manuscript.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Availability of data and materials
The data that supports this article cannot be shared openly due to protection of the study participants privacy. The data is kept in an online database at the neurosurgical department in Odense University Hospital, Denmark.
References
- Sahni R, Weinberger J. Management of intracerebral hemorrhage. Vasc Health Risk Manag 3 (2007): 701-709.
- Krishnamurthi RV, Feigin VL, Forouzanfar MH, et al. Global and regional burden of first-ever ischaemic and haemorrhagic stroke during 1990-2010: findings from the Global Burden of Disease Study 2010. Lancet Glob Health 1 (2013): e259-281.
- Dennis MS. Outcome after brain haemorrhage. Cerebrovasc Dis 1 (2003): 9-13.
- Van Asch CJ, Luitse MJ, Rinkel GJ, et al. Incidence, case fatality, and functional outcome of intracerebral haemorrhage over time, according to age, sex, and ethnic origin: a systematic review and meta-analysis. Lancet Neurol 9 (2010): 167-176.
- Langhorne P, Fearon P, Ronning OM, et al. Stroke unit care benefits patients with intracerebral hemorrhage: systematic review and meta-analysis. Stroke 44 (2013): 3044-3049.
- Schreuder FH, Sato S, Klijn CJ, Anderson CS. Medical management of intracerebral haemorrhage. J Neurol Neurosurg Psychiatry 88 (2017): 76-84.
- Anderson CS, Heeley E, Huang Y, et al. Rapid blood-pressure lowering in patients with acute intracerebral hemorrhage. N Engl J Med 368 (2013): 2355-2365.
- Anderson CS, Huang Y, Wang JG, et al. Intensive blood pressure reduction in acute cerebral haemorrhage trial (INTERACT): a randomised pilot trial. Lancet Neurol 7 (2008): 391-399.
- Qureshi AI, Palesch YY, Martin R, et al. Effect of systolic blood pressure reduction on hematoma expansion, perihematomal edema, and 3-month outcome among patients with intracerebral hemorrhage: results from the antihypertensive treatment of acute cerebral hemorrhage study. Arch Neurol 67 (2010): 570-576.
- De Oliveira Manoel AL. Surgery for spontaneous intracerebral hemorrhage. Crit Care 24 (2020): 45.
- Mendelow AD, Gregson BA, Fernandes HM, et al. Early surgery versus initial conservative treatment in patients with spontaneous supratentorial intracerebral haematomas in the International Surgical Trial in Intracerebral Haemorrhage (STICH): a randomised trial. Lancet 365 (2005): 387-397.
- Mendelow AD, Gregson BA, Rowan EN, et al. Early surgery versus initial conservative treatment in patients with spontaneous supratentorial lobar intracerebral haematomas (STICH II): a randomised trial. Lancet 382 (2013): 397-408.
- Hammond MD, Ai Y, Sansing LH. Gr1+ Macrophages and Dendritic Cells Dominate the Inflammatory Infiltrate 12 Hours After Experimental Intracerebral Hemorrhage. Transl Stroke Res 3 (2021): s125-s31.
- Venkatasubramanian C, Mlynash M, Finley-Caulfield A, et al. Natural history of perihematomal edema after intracerebral hemorrhage measured by serial magnetic resonance imaging. Stroke 42 (2011): 73-80.
- Wang WZ, Jiang B, Liu HM, et al. Minimally invasive craniopuncture therapy vs. conservative treatment for spontaneous intracerebral hemorrhage: results from a randomized clinical trial in China. Int J Stroke 4 (2009): 11-16.
- Hattori N, Katayama Y, Maya Y, et al. Impact of stereotactic hematoma evacuation on activities of daily living during the chronic period following spontaneous putaminal hemorrhage: a randomized study. J Neurosurg 101 (2004): 417-420.
- Moullaali TJ, Wang X, Martin RH, et al. Blood pressure control and clinical outcomes in acute intracerebral haemorrhage: a preplanned pooled analysis of individual participant data. Lancet Neurol 18 (2019): 857-864.
- Pedersen CB, Andersen MS, Poulsen FR, et al. Endoscopic evacuation of deep intracerebral spontaneous haematoma. Ugeskr Laeger 183 (2021).
- Houben R, Schreuder F, Bekelaar KJ, et al. Predicting Prognosis of Intracerebral Hemorrhage (ICH): Performance of ICH Score Is Not Improved by Adding Oral Anticoagulant Use. Front Neurol 9 (2018): 100.
- Broderick JP, Brott TG, Duldner JE, et al. Volume of intracerebral hemorrhage. A powerful and easy-to-use predictor of 30-day mortality. Stroke 24 (1993): 987-993.
- Rassen JA, Shelat AA, Myers J, et al. One-to-many propensity score matching in cohort studies. Pharmacoepidemiol Drug Saf 21 (2012): 69-80.
- Divani AA, Majidi S, Luo X, et al. The ABCs of accurate volumetric measurement of cerebral hematoma. Stroke 42 (2011): 1569-1574.
- Saulle MF, Schambra HM. Recovery and Rehabilitation after Intracerebral Hemorrhage. Semin Neurol 36 (2016): 306-312.
- Scaggiante J, Zhang X, Mocco J, et al. Minimally Invasive Surgery for Intracerebral Hemorrhage. Stroke 49 (2018): 2612-2620.
- Hanley DF, Thompson RE, Rosenblum M, et al. Efficacy and safety of minimally invasive surgery with thrombolysis in intracerebral haemorrhage evacuation (MISTIE III): a randomised, controlled, open-label, blinded endpoint phase 3 trial. Lancet 393 (2019): 1021-1032.
- Fiorella D, Arthur A, Schafer S. Minimally invasive cone beam CT-guided evacuation of parenchymal and ventricular hemorrhage using the Apollo system: proof of concept in a cadaver model. J Neurointerv Surg 7 (2015): 569-573.
- Fiorella D, Gutman F, Woo H, et al. Minimally invasive evacuation of parenchymal and ventricular hemorrhage using the Apollo system with simultaneous neuronavigation, neuroendoscopy and active monitoring with cone beam CT. J Neurointerv Surg 7 (2015): 752-757.
- Spiotta AM, Fiorella D, Vargas J, et al. Initial multicenter technical experience with the Apollo device for minimally invasive intracerebral hematoma evacuation. Neurosurgery 11 (2015): 243-251.
- Goyal N, Tsivgoulis G, Malhotra K, et al. Minimally invasive endoscopic hematoma evacuation vs best medical management for spontaneous basal-ganglia intracerebral hemorrhage. J Neurointerv Surg 11 (2019): 579-583.


 Impact Factor:
* 4.2
Impact Factor:
* 4.2
 Acceptance Rate:
72.62%
Acceptance Rate:
72.62%
 Time to first decision: 10.4 days
Time to first decision: 10.4 days
 Time from article received to acceptance:
2-3 weeks
Time from article received to acceptance:
2-3 weeks
